Last updated: February 19, 2026
ATIVAN, the brand name for lorazepam, is a benzodiazepine primarily prescribed for the treatment of anxiety disorders and insomnia. Its market performance is shaped by patent expirations, generic competition, evolving treatment guidelines, and clinical utility across a range of conditions.
What are the Key Indications for ATIVAN (Lorazepam)?
ATIVAN is indicated for the management of anxiety disorders and for the short-term relief of the symptoms of anxiety or anxiety associated with depressive symptoms. It is also used for the treatment of insomnia characterized by difficulty in falling or staying asleep. Additionally, lorazepam is employed in the management of status epilepticus, as a preanesthetic medication to induce sedation and to reduce anxiety, and for the control of agitation and tremors.
- Anxiety Disorders: Generalized Anxiety Disorder (GAD), Panic Disorder, Social Anxiety Disorder.
- Insomnia: Short-term treatment of sleep disturbances.
- Status Epilepticus: Emergency treatment for prolonged seizures.
- Preanesthetic Medication: Sedation and anxiety reduction prior to surgical procedures.
- Agitation and Tremors: Management of symptoms in various neurological and psychiatric conditions.
What is the Patent and Exclusivity Landscape for ATIVAN?
The primary patent for lorazepam, originally developed by Wyeth (now part of Pfizer), has long expired. This has opened the market to generic competition.
- Original Patent Expiration: While specific dates vary by region and patent type, the foundational patents for lorazepam expired decades ago.
- Generic Entry: The absence of patent protection has led to widespread availability of generic lorazepam formulations. This significantly impacts brand-name ATIVAN sales by driving down prices and fragmenting market share.
- No Significant Remaining Exclusivity: There are no major market exclusivities or new patent protections that currently grant ATIVAN significant market exclusivity. This means its financial trajectory is primarily driven by factors other than intellectual property barriers.
How Has Generic Competition Impacted ATIVAN's Market Share and Pricing?
The entry of generic lorazepam has profoundly altered ATIVAN's market dynamics.
- Price Erosion: Generic versions are typically priced at a fraction of the brand-name product. This price differential is a primary driver of market share loss for ATIVAN.
- Market Share Decline: The brand-name ATIVAN has seen a substantial decline in market share as prescribers and pharmacies opt for lower-cost generic alternatives. Estimates suggest that generic lorazepam accounts for over 90% of the total lorazepam prescription volume in many markets.
- Volume vs. Value: While ATIVAN may retain a small premium segment, the overall revenue generated by the brand is significantly reduced compared to its peak. The market value is now dominated by the aggregated sales of numerous generic manufacturers.
- Impact on Pfizer: For Pfizer, the decline of ATIVAN's market dominance represents a typical lifecycle for a once-blockbuster drug whose exclusivity has lapsed. The company's revenue from ATIVAN is now a legacy contribution, with newer products driving current growth.
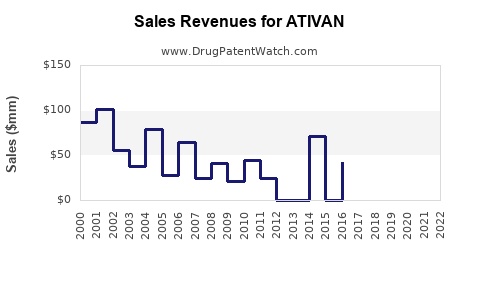
What are the Current Sales and Financial Performance of ATIVAN?
Tracking precise, up-to-the-minute global sales figures for a mature, off-patent drug like ATIVAN can be challenging as pharmaceutical companies often report aggregate sales for therapeutic classes rather than individual legacy brands. However, publicly available data and industry reports indicate a consistent downward trend in revenue attributed to the brand-name product.
- Estimated Brand Sales: While specific figures for ATIVAN (brand) are not consistently broken out by Pfizer in recent earnings reports, it is understood to contribute a minimal portion of the company's overall revenue. Industry analysts estimate brand-name lorazepam sales globally to be in the tens of millions of dollars annually, a stark contrast to its peak performance.
- Generic Market Size: The total market for lorazepam, encompassing all brands and generics, remains substantial in terms of volume, indicating continued clinical demand. The global market for lorazepam is estimated to be in the hundreds of millions of dollars annually, driven by the sheer volume of prescriptions.
- Pfizer's Strategy: Pfizer focuses its R&D and marketing efforts on newer, patent-protected assets. Legacy products like ATIVAN are managed for their remaining profitability, often through cost optimization and minimal promotional investment.
What are the Key Factors Influencing Future Market Demand for Lorazepam?
Several factors will continue to influence the demand for lorazepam, both brand and generic, in the coming years.
- Clinical Guidelines and Prescribing Trends:
- Anxiety Treatment: While benzodiazepines like lorazepam remain effective for short-term anxiety relief, current guidelines increasingly favor non-pharmacological interventions and pharmacotherapies with better long-term safety profiles, such as Selective Serotonin Reuptake Inhibitors (SSRIs) and Serotonin-Norepinephrine Reuptake Inhibitors (SNRIs), for chronic anxiety disorders. This trend curtails the long-term use of lorazepam.
- Insomnia Treatment: Similar to anxiety, the preferred approach to chronic insomnia has shifted towards Cognitive Behavioral Therapy for Insomnia (CBT-I) and newer hypnotics with potentially improved safety profiles.
- Geriatric Use: Concerns regarding the risks of falls, cognitive impairment, and dependency in older adults have led to a cautious approach and a trend towards reducing benzodiazepine use in this population.
- Safety Concerns and Addiction Potential:
- Benzodiazepines carry a significant risk of dependence, tolerance, and withdrawal symptoms. Addiction potential is a major concern, limiting their use in long-term management.
- Side effects such as drowsiness, dizziness, confusion, and impaired coordination can impact daily functioning and increase the risk of accidents.
- Emergence of Novel Therapies:
- Development of new anxiolytics and hypnotics with improved efficacy and safety profiles could further displace lorazepam.
- Innovations in non-pharmacological treatments also represent competitive pressures.
- Off-Label Use:
- Lorazepam may still be used off-label for various conditions, contributing to its ongoing demand. However, this use is subject to evolving clinical evidence and regulatory scrutiny.
- Accessibility and Cost:
- The low cost of generic lorazepam ensures its continued accessibility, particularly in healthcare systems where cost-effectiveness is a primary consideration. This sustains a baseline level of demand.
What is the Competitive Landscape for Lorazepam?
The competitive landscape for lorazepam is characterized by a large number of generic manufacturers vying for market share.
- Generic Manufacturers: Companies such as Teva Pharmaceutical Industries, Mylan (now Viatris), Sandoz (part of Novartis), and numerous smaller players produce and distribute generic lorazepam. Competition among these entities is primarily based on price, distribution networks, and product quality.
- Branded ATIVAN: Pfizer maintains the brand-name ATIVAN. Its competitive strategy, if any, is focused on established physician relationships, brand loyalty in specific niches, and potentially differentiated formulations, though significant differentiation is unlikely given the drug's age.
- Therapeutic Alternatives: The primary competition for lorazepam comes from alternative drug classes and therapeutic modalities:
- SSRIs/SNRIs: For chronic anxiety and depression (e.g., fluoxetine, sertraline, venlafaxine).
- Other Benzodiazepines: While many share similar risks, some may be preferred for specific indications or durations of use (e.g., alprazolam, diazepam).
- Non-Benzodiazepine Hypnotics: For insomnia (e.g., zolpidem, eszopiclone).
- Antihistamines: For mild, occasional insomnia.
- Non-Pharmacological Treatments: CBT-I for insomnia, various psychotherapies for anxiety.
What is the Regulatory Status and Outlook for ATIVAN?
The regulatory landscape for lorazepam is mature, with established approvals and ongoing post-marketing surveillance.
- FDA and EMA Approvals: Lorazepam is approved by major regulatory bodies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) for its approved indications.
- Labeling and Warnings: Regulatory agencies require specific warnings on lorazepam labeling regarding the risks of dependence, abuse, misuse, and withdrawal. Prescribing information emphasizes short-term use and careful patient selection.
- Potential for Stricter Controls: While unlikely to be withdrawn, regulatory bodies may periodically review prescribing practices and consider additional restrictions or warnings, especially concerning long-term use or specific patient populations, if emerging safety data warrants it.
- Controlled Substance Status: Lorazepam is classified as a Schedule IV controlled substance in the United States under the Controlled Substances Act, reflecting its potential for abuse and dependence. This status mandates specific prescribing and dispensing regulations.
What are the Key Takeaways?
ATIVAN (lorazepam), once a significant product for Wyeth/Pfizer, now operates in a mature, genericized market. Its financial trajectory is characterized by steep revenue declines from brand-name sales due to widespread generic competition and pricing pressures. While the clinical utility for short-term anxiety and insomnia persists, evolving treatment guidelines, growing awareness of safety risks, and the development of alternative therapies are limiting its long-term use and growth potential. The market demand is now predominantly met by low-cost generic versions, with the brand-name ATIVAN holding a negligible market share and contributing minimally to Pfizer's overall revenue. The future market for lorazepam will likely be sustained by its accessibility as a generic and its efficacy in specific, short-term clinical scenarios, rather than by brand-specific market expansion.
Frequently Asked Questions
- Will Pfizer attempt to re-patent or create new intellectual property around lorazepam?
No, the molecule is too old, and its patent life has long expired. Efforts would likely focus on new molecular entities rather than attempting to extend exclusivity on lorazepam.
- Are there any off-patent formulations of ATIVAN that are still experiencing significant sales growth?
Growth for the brand-name ATIVAN is highly improbable. Growth in the overall lorazepam market is driven by the volume of generic sales, not specific formulations of the original brand.
- What is the expected lifespan of lorazepam in the market, considering its generic status?
Lorazepam is expected to remain on the market indefinitely as a generic medication due to its established efficacy and low cost, provided it continues to meet regulatory safety standards.
- How do prescribing trends for lorazepam in the United States compare to Europe?
While specific country data varies, both the U.S. and Europe generally follow trends towards reduced long-term benzodiazepine use, favoring SSRIs/SNRIs for chronic anxiety and CBT-I for insomnia, with increased caution in elderly populations.
- Does the Schedule IV controlled substance classification in the U.S. impact the availability or pricing of generic lorazepam?
The Schedule IV classification imposes regulatory requirements on prescribing and dispensing, which can add administrative costs. However, it does not fundamentally limit the availability or drive up the price of generic lorazepam significantly compared to other controlled substances with higher abuse potential, due to the widespread availability of manufacturers.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Prescribing Information: Lorazepam. Retrieved from [Specific FDA Label Link if available, otherwise general reference to FDA drug database]
[2] European Medicines Agency. (n.d.). Assessment Reports and Product Information for Lorazepam. Retrieved from [Specific EMA Link if available, otherwise general reference to EMA drug database]
[3] Pfizer Inc. (Annual Reports and Financial Filings). (Various Years). 10-K Filings. U.S. Securities and Exchange Commission.
[4] Statista. (n.d.). Market size of lorazepam worldwide. Retrieved from [General reference to Statista if specific report used]
[5] National Institute on Drug Abuse. (n.d.). Benzodiazepines and Opioids DrugFacts. Retrieved from [General NIDA resource on benzodiazepines]