ARAZLO Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Arazlo, and what generic alternatives are available?
Arazlo is a drug marketed by Bausch and is included in one NDA. There are two patents protecting this drug and one Paragraph IV challenge.
This drug has twenty-seven patent family members in nineteen countries.
The generic ingredient in ARAZLO is tazarotene. There are eight drug master file entries for this compound. Eleven suppliers are listed for this compound. Additional details are available on the tazarotene profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Arazlo
A generic version of ARAZLO was approved as tazarotene by TARO on April 3rd, 2017.
Summary for ARAZLO
| International Patents: | 27 |
| US Patents: | 2 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 129 |
| Clinical Trials: | 1 |
| Patent Applications: | 4,408 |
| Formulation / Manufacturing: | see details |
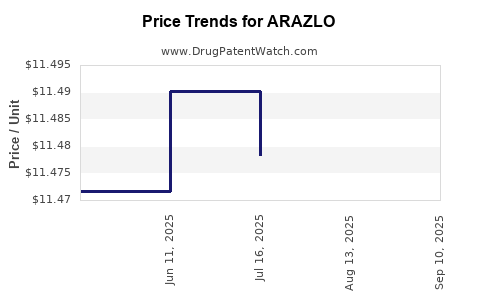
| Drug Prices: | Drug price information for ARAZLO |
| What excipients (inactive ingredients) are in ARAZLO? | ARAZLO excipients list |
| DailyMed Link: | ARAZLO at DailyMed |
Recent Clinical Trials for ARAZLO
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Dr. Emmy Graber | Phase 4 |
Pharmacology for ARAZLO
| Drug Class | Retinoid |
Anatomical Therapeutic Chemical (ATC) Classes for ARAZLO
Paragraph IV (Patent) Challenges for ARAZLO
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| ARAZLO | Topical Lotion | tazarotene | 0.045% | 211882 | 1 | 2022-05-12 |
US Patents and Regulatory Information for ARAZLO
ARAZLO is protected by two US patents.
Patents protecting ARAZLO
Topical compositions and methods for treating skin diseases
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TOPICAL TREATMENT OF ACNE VULGARIS IN PATIENTS 9 YEARS OF AGE AND OLDER
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bausch | ARAZLO | tazarotene | LOTION;TOPICAL | 211882-001 | Dec 18, 2019 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Bausch | ARAZLO | tazarotene | LOTION;TOPICAL | 211882-001 | Dec 18, 2019 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for ARAZLO
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Bausch | ARAZLO | tazarotene | LOTION;TOPICAL | 211882-001 | Dec 18, 2019 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for ARAZLO
See the table below for patents covering ARAZLO around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Denmark | 3310389 | ⤷ Try a Trial | |
| Argentina | 030093 | SISTEMA PARA LA DISTRIBUCION TOPICA DEL GEL | ⤷ Try a Trial |
| Portugal | 2052714 | ⤷ Try a Trial | |
| Slovenia | 1304992 | ⤷ Try a Trial | |
| European Patent Office | 3621614 | COMPOSITIONS TOPIQUES ET MÉTHODES DE TRAITEMENT DE MALADIES CUTANÉS (TOPICAL COMPOSITIONS AND METHODS FOR TREATING SKIN DISEASES) | ⤷ Try a Trial |
| Japan | 4988129 | ⤷ Try a Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for ARAZLO
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1304992 | 474 | Finland | ⤷ Try a Trial | |
| 1304992 | SPC/GB13/061 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: CLINDAMYCIN PHOSPHATE IN COMBINATION WITH TRETINOIN.; REGISTERED: IE PA1332/043/001 20130322; UK PL15142/0249 20130624 |
| 1304992 | 1390049-3 | Sweden | ⤷ Try a Trial | PRODUCT NAME: KLINDAMYCIN (SOM KLINDAMYCINFOSFAT) OCH TRETINOIN; NAT. REG. NO/DATE: MTNR 46193 20130503; FIRST REG.: IR PA1332/043/001 20130322 |
| 1304992 | C300617 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: COMBINATIE VAN CLINDAMYCINE, DESGEWENST IN DE VORM VAN CLINDAMYCINEFOSFAAT EN TRETINOINE; NAT. REGISTRATION NO/DATE: RVG 109745 20130626; FIRST REGISTRATION: PA1332/043/001 20130322 |
| 1304992 | 132013902214376 | Italy | ⤷ Try a Trial | PRODUCT NAME: CLINDAMICINA FOSFATO E TRETINOINA(ACNATAC); AUTHORISATION NUMBER(S) AND DATE(S): PA1332/043/001, 20130322;042056010/M - 022/M, 20130718 |
| 1304992 | C01304992/01 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: CLINDAMYCINI PHOSPHAS ET TRETINOINUM; REGISTRATION NO/DATE: SWISSMEDIC 62513 28.03.2014 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |


