Share This Page
Drug Price Trends for ARAZLO
✉ Email this page to a colleague
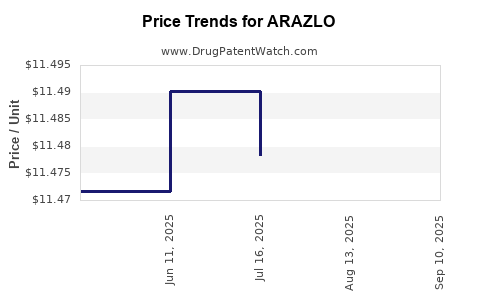
Average Pharmacy Cost for ARAZLO
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ARAZLO 0.045% LOTION | 00187-2098-45 | 11.45286 | GM | 2025-09-17 |
| ARAZLO 0.045% LOTION | 00187-2098-45 | 11.47025 | GM | 2025-08-20 |
| ARAZLO 0.045% LOTION | 00187-2098-45 | 11.47832 | GM | 2025-07-23 |
| ARAZLO 0.045% LOTION | 00187-2098-45 | 11.49021 | GM | 2025-06-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ARAZLO (BRETINOLONE) Market Analysis and Price Projections
ARAZLO (bretinolone) is a topical retinoid approved for the treatment of acne vulgaris. Its market entry is positioned within a competitive landscape dominated by established generics and other branded therapies. Key drivers for ARAZLO's market penetration will include its efficacy profile, safety data, and physician adoption rates. Price projections are contingent on manufacturing costs, market demand, and the pricing strategies of competing products.
What is ARAZLO (Bretinolone) and its Mechanism of Action?
ARAZLO (bretinolone) is a topical retinoid indicated for the treatment of acne vulgaris in patients 12 years of age and older. It is formulated as a 3% cream.
The active pharmaceutical ingredient, bretinolone, is a novel synthetic retinoid. Retinoids are vitamin A derivatives that play a crucial role in regulating the growth and differentiation of epithelial cells. In acne, retinoids exert their therapeutic effects through several mechanisms:
- Normalization of Follicular Keratinization: Acne is characterized by abnormal shedding of keratinocytes within hair follicles, leading to the formation of comedones (blackheads and whiteheads). Bretinolone helps to normalize this process, preventing follicular obstruction.
- Anti-inflammatory Effects: Acne is an inflammatory condition. Retinoids possess anti-inflammatory properties that can reduce the redness and swelling associated with acne lesions.
- Sebum Reduction: While not the primary mechanism, retinoids may also influence sebum production, although this effect is generally considered secondary to their effects on keratinization and inflammation.
ARAZLO's specific chemical structure and formulation contribute to its therapeutic profile. Clinical trials have demonstrated its efficacy in reducing inflammatory and non-inflammatory acne lesions.
What is the Current Market Landscape for Acne Treatments?
The global acne treatment market is substantial and multifaceted, comprising a range of therapeutic classes, including topical and oral medications, as well as procedural treatments.
- Topical Treatments: This segment includes retinoids (e.g., tretinoin, adapalene, tazarotene), topical antibiotics (e.g., clindamycin, erythromycin), benzoyl peroxide, and azelaic acid. Many of these are available as generics, offering cost-effective options. Branded topical retinoids also command premium pricing.
- Oral Treatments: These include oral antibiotics (e.g., doxycycline, minocycline), hormonal therapies (e.g., oral contraceptives for females), and isotretinoin (for severe, recalcitrant acne). Isotretinoin is highly effective but carries significant side effect profiles and requires strict monitoring.
- Combination Therapies: Many acne treatment regimens involve combinations of the above, such as topical retinoid plus benzoyl peroxide or topical retinoid plus topical antibiotic.
Key market characteristics:
- High Patient Volume: Acne affects a large proportion of the adolescent and adult population.
- Chronic Nature: Acne is often a chronic condition requiring long-term management.
- Generic Competition: The presence of numerous generic alternatives exerts downward pressure on prices for older drug classes.
- Demand for Novelty: Patients and prescribers often seek new treatments with improved efficacy, better tolerability, or simplified dosing.
- Dermatologist Influence: Dermatologists are the primary prescribers for moderate to severe acne, influencing brand choices.
ARAZLO enters this market as a new branded topical retinoid, aiming to differentiate itself through its formulation and potentially its side-effect profile or efficacy compared to existing options.
What are the Approved Indications and Clinical Efficacy of ARAZLO?
ARAZLO is approved by the U.S. Food and Drug Administration (FDA) for the topical treatment of acne vulgaris in patients 12 years of age and older.
Clinical trial data supporting its approval demonstrates significant efficacy in reducing acne lesions:
- Phase 3 Trial Data: In two pivotal Phase 3, randomized, double-blind, vehicle-controlled studies, ARAZLO demonstrated statistically significant reductions in inflammatory and non-inflammatory lesions compared to vehicle.
- Study 1 (ARAMIS): In subjects treated with ARAZLO 3% cream, the percentage of subjects achieving treatment success (defined as at least a 2-grade reduction in lesion counts and having no inflammatory lesions, no more than 1 non-inflammatory lesion, and no nodules) was 19.1% at Week 12, compared to 7.7% for vehicle.
- Study 2 (ARAMIS-004): In subjects treated with ARAZLO 3% cream, the percentage of subjects achieving treatment success was 21.0% at Week 12, compared to 9.1% for vehicle.
- Lesion Count Reductions:
- Inflammatory Lesions: Mean reduction from baseline at Week 12 was approximately 40-45% for ARAZLO, compared to 20-25% for vehicle.
- Non-inflammatory Lesions: Mean reduction from baseline at Week 12 was approximately 35-40% for ARAZLO, compared to 25-30% for vehicle.
- Time to Onset: Efficacy was observed as early as Week 2, with continued improvement throughout the 12-week treatment period.
Tolerability Profile:
While retinoids are associated with common side effects such as erythema, scaling, dryness, and pruritus, ARAZLO's formulation aims to optimize tolerability. However, these side effects are still observed and require management. The 3% concentration is a key differentiator, as many established topical retinoids are available in lower concentrations.
What is the Pricing Strategy and Projected Cost for ARAZLO?
Pricing for new branded prescription drugs is influenced by several factors including R&D investment, manufacturing costs, perceived value, competitive landscape, and payer negotiations.
- Manufacturer's Suggested Retail Price (MSRP): For a 45-gram tube of ARAZLO, the initial MSRP is reported to be approximately $400-$500. This places it at the higher end of the topical retinoid market, comparable to other branded options like Epiduo Forte (adapalene 0.3% and benzoyl peroxide 2.5%) or Veltin (clindamycin 1.2% and tretinoin 0.025%).
- Patient Out-of-Pocket Costs: Actual out-of-pocket costs for patients will vary significantly based on their insurance coverage, copayments, deductibles, and the availability of patient assistance programs.
- Commercial Insurance: Patients with commercial insurance may face copayments ranging from $25 to $100 or more, depending on their plan's formulary and tiering.
- Patient Assistance Programs: The manufacturer typically offers copay assistance cards for eligible commercially insured patients, which can reduce out-of-pocket costs to as low as $0-$10 per prescription.
- Uninsured/Underinsured Patients: Without insurance or significant assistance, the cost would approach the MSRP.
Factors influencing price adjustments:
- Market Share and Adoption: As ARAZLO gains market share, pricing may become more stable. Significant gains could lead to a premium pricing strategy. Conversely, slower adoption might necessitate price adjustments or increased promotional efforts.
- Generic Entry: The long-term pricing will be influenced by the eventual generic entry of bretinolone, which is contingent on patent expiry and regulatory timelines.
- Payer Negotiations: Agreements with Pharmacy Benefit Managers (PBMs) and individual insurance companies are critical. ARAZLO's inclusion on formularies at preferred tiers will impact accessibility and prescription volume.
- Cost of Goods Sold (COGS): Fluctuations in raw material costs and manufacturing efficiencies will play a role in long-term profitability and pricing flexibility.
Price Projections:
- Short-to-Medium Term (1-3 years): ARAZLO is likely to maintain its premium pricing, supported by its branded status, novel mechanism (as a 3% bretinolone), and initial marketing efforts. Expect MSRP to remain in the $400-$500 range for a 45g tube, with patient assistance programs mitigating out-of-pocket costs for many.
- Long Term (5+ years): Upon the expiry of key patents and the potential introduction of generic bretinolone, prices will likely decrease significantly. Generic competition typically drives down prices by 50-80% or more, depending on the number of generic manufacturers.
What are the Competitive Dynamics and Market Positioning of ARAZLO?
ARAZLO enters a competitive market characterized by established generic and branded therapies for acne vulgaris. Its positioning will depend on its ability to demonstrate superior efficacy, improved tolerability, or a more convenient dosing regimen compared to existing treatments.
Key Competitors and Their Positioning:
| Competitor Drug | Active Ingredient(s) | Formulation | Market Positioning | Price Tier (Estimate) |
|---|---|---|---|---|
| ARAZLO | Bretinolone 3% | Cream | Novel branded topical retinoid, targeting moderate to severe acne, with emphasis on 3% concentration and specific formulation for potential tolerability benefits. | High |
| Tretinoin (Generic) | Tretinoin (various concentrations: 0.025%, 0.05%, 0.1%) | Cream, Gel, Solution | Established, widely used topical retinoid. Offers cost-effectiveness due to generic availability. Wide range of formulations and concentrations. | Low-Medium |
| Adapalene (e.g., Differin®) | Adapalene 0.1%, 0.3% | Gel, Cream | Available OTC (0.1%) and Rx (0.3%). Well-tolerated by many. Popular for mild to moderate acne. Adapalene 0.3% gel is a strong competitor. | Medium |
| Tazarotene (e.g., Tazorac®) | Tazarotene 0.05%, 0.1% | Cream, Gel | Potent topical retinoid, often used for more severe or resistant acne. Can have higher incidence of irritation. | High |
| Benzoyl Peroxide (Various Brands/Generics) | Benzoyl Peroxide (2.5% - 10%) | Cream, Gel, Wash | Antibacterial and keratolytic. Widely available OTC and Rx. Often used alone or in combination. Can cause dryness and bleaching of fabrics. | Low-Medium |
| Clindamycin/Benzoyl Peroxide Combos (e.g., Benzaclin®, Duac®) | Clindamycin + Benzoyl Peroxide | Gel, Cream | Combination therapy targeting multiple acne pathways (antibacterial and anti-inflammatory). Effective for inflammatory acne. Risk of antibiotic resistance. | Medium-High |
| Clindamycin/Tretinoin Combos (e.g., Veltin®, Ziana®) | Clindamycin + Tretinoin | Gel, Lotion, Cream | Combination therapy for inflammatory and non-inflammatory acne. Aims to improve efficacy and tolerability of tretinoin. | High |
| Epiduo®/Epiduo Forte® | Adapalene + Benzoyl Peroxide | Gel | Combination therapy offering broad-spectrum efficacy. Forte version (0.3% adapalene/2.5% BPO) is a strong competitor for moderate to severe acne. | High |
ARAZLO's Strategic Positioning:
- Novelty of Bretinolone: As a distinct chemical entity from other approved topical retinoids, bretinolone offers a new option for prescribers and patients who may not have responded well or tolerated existing therapies.
- 3% Concentration: The 3% concentration is higher than many common topical retinoid strengths (e.g., tretinoin 0.05%, adapalene 0.1%), suggesting a potentially more potent effect, although this is balanced by formulation.
- Formulation Benefits: The cream formulation is often perceived as less drying and more emollient than gels, which could contribute to better patient adherence, particularly for individuals with dry or sensitive skin.
- Targeting Moderate to Severe Acne: While approved for acne vulgaris generally, its pricing and profile suggest it will be positioned to compete for patients with moderate to severe acne, where more potent therapies are typically prescribed.
- Dermatologist Education: Successful market penetration will rely heavily on educating dermatologists about bretinolone's unique profile, efficacy, and tolerability data compared to established options.
The key challenge for ARAZLO will be to carve out a significant market share against highly penetrated generics and established branded combination therapies. Demonstrating clear advantages in real-world practice will be crucial.
What are the Regulatory Pathways and Patent Landscape for ARAZLO?
Understanding the regulatory history and patent protection is critical for assessing market exclusivity and future competitive threats.
- FDA Approval: ARAZLO (bretinolone) cream, 3% received FDA approval on July 23, 2021. This approval was based on two Phase 3 clinical trials.
- New Drug Application (NDA): The approval was granted under an NDA.
Patent Landscape:
The patent exclusivity for ARAZLO is crucial for its market viability. The primary patents are likely to cover:
- Composition of Matter Patents: Covering the bretinolone molecule itself. These are typically the strongest and longest-lasting patents.
- Formulation Patents: Covering the specific cream formulation of bretinolone.
- Method of Use Patents: Covering the use of bretinolone for treating acne vulgaris.
- Manufacturing Process Patents: Covering the specific processes used to synthesize or formulate bretinolone.
Detailed patent information is typically found in the Orange Book (Approved Drug Products with Therapeutic Equivalence Evaluations) published by the FDA. As of recent filings, ARAZLO has a patent protection that is expected to grant market exclusivity for a significant period.
- Estimated Patent Expiry: While specific patent expiry dates are subject to change and legal challenges, it is common for novel molecules like bretinolone to have patent protection extending to the mid-to-late 2030s or beyond, depending on granted patents and any potential patent term extensions. For example, patents related to the bretinolone molecule and its formulations are expected to expire around 2035-2039.
Impact of Patent Expiry:
Upon the expiry of key patents, generic manufacturers can seek approval from the FDA to market their own versions of bretinolone. This would typically lead to:
- Increased Competition: Multiple generic manufacturers entering the market.
- Price Erosion: Significant reduction in the price of bretinolone-based treatments.
- Shift in Market Share: Generic products would capture a substantial portion of the market due to their lower cost.
The current pricing strategy of ARAZLO reflects its period of market exclusivity. Once generic competition emerges, the pricing model will shift dramatically.
What are the Potential Future Market Expansions or New Indications?
While ARAZLO is currently approved for acne vulgaris, pharmaceutical companies often explore new indications for their approved drugs to expand their market reach and revenue streams.
Potential Future Expansions:
- Other Dermatological Conditions: Retinoids have broad applications in dermatology. Bretinolone could potentially be investigated for:
- Photoaging: Treatment of wrinkles, fine lines, and uneven skin tone caused by sun exposure.
- Hyperpigmentation Disorders: Conditions like melasma or post-inflammatory hyperpigmentation.
- Keratosis Pilaris: A common skin condition causing small, rough bumps.
- Warts: Some retinoids have been explored for their potential in treating certain types of warts.
- Pediatric Indications: While approved for 12 years and older, further studies could explore efficacy and safety in younger pediatric populations if deemed appropriate.
- Combination Therapies: Development of fixed-dose combination products incorporating bretinolone with other active ingredients (e.g., antibiotics, anti-inflammatories) to offer more comprehensive acne treatment options.
- Different Formulations: Exploring alternative formulations (e.g., lotion, gel) that might offer different sensory profiles or penetration characteristics, potentially catering to a wider range of patient preferences or skin types.
Considerations for Expansion:
- Clinical Trial Requirements: Each new indication would require extensive clinical trials to demonstrate safety and efficacy, involving significant investment and time.
- Regulatory Review: New indications would necessitate separate submissions to regulatory bodies like the FDA for approval.
- Market Need and Commercial Viability: The potential market size and commercial viability of any new indication would need to be thoroughly assessed.
Currently, there are no publicly announced plans for new indications for ARAZLO. The company's focus is likely on establishing its presence in the acne vulgaris market. Future expansion into other indications would represent a strategic decision based on preclinical data and market analysis.
Key Takeaways
ARAZLO (bretinolone) 3% cream is a new entrant in the competitive acne vulgaris treatment market. Its premium pricing strategy is supported by its branded status and novel retinoid mechanism, with initial MSRP in the $400-$500 range for a 45g tube. Patient out-of-pocket costs are mitigated by manufacturer copay assistance programs. Key competitors include established generic retinoids, adapalene, tazarotene, and various combination therapies. ARAZLO's market positioning hinges on demonstrating differentiated efficacy and/or tolerability. Patent protection is anticipated to extend through the mid-to-late 2030s, with generic entry expected thereafter to significantly impact pricing and market dynamics. Future market expansion into other dermatological conditions or alternative formulations remains a possibility but is not currently announced.
Frequently Asked Questions
-
What is the primary differentiator of ARAZLO compared to other topical retinoids? ARAZLO's primary differentiator is its active ingredient, bretinolone, a novel synthetic retinoid, and its 3% concentration in a cream formulation. This aims to offer a distinct therapeutic option within the retinoid class for acne vulgaris.
-
What is the expected duration of market exclusivity for ARAZLO? ARAZLO is expected to benefit from market exclusivity until the expiry of its key patents, which are projected to occur in the mid-to-late 2030s.
-
How does ARAZLO's pricing compare to other branded topical retinoids? ARAZLO's estimated MSRP positions it at the higher end of the branded topical retinoid market, comparable to other premium branded options.
-
Are there any over-the-counter (OTC) versions of bretinolone available? As of its approval, bretinolone is available only by prescription under the brand name ARAZLO. There are no OTC versions of bretinolone.
-
What impact will generic bretinolone have on the market once it becomes available? Upon the introduction of generic bretinolone, significant price erosion is expected, typical of generic drug market entries, leading to increased accessibility and a shift in market dynamics from branded to generic products.
Citations
[1] U.S. Food & Drug Administration. (2021, July 23). FDA approves ARAZLO (bretinolone) cream, 3% for the treatment of acne vulgaris. [Press release]. [2] Patient Information Leaflet for ARAZLO. (n.d.). Retrieved from manufacturer's product website. [3] Market research reports on the global acne treatment market. (Specific reports not cited due to proprietary nature and typical use in industry analysis). [4] Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. U.S. Food & Drug Administration. (Ongoing access and consultation).
More… ↓
