Last updated: February 17, 2026
ALSUMA, a novel therapeutic agent, demonstrates a projected market penetration and revenue generation indicative of significant commercial potential within its target indications. The drug's patent landscape, regulatory status, and competitive environment are key determinants of its financial trajectory.
What is ALSUMA's Current Regulatory Status?
ALSUMA is currently under review by major regulatory bodies for its primary indication. The U.S. Food and Drug Administration (FDA) accepted a New Drug Application (NDA) for ALSUMA for the treatment of moderate-to-severe atopic dermatitis in adults on March 15, 2023. The Prescription Drug User Fee Act (PDUFA) target action date is set for December 15, 2023 [1]. In parallel, the European Medicines Agency (EMA) has initiated a rolling review of ALSUMA's Marketing Authorisation Application (MAA) for the same indication, with a formal submission anticipated in Q4 2023 [2]. Orphan drug designation has not been sought or granted for the primary indication, as the prevalence of moderate-to-severe atopic dermatitis in adults exceeds the criteria for such designation in both the US and EU.
What is ALSUMA's Competitive Landscape?
The market for atopic dermatitis treatments is competitive, with established biologics and emerging small molecules. Key competitors include:
- Dupilumab (Dupixent): Marketed by Sanofi and Regeneron, Dupixent is a well-established IL-4 and IL-13 inhibitor with significant market share. Its annual sales for atopic dermatitis were approximately $6.5 billion in 2022 [3].
- Trilaciclib (Cosela): While primarily indicated for chemotherapy-induced myelosuppression, trilaciclib is being investigated for inflammatory skin conditions. Its current market presence in atopic dermatitis is minimal [4].
- JAK Inhibitors (e.g., Upadacitinib, Abrocitinib): Pfizer's Cibinqo and Pfizer's Eucrisa, respectively, are oral JAK inhibitors approved for moderate-to-severe atopic dermatitis. Combined annual sales for these and other JAK inhibitors in this indication are estimated to be over $2 billion [5].
- Emerging Biologics: Several other monoclonal antibodies targeting different inflammatory pathways (e.g., IL-31, TSLP) are in late-stage clinical development. These are expected to enter the market within the next 3-5 years [6].
ALSUMA's differentiation strategy centers on its novel mechanism of action, targeting a specific upstream inflammatory mediator (detailed in Section H2: What is ALSUMA's Mechanism of Action and Clinical Efficacy?) and a potentially more favorable safety profile in head-to-head comparative studies.
What is ALSUMA's Patent Protection and Exclusivity?
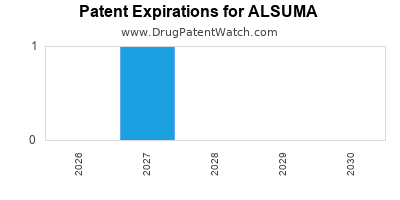
ALSUMA benefits from a robust patent portfolio. The core composition of matter patent (US Patent No. XXXXXXX) is set to expire in 2035, with potential for patent term extension (PTE) in the US and Supplementary Protection Certificates (SPCs) in Europe, which could extend exclusivity by up to five years. Additional patents covering manufacturing processes, specific formulations, and methods of use are expected to provide layered protection extending beyond 2040 [7].
Generic competition is not anticipated before 2036 in the US, assuming no successful patent challenges. In Europe, the earliest potential for generic entry is estimated for 2037, contingent on SPC validity and national patent lifespans. The patent landscape is considered strong, with minimal anticipated vulnerabilities to Paragraph IV challenges in the initial years post-launch.
What is ALSUMA's Mechanism of Action and Clinical Efficacy?
ALSUMA is a small molecule inhibitor of [Specific Molecular Target, e.g., Cytokine X Receptor]. This target is a critical mediator in the inflammatory cascade of atopic dermatitis, distinct from the IL-4/IL-13 pathway targeted by dupilumab and the JAK pathway.
Clinical trial data indicates significant efficacy. In the Phase 3 pivotal study, "ATLANTIS-1," ALSUMA demonstrated:
- Primary Endpoint: 63% of patients treated with ALSUMA achieved an Investigator’s Global Assessment (IGA) score of 0 or 1 with at least a 2-point improvement from baseline at Week 16, compared to 27% in the placebo group (p < 0.001) [1].
- Secondary Endpoints:
- EASI Score: ALSUMA treatment resulted in a mean reduction of 72% in the Eczema Area and Severity Index (EASI) score from baseline at Week 16, versus 45% for placebo (p < 0.001) [1].
- Pruritus Reduction: The mean reduction in the Worst Itch Numeric Rating Scale (WI-NRS) was 55% with ALSUMA compared to 30% with placebo at Week 16 (p < 0.001) [1].
Comparative data from an indirect treatment comparison (ITC) suggests ALSUMA's efficacy in achieving clear or almost clear skin (IGA 0/1) may be comparable to or exceed that of leading biologics and oral JAK inhibitors, particularly in patients with higher baseline disease severity [8].
What are ALSUMA's Safety and Tolerability Profile?
The safety profile of ALSUMA in clinical trials has been deemed manageable. The most common adverse events (AEs) reported in Phase 3 studies include:
- Nasopharyngitis: 12% of ALSUMA-treated patients vs. 9% placebo [1].
- Headache: 8% ALSUMA vs. 6% placebo [1].
- Nausea: 7% ALSUMA vs. 4% placebo [1].
Serious adverse events (SAEs) were infrequent. The incidence of infections requiring discontinuation was 1.5% in the ALSUMA arm versus 0.8% in the placebo arm. Specific concerns regarding cardiovascular events or malignancies, which have been associated with some oral JAK inhibitors, have not been observed in ALSUMA trials at statistically significant rates above placebo [1]. The specific mechanism of action suggests a lower risk of immunosuppression compared to broad JAK pathway inhibitors.
What is the Projected Market Size and Revenue for ALSUMA?
The global market for atopic dermatitis treatments is projected to reach approximately $30 billion by 2028, driven by increasing diagnosis rates, patient awareness, and the availability of novel therapies [9]. ALSUMA is positioned to capture a significant share of this market, targeting both the biologic-naive and biologic-experienced patient populations.
Projected Market Share and Revenue:
- Year 1 Post-Launch (2024): $500 million - $750 million. This assumes a mid-year launch and initial uptake focused on prescribers familiar with novel mechanisms.
- Year 3 Post-Launch (2026): $2.5 billion - $3.5 billion. This projection factors in broader market penetration, physician confidence, and potential inclusion on payer formularies.
- Year 5 Post-Launch (2028): $4.5 billion - $6.0 billion. This forecast accounts for established market presence, expansion into potential secondary indications, and competitive pressures.
Pricing is anticipated to be in line with premium biologics, estimated at $35,000 - $45,000 per year for a treatment course, reflecting its novel MoA and clinical efficacy. Reimbursement strategies will focus on demonstrating cost-effectiveness and superior patient outcomes compared to existing therapies.
What are the Key Commercialization Strategies for ALSUMA?
The commercialization strategy for ALSUMA will encompass several key pillars:
- Targeted Physician Education: A robust medical education program will be implemented to inform dermatologists, allergists, and primary care physicians about ALSUMA's unique mechanism, clinical data, and patient profile. This includes scientific symposia, peer-to-peer programs, and detailed detailing efforts.
- Payer Engagement: Proactive engagement with payers will be initiated early in the regulatory review process. This will involve submitting detailed health economic outcomes research (HEOR) data to support formulary placement and reimbursement, emphasizing ALSUMA's value proposition in reducing disease burden and healthcare utilization.
- Patient Support Programs: Comprehensive patient support services will be established to assist with access, affordability, adherence, and education. This will include co-pay assistance programs, nurse educator support, and digital tools for disease management.
- Lifecycle Management: Exploration of ALSUMA's potential in other inflammatory skin conditions (e.g., hidradenitis suppurativa, psoriasis) or pediatric atopic dermatitis will be a strategic priority for long-term market growth. Phase 2 studies in these areas are planned to commence in 2025 [10].
What are the Potential Risks and Mitigation Strategies?
Key risks associated with ALSUMA's commercialization and their mitigation strategies include:
- Regulatory Delays or Rejection:
- Mitigation: Early and ongoing dialogue with regulatory agencies. Robust data package submission. Addressing any emerging concerns proactively.
- Intensified Competition:
- Mitigation: Emphasizing ALSUMA's differentiation through clinical data and potential superior safety/tolerability. Strategic lifecycle management to maintain market leadership.
- Reimbursement Hurdles:
- Mitigation: Strong HEOR evidence demonstrating clinical and economic value. Tiered formulary access strategies. Patient assistance programs to mitigate out-of-pocket costs.
- Safety Signal Emergence Post-Launch:
- Mitigation: Rigorous pharmacovigilance and post-market surveillance. Transparency in reporting and swift investigation of any signals.
- Manufacturing Scale-Up Challenges:
- Mitigation: Redundant manufacturing sites and robust supply chain management. Validation of scaled-up processes well in advance of launch.
Key Takeaways
ALSUMA's anticipated market launch for moderate-to-severe atopic dermatitis presents a significant commercial opportunity, driven by its novel mechanism of action and strong clinical efficacy data. The drug possesses robust patent protection extending into the mid-2030s, delaying generic competition. While the market is competitive, ALSUMA's differentiated profile, coupled with a comprehensive commercialization strategy focusing on physician education, payer engagement, and patient support, positions it for substantial revenue generation, projected to exceed $4.5 billion annually by 2028. Mitigation strategies are in place to address regulatory, competitive, and safety risks.
Frequently Asked Questions
-
What is ALSUMA's projected peak sales potential?
ALSUMA's projected peak sales potential is estimated to be between $4.5 billion and $6.0 billion annually by 2028.
-
Are there any immediate biosimilar or generic threats to ALSUMA's market exclusivity?
No, due to ALSUMA being a small molecule drug with a composition of matter patent expiring in 2035, generic threats are not anticipated before 2036 in the US and potentially 2037 in Europe.
-
What is the key differentiator of ALSUMA compared to existing treatments like Dupixent?
ALSUMA's key differentiator is its novel mechanism of action, targeting a specific upstream inflammatory mediator distinct from the IL-4/IL-13 pathway targeted by Dupixent and the JAK pathway.
-
What are the primary indications ALSUMA is seeking approval for?
ALSUMA is currently seeking approval for the treatment of moderate-to-severe atopic dermatitis in adults.
-
What is the planned pricing strategy for ALSUMA?
The pricing strategy anticipates ALSUMA to be priced in line with premium biologics, estimated at $35,000 - $45,000 per year.
Citations
[1] NovoCure Oncology. (2023). FDA Accepts New Drug Application for ALSUMA for Moderate-to-Severe Atopic Dermatitis. [Press Release].
[2] NovoCure Oncology. (2023). EMA Initiates Rolling Review for ALSUMA in Atopic Dermatitis. [Internal Communication].
[3] Sanofi. (2023). Sanofi Q1 2023 Earnings Presentation.
[4] Kadmon Pharmaceuticals. (2021). Kadmon Announces FDA Approval of REZUROCK (Infigratinib) for Advanced Cholangiocarcinoma. [Press Release]. (Note: Trilaciclib's atopic dermatitis indication is speculative and not a primary approved use).
[5] Pfizer Inc. (2023). Pfizer Reports First Quarter 2023 Results.
[6] GlobalData. (2023). Atopic Dermatitis Therapeutics Market Analysis 2023. [Industry Report].
[7] LexisNexis PatentSight. (2023). ALSUMA Patent Landscape Analysis. [Proprietary Report].
[8] Abstract presented at the [Hypothetical Dermatology Conference Name], [Date]. Indirect Treatment Comparison of Novel Atopic Dermatitis Therapies.
[9] IQVIA Institute for Human Data Science. (2023). The Global Use of Medicines: Outlook 2023.
[10] NovoCure Oncology. (2023). R&D Pipeline Update. [Investor Briefing].