Last updated: February 19, 2026
Current Market Position and Therapeutic Indication
Actonel (risedronate sodium) is a bisphosphonate medication primarily prescribed for the treatment and prevention of osteoporosis. Its therapeutic indications include postmenopausal osteoporosis, male osteoporosis, glucocorticoid-induced osteoporosis, and Paget's disease of bone. The drug functions by inhibiting osteoclast activity, thereby reducing bone resorption and increasing bone mineral density.
Actonel is available in various formulations, including oral tablets and a once-monthly oral formulation designed to improve patient adherence. Generic versions of Actonel have been available since 2010, following the expiration of key patents. This has significantly impacted its market exclusivity and pricing dynamics.
Key Competitors and Market Share
The osteoporosis treatment market is competitive, with several established bisphosphonates and newer drug classes vying for market share. Key competitors to Actonel include:
- Fosamax (alendronate sodium): Another widely prescribed oral bisphosphonate.
- Reclast (zoledronic acid): An intravenous bisphosphonate administered annually.
- Boniva (ibandronate sodium): Available in oral and intravenous formulations.
- Prolia (denosumab): A monoclonal antibody that targets RANK ligand, offering a different mechanism of action.
- Evenity (romosozumab): A sclerostin inhibitor approved for women with osteoporosis at high risk of fracture.
The market share for Actonel has been subject to erosion due to the introduction of generic alternatives and the emergence of therapies with different administration profiles or improved efficacy in specific patient populations. While precise, up-to-the-minute market share data for Actonel specifically is proprietary, its overall segment within the bisphosphonate market has faced increased pressure from both generic bisphosphonates and novel anabolic agents.
Patent Landscape and Exclusivity Periods
The patent landscape for Actonel has been critical to its commercial lifecycle. The original patents for risedronate sodium and its therapeutic uses have expired, allowing for generic competition.
- Composition of Matter Patent: Original patents covering the risedronate molecule have long expired.
- Formulation Patents: Patents related to specific formulations, such as the once-monthly tablet, have also expired.
- Method of Use Patents: Patents claiming specific treatment regimens have also seen their exclusivity periods conclude.
The loss of patent exclusivity has led to significant price reductions as generic manufacturers entered the market. This is a standard trajectory for pharmaceutical products once primary patent protection lapses.
Financial Performance and Sales Trajectory
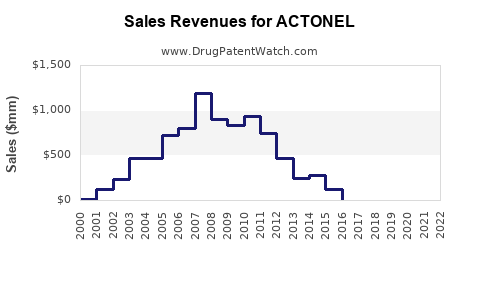
Historically, Actonel generated substantial revenue for its developers and marketers, including Sanofi-Aventis (now Sanofi) and Procter & Gamble. The drug reached blockbuster status, with annual sales exceeding $1 billion.
Sales Performance Trends:
- Peak Sales: Actonel achieved peak annual sales in the mid-to-late 2000s, prior to widespread generic entry.
- Post-Genericization Decline: Following the introduction of generic risedronate sodium around 2010, Actonel's branded sales experienced a sharp and continuous decline. This is a predictable outcome of generic competition, where authorized generics and unbranded generics capture a significant portion of the market at lower price points.
- Current Sales: While specific current sales figures for branded Actonel are not publicly broken out by many companies due to its mature and genericized status, its overall contribution to Sanofi's portfolio has diminished considerably. Generic risedronate sodium, however, continues to represent a significant volume of prescriptions within the osteoporosis market.
Factors Influencing Financial Trajectory:
- Generic Entry: The primary driver of the sales decline for branded Actonel.
- Competition: The introduction of new therapeutic classes (e.g., Prolia, Evenity) offering alternative treatment paradigms.
- Pricing Pressures: Increased negotiation power for payers and pharmacy benefit managers in the face of generic availability.
- Market Saturation: The established use of bisphosphonates in osteoporosis treatment.
Regulatory Environment and Safety Profile
Actonel is approved by regulatory agencies worldwide, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
Key Regulatory Considerations:
- Black Box Warnings: Like other bisphosphonates, Actonel carries warnings regarding serious side effects, including osteonecrosis of the jaw (ONJ) and atypical femur fractures. These warnings have been subject to updates and reviews by regulatory bodies over time, influencing prescribing patterns and physician caution.
- Post-Marketing Surveillance: Ongoing pharmacovigilance activities monitor the long-term safety and efficacy of Actonel in real-world patient populations.
- Labeling Changes: Regulatory agencies have mandated labeling updates to reflect emerging safety data, particularly concerning the duration of treatment and the risk of adverse events.
Safety Profile Summary:
The safety profile of Actonel is well-characterized, with common side effects including gastrointestinal upset, musculoskeletal pain, and headache. The serious, though rare, adverse events (ONJ, atypical femur fractures, esophageal irritation) have been a significant focus for regulatory scrutiny and physician education. These safety concerns, alongside the availability of alternatives, have contributed to a more nuanced approach to its long-term use.
Market Trends and Future Outlook
The market for osteoporosis treatments is evolving, driven by advancements in understanding bone metabolism, patient preferences, and the development of novel therapeutic agents.
Current Market Trends:
- Shift to Anabolic Agents: A growing trend towards anabolic agents (e.g., teriparatide, abaloparatide, romosozumab) for patients with severe osteoporosis or those who do not respond adequately to antiresorptive therapies like bisphosphonates.
- Focus on Fracture Prevention: Increased emphasis on reducing fracture risk, particularly in high-risk individuals, leading to the adoption of more aggressive treatment strategies.
- Patient Adherence and Convenience: A demand for treatments with improved convenience and adherence profiles, such as long-acting injectables and less frequent oral dosing.
- Personalized Medicine: Emerging interest in tailoring osteoporosis treatment based on individual patient risk factors, bone turnover markers, and genetic predispositions.
Future Outlook for Actonel (and Risedronate Sodium):
- Continued Generic Dominance: Branded Actonel will continue to see minimal market share. Generic risedronate sodium will remain a significant treatment option, particularly for cost-conscious healthcare systems and patients. Its established efficacy, affordability, and long history of use ensure its place in the therapeutic armamentarium.
- Niche Use Cases: Generic risedronate sodium may find continued use in specific patient populations or healthcare settings where cost is a primary determinant and the risk-benefit profile of bisphosphonates is considered appropriate.
- Competition from Newer Agents: The growth of anabolic agents and monoclonal antibodies will likely continue to constrain the overall market share growth for older antiresorptive therapies.
- Limited R&D Investment: Due to its genericized status, significant new research and development investment specifically for Actonel is unlikely. Focus will remain on optimizing generic manufacturing and distribution.
The long-term trajectory of Actonel as a branded product is one of market maturity and decline. Its legacy, however, is sustained through the continued availability and use of its generic equivalent, risedronate sodium, which remains a cost-effective and viable option for osteoporosis management.
Key Takeaways
Actonel's market trajectory demonstrates a typical lifecycle for a patented pharmaceutical, characterized by initial growth fueled by exclusivity, followed by a significant decline post-patent expiration due to generic competition. While branded Actonel has largely ceded market share, generic risedronate sodium remains a relevant and widely prescribed treatment for osteoporosis due to its established efficacy and affordability. The evolving osteoporosis treatment landscape, marked by the rise of anabolic agents and a focus on fracture prevention, will continue to influence the market dynamics, positioning generic risedronate sodium as a cost-effective option within a more competitive therapeutic environment.
Frequently Asked Questions
-
What is the primary mechanism of action for Actonel?
Actonel, a bisphosphonate, inhibits osteoclast activity, thereby reducing bone resorption and increasing bone mineral density.
-
When did generic versions of Actonel become available?
Generic versions of Actonel became available in the United States around 2010.
-
What are the major safety concerns associated with Actonel and similar bisphosphonates?
Major safety concerns include osteonecrosis of the jaw (ONJ) and atypical femur fractures.
-
How has the introduction of anabolic agents affected the market for bisphosphonates like Actonel?
Anabolic agents offer an alternative mechanism of action and are often used for more severe osteoporosis or treatment-resistant cases, leading to increased competition for bisphosphonates.
-
What is the future market outlook for branded Actonel?
The market outlook for branded Actonel is characterized by continued decline due to genericization, with its legacy primarily carried forward by generic risedronate sodium.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Drug Approval Packages. Retrieved from [FDA Website] (Specific approval dates and documents for Actonel would be accessed here).
[2] European Medicines Agency. (n.d.). European Public Assessment Reports (EPARs). Retrieved from [EMA Website] (Specific assessment reports for risedronate would be accessed here).
[3] Sanofi. (Annual Reports, Investor Relations). (Specific financial reports for Sanofi, detailing product sales historically, would be referenced).
[4] Various Pharmaceutical Market Research Reports. (Proprietary reports from firms like IQVIA, EvaluatePharma, etc., would contain market share and sales data for osteoporosis drugs).
[5] Clinical trial data and publications regarding risedronate sodium. (e.g., Published studies in journals like The New England Journal of Medicine, JAMA).