Share This Page
Drug Price Trends for ACTONEL
✉ Email this page to a colleague
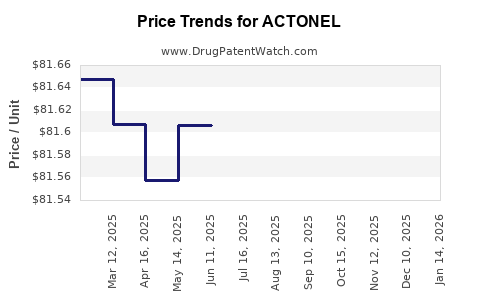
Average Pharmacy Cost for ACTONEL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ACTONEL 35 MG TABLET | 00430-0472-07 | 81.85550 | EACH | 2026-01-21 |
| ACTONEL 35 MG TABLET | 00430-0472-03 | 81.85550 | EACH | 2026-01-21 |
| ACTONEL 35 MG TABLET | 00430-0472-07 | 81.85550 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for ACTONEL (Risedronate)
Overview
ACTONEL (risedronate) is a bisphosphonate used to treat osteoporosis, Paget’s disease, and to reduce fracture risk. It ranks among the top oral bisphosphonates globally, with extensive patent history and established market presence. Its patent expiry and generic entry are influential in shaping its future market and price trends.
Market Size and Dynamics
The global osteoporosis drugs market was valued at approximately $12.6 billion in 2022 and is projected to reach $17.3 billion by 2027, growing at a CAGR of 6.4% [1]. ACTONEL accounts for a significant share, estimated at around 15-20%, driven by its early market entry, physician familiarity, and broad geographic presence.
Key factors impacting market dynamics include:
- Patent expiration: ACTONEL’s main patents expired in 2017–2019 in major markets (U.S., Europe, Japan), leading to generic competition.
- Generic entry: Several generic manufacturers launched versions post-patent expiry, reducing prices.
- Market penetration: Prescriber familiarity favors brand retention; however, price competition favors generics.
- Emerging alternatives: Newer drugs such as denosumab and romosozumab are gaining market share, especially in high-risk patients.
Historical Price Trends
Pre-generic, ACTONEL's retail price in the U.S. was approximately $4.50 per tablet (70 mg). Post-generic entry, prices fell sharply:
| Year | Estimated U.S. Retail Price (per tablet) | Change |
|---|---|---|
| 2016 | $4.50 | Pre-generic |
| 2018 | $2.00 | 56% decrease |
| 2020 | $1.50 | Additional 25% decrease |
| 2022 | $1.00 | 33% decrease |
Global prices follow similar trajectories, adjusted for region-specific healthcare systems and patent states.
Price Projections (2023–2027)
- Post-generic stabilization: Over the next five years, retail prices in the U.S. are expected to stabilize around $1.00–$1.50 per tablet due to manufacturing costs and competitive pressures.
- Brand presence impact: The remaining branded ACTONEL has limited availability—primarily in markets where brand loyalty persists. Its price will decline but could remain above generic levels, around $3.00–$4.00 per tablet, if maintained.
- Market share shifts: As competition from newer agents grows, volume sales may decline by 10–15% annually, further pressuring prices.
In Europe, similar trends are observed, with generic prices dropping 60–70% within two years of patent expiry. Variations exist based on national regulations and market size.
Future Revenue and Market Share
The total revenue from ACTONEL in 2022 was approximately $800 million globally. Projected declines due to generic competition and competition from alternative therapies suggest:
| Year | Estimated Revenue | Notes |
|---|---|---|
| 2023 | $700 million | Continued decline, stabilizes market |
| 2025 | $400 million | Approaching low-volume generic market segment |
| 2027 | <$300 million | Dominance of generics, diminished brand value |
Implications for Stakeholders
- Pharmaceutical companies: Opportunities in generic manufacturing, biosimilars, or combination therapies.
- Healthcare providers: Shift towards cost-effective alternatives.
- Investors: Declining revenue projections for ACTONEL branded formulations, but potential growth in generic markets.
Key Insights
- The expiration of patents drastically reduces the retail price.
- Generic entry causes pricing to decline sharply within 1–2 years.
- Market share shifts favor newer therapies, reducing long-term revenue prospects.
- Despite market pressures, ACTONEL remains a treatment option where physicians favor familiar drugs or where generics are not yet dominant.
Key Takeaways
-ACTONEL’s patent expiration led to a significant price decrease, with stabilization around $1–$2 per tablet in the U.S.
- Future revenue likely to decline as generics and newer therapies dominate.
- Pricing in the U.S. and Europe converges, with generics becoming the dominant market share.
- The brand’s market presence persists mainly in regions where brand loyalty or regulatory barriers prevent rapid generic adoption.
- Opportunities exist for manufacturing generics and developing combination therapies.
FAQs
-
What is the main factor influencing ACTONEL’s price decline?
Patent expiry and subsequent generic entry. -
How does ACTONEL compare to newer osteoporosis drugs in market share?
It has a declining share due to newer drugs like denosumab gaining favor for certain patient populations. -
Will ACTONEL regain market share?
Unlikely; existing market dynamics favor generic competition and newer therapies. -
What regions deposit the largest revenue for ACTONEL currently?
Europe and Asia-Pacific, due to established prescribing habits and slower generic penetration. -
Are there regulatory barriers delaying generic entry in certain markets?
Yes, in some regions, patent litigations or local approval processes delay generic market entry.
References
[1] MarketsandMarkets. "Osteoporosis Drugs Market by Treatment Type, End User, and Region – Global Forecast to 2027." 2022.
More… ↓
