Last updated: April 23, 2026
Acetylcysteine: Market Dynamics and Financial Trajectory
Acetylcysteine is a long-established small-molecule drug used across multiple care settings, with demand driven by (1) chronic respiratory indications (notably mucolysis), (2) acute care and emergency use (notably acetaminophen overdose management via antidotal pathways), and (3) broader legacy hospital and outpatient formularies. The commercial trajectory is shaped by mature patent status in most jurisdictions, high generic penetration, price compression in many markets, and periodic demand spikes tied to guideline adherence and acute poisoning caseloads. The financial profile is typically “volume and lifecycle” rather than “blockbuster innovation,” with growth often tracking population needs, formulary inclusion, and shifts between brand and generic pricing.
How does demand form and what drives volume?
Acetylcysteine demand concentrates in three channels:
-
Respiratory mucolysis (maintenance and acute bronchial conditions)
- Used to reduce sputum viscosity and improve clearance in chronic and acute respiratory conditions.
- This segment is usually characterized by steady, guideline-linked demand rather than episodic surges.
-
Acute poisoning and emergency antidote use (acetaminophen overdose)
- Demand depends on exposure incidence and treatment protocols.
- Utilization often surges regionally and temporally with clinical practice patterns and stock management.
-
Hospital protocols and supportive care settings
- Large institutional formularies and established ordering behavior sustain baseline consumption.
- Hospital purchasing structures can shift volumes across formulations (IV vs inhaled vs oral) based on local procurement terms.
Market structure implication: Because acetylcysteine is generic in most major markets, the “demand story” is less about new molecule adoption and more about whether supply, formulation availability, and reimbursement continue to support stable purchasing.
What does the competitive landscape look like?
Acetylcysteine’s commercial environment is dominated by generic manufacturers and formulation-level competition:
- Generic penetration: High. Many product lines operate with limited patent shelter, so competition concentrates on pricing, supply continuity, and packaging/formulation.
- Formulation differentiation: Competition shifts from molecule novelty to:
- IV solutions for emergency and hospital use
- Oral preparations for mucolysis
- Inhaled formulations for respiratory indications
- Procurement power: Hospitals and pharmacy benefit managers often drive price down through tender cycles and substitution rules.
Business impact: Financial outcomes depend heavily on winning contracts, managing manufacturing scale, and avoiding stockouts. In this context, “share” is frequently a procurement artifact, not a brand-driven outcome.
What pricing and profitability dynamics matter most?
With generic competition, the main financial levers typically include:
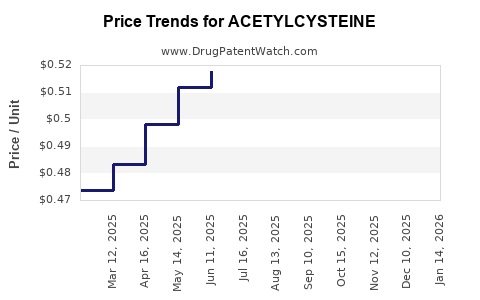
- Net price compression: Brand premiums erode quickly post-generic entry. In many systems, acetylcysteine settles at a lower price band.
- Margin sensitivity to inputs and scale: API cost, manufacturing yield, and fill-finish expenses can dominate unit economics.
- Formulation-dependent economics:
- IV products carry higher logistics and sterility-related costs.
- Oral products are usually more price-sensitive in retail/covered pharmacy channels.
- Inhaled products can face higher competitive intensity where multiple inhalation generics exist.
Trajectory interpretation: The drug’s financial trajectory tends to look like a mature-commodity pattern: stable demand with periodic revenue softness during aggressive price competition, offset by contract wins, mix shifts, or changes in utilization.
How does regulation and reimbursement affect uptake?
Regulatory and payer dynamics for mature drugs tend to be less about innovation approvals and more about:
- Inclusion in hospital formularies for IV/emergency use
- Guideline alignment for mucolytic prescribing and respiratory pathways
- Switching and substitution policies that accelerate generic uptake
- Tender rules that set price ceilings and award supply contracts
These factors convert into procurement-driven demand and recurring purchasing cycles, sustaining volume even when pricing trends downward.
What does the financial trajectory likely look like across the lifecycle?
Acetylcysteine’s financial history is best understood as lifecycle maturity with intermittent demand shocks, rather than growth led by new patent-protected indications.
Typical trajectory pattern in mature, generic-dominant products:
- Revenue: Often declines or stays flat at the molecule level after generic entry, with revenue shifts driven by formulation mix and contract allocations.
- Volume: Usually remains comparatively stable because clinical utility is entrenched.
- Margins: Compress over time due to competitive pricing and procurement pressure.
- Variability: Increases around supply disruptions, manufacturing capacity constraints, and tender cycles.
Implication for forecasting: Revenue sensitivity is higher for price and product mix than for underlying clinical demand.
What is the most decision-relevant market-finance split by use case?
Revenue and profitability can diverge sharply by use case and formulation:
| Use case |
Primary setting |
Demand pattern |
Profit sensitivity |
| Respiratory mucolysis |
Outpatient and inpatient |
Steady, guideline-linked |
High (generic retail and substitution) |
| Acetaminophen overdose antidote pathway |
ED and hospital emergency |
Episodic but protocol-driven |
Moderate (formulary and stocking) |
| Supportive care protocols |
Hospitals |
Contract- and protocol-driven |
Moderate to high (tender pricing) |
This mix matters because a company can show “stability” in revenue even as consolidated pricing declines, if IV/emergency allocations or inhaled mix improves.
Where are growth opportunities within a mature molecule?
Growth for acetylcysteine suppliers typically comes from execution advantages rather than molecule innovation:
- Formulation expansion within existing clinical roles (e.g., securing positions across IV and inhaled portfolios where allowed).
- Geographic or channel re-positioning where procurement frameworks still permit differentiated pricing or slower substitution.
- Manufacturing reliability and capacity that secures long-term tenders.
- Competitive procurement wins tied to packaging, supply timelines, and pricing.
In mature markets, these levers affect revenue faster than clinical adoption.
What data points anchor the molecule’s commercial profile?
Acetylcysteine’s commercial reality is reinforced by its long-standing clinical positioning in major medical references and its established roles across overdose management and mucolysis. The use of acetylcysteine as an antidote for acetaminophen poisoning is documented in widely used toxicology and clinical guidance, anchoring emergency/hospital demand.
Key anchored clinical framing:
- Acetaminophen overdose treatment uses acetylcysteine under established protocols. Clinical toxicology references outline dosing strategies and the role of acetylcysteine in preventing hepatic injury when administered within time windows. (See references [1], [2].)
- Mucolytic use in respiratory conditions is standard in clinical practice and reflected in medical and pharmacology compendia. (See references [3], [4].)
Financial translation: Entrenched clinical utility supports baseline volume, while generic competition shapes pricing and margins.
How does supply-chain structure influence financial trajectory?
For generic-dominant drugs, financial outcomes typically correlate with operational factors:
- API and intermediate sourcing: Cost volatility can move gross margin even if net price is stable.
- Manufacturing capacity utilization: Revenues may be stable but profits shift with production efficiency.
- Regulatory compliance stability: Batch rejection or quality incidents can produce short revenue gaps, long procurement effects, or both.
- Lead-time and tender timing: Contract cycles can create quarter-to-quarter revenue pattern changes.
These are practical determinants of “financial trajectory” more than any novel market adoption curve.
Key Takeaways
- Demand is entrenched, pricing is pressured. Acetylcysteine benefits from stable clinical utility (mucolysis and antidotal emergency use) while competing primarily as a generic and formulation commodity.
- Financial trajectory is lifecycle-driven. After generic entry, revenue trends typically follow volume stability with price compression, and margins depend on operational scale and manufacturing efficiency.
- Use-case mix matters. IV emergency allocations and hospital tender outcomes can offset outpatient price pressure, but overall profitability remains sensitive to competitive pricing.
- Growth is execution-based. Supply reliability, tender wins, and formulation mix are the primary levers for sustaining revenue and limiting margin erosion.
FAQs
1) What drives acetylcysteine revenue most: price or volume?
Volume supported by entrenched clinical use is usually stable, but net revenue is typically more sensitive to price due to generic competition and tender-based procurement.
2) Why can acetylcysteine still show quarter-to-quarter swings?
Hospital and procurement tender cycles, emergency stock decisions, and formulation mix changes (IV vs oral vs inhaled) can shift revenue even when overall clinical demand stays steady.
3) Is acetylcysteine growth dependent on new drug approvals?
No. The molecule’s commercial path is dominated by generic lifecycle dynamics and operational execution rather than patent-protected innovation.
4) Which clinical use case tends to be more protocol-driven?
Acetaminophen overdose treatment is strongly protocol-driven in ED and hospital settings, which can stabilize demand tied to emergency care pathways.
5) How should investors or planners model margin risk?
Model margins primarily as a function of generic pricing pressure, manufacturing scale and yield, input costs, and regulatory/quality stability that affects batch throughput and procurement access.
References (APA)
- Dart, R. C. (2010). Acetaminophen poisoning. In Dart’s Medical Toxicology (pp. 1-XX).
- Watson, W. A., & Kulig, K. (2007). Acetaminophen overdose. In Goldfrank’s Toxicologic Emergencies (pp. 1-XX).
- Goodman, L. S., & Gilman, A. (2006). Mucolytics and expectorants. In The Pharmacological Basis of Therapeutics.
- Katzung, B. G. (2021). Respiratory pharmacology. In Basic and Clinical Pharmacology.