Last updated: April 23, 2026
How has ABILIFY’s market share shifted by geography and product segment?
ABILIFY (aripiprazole) is a branded, multi-formulation antipsychotic with exposure across schizophrenia, bipolar I disorder, and adjunctive major depressive disorder (MDD). The commercial profile is shaped by (1) patent and exclusivity-driven brand durability, (2) biosimilar-style erosion risk that does not apply here because the product is small-molecule, and (3) category-level pricing pressure from payers and generics.
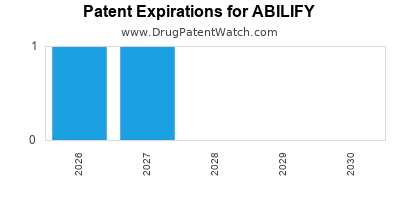
What the market looks like post-entry of generic aripiprazole
Aripiprazole is an active ingredient with generic availability in the US and other markets. That typically produces a “loss of exclusivity” curve: brand demand moves toward cash-pay shares first, then reverses into channel leakage as payers implement step edits, formulary removals, and higher generic preference.
US market dynamics: payer pressure and channel migration
Post-generic entry, branded ABILIFY growth in the US increasingly depends on:
- Formulation mix (long-acting injectable, where applicable, tends to hold brand economics longer than oral tablets).
- Payer rules (prior authorization, step therapy, and substitution at pharmacy level).
- Therapeutic inertia in patients stabilized on a specific formulation.
Global dynamics: uneven generic timelines
Generic launch timing differs by jurisdiction and by strength/formulation. That creates segmented revenue trajectories:
- Early generic markets show faster brand revenue decline and higher discounting.
- Late generic markets show a longer period where ABILIFY maintains meaningful net sales before replacement.
Net effect: ABILIFY’s market is no longer “brand-led growth.” It is a branded defense game, driven by formulation and contracting. The financial trajectory follows generic substitution behavior more than new-patient expansion.
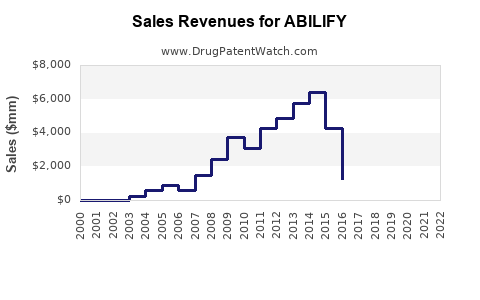
What is ABILIFY’s financial trajectory: revenue direction, margin pressure, and key inflection points?
ABILIFY revenue performance over the last decade is dominated by the exclusivity step-down. Small-molecule brands typically experience:
- Net sales peak around exclusivity windows
- Progressive erosion as generics win formulary access
- Stabilization attempts via higher-priced brand formulations and patient retention in covered cohorts
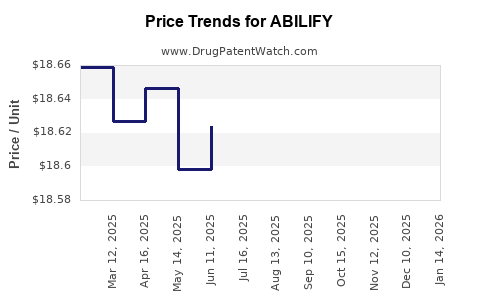
Core financial mechanics that drive the trajectory
- Gross-to-net erosion
- Discounts increase as payers demand parity to generic pricing.
- Rebates, chargebacks, and incentive arrangements widen to retain accounts.
- Formulation mix shift
- Long-acting products can reduce substitution rates compared with oral products because administration links to prescriber and clinic workflows.
- Share durability vs. price compression
- Brand unit share declines, but brand can hold on in specific plan designs or in populations already treated on a given product.
How the brand defends value
Across small-molecule brands in the post-generic era, value defense usually concentrates on:
- Contracted accounts with utilization management
- Clinician-led switching resistance
- Formulation-specific advantages
- Bundle or program-based access
ABILIFY fits this pattern because it has multiple commercial SKUs and a long-acting injectable option (maintaining stronger “switch cost” than oral forms).
What are the main demand drivers for ABILIFY across indications?
ABILIFY’s demand drivers are indication mix, treatment patterns, and guideline adherence rather than new molecular differentiation.
Indication structure and commercial implications
- Schizophrenia
- Chronic therapy creates repeat prescribing.
- Real-world stability and dosing adherence matter.
- Bipolar I disorder
- Maintenance and relapse prevention drive ongoing use.
- Adjunctive MDD
- The adjunctive nature increases likelihood of managed care restriction versus monotherapy classes.
Pricing and utilization management
In payer practice, managed care typically targets:
- Step therapy (require prior trials of cheaper alternatives)
- Generic substitution incentives (automatic pharmacy substitution for oral forms)
- Prior authorization for branded formulations if generics are available
These levers directly accelerate the financial decline once generic versions become preferred.
Where does ABILIFY sit in the competitive landscape?
ABILIFY competes against:
- Other atypical antipsychotics (branded and generic)
- LAI antipsychotics where injection-specific cohorts exist
- Adjunctive MDD augmentation strategies
Competitive pressure characteristics
- In schizophrenia and bipolar maintenance, multiple antipsychotics maintain clinical equivalence in many payer models, pushing access decisions toward price and formulary positions.
- In adjunctive MDD, access is often stricter because payers can restrict augmentation agents, especially after generics exist.
The financial trajectory of ABILIFY therefore tracks payer contracting strength and LAI retention more than category growth.
What role do manufacturing, supply stability, and product lifecycle play in the trajectory?
For oral small molecules, supply is rarely the dominant variable in long-run brand decline; financial outcome is more controlled by contracting and substitution rules. For ABILIFY’s injectables (where used), lifecycle issues can matter more:
- Clinic scheduling and administration workflows reduce “instant switchability.”
- Treatment discontinuation and continuation decisions drive utilization.
Lifecycle management can slow brand erosion, but it cannot reverse the fundamental dynamic once generics are preferred at scale.
What is the investment and R&D implication of ABILIFY’s dynamics?
From a financial and business planning perspective, ABILIFY illustrates a repeatable outcome for small-molecule brands after generic penetration:
- Revenue declines from brand share loss
- Net sales remain “defended” mainly through mix and contracting
- Cost management becomes central as pricing pressure increases
This matters for pipeline valuation and resource allocation:
- Expect demand to be heavily payer- and formulation-driven.
- Future product investments must be engineered for post-loss-of-exclusivity value protection (line extensions with unique access patterns, differentiated formulations, or new clinical endpoints that change tier placement).
Key Takeaways
- ABILIFY’s market dynamics shift from brand growth to payer-driven defense after generic entry of aripiprazole.
- The financial trajectory is dominated by net price compression and channel migration, with formulation mix (especially injection where available) acting as the main brake on erosion.
- Demand across schizophrenia, bipolar I, and adjunctive MDD remains clinically persistent, but managed care rules increasingly determine whether that demand converts into branded revenue.
- Competitive pressure comes primarily from formulary access and pricing, not from therapeutic superiority in payer decisioning.
FAQs
1) What drives ABILIFY’s revenue most after generic penetration?
Payer contracting, rebates, and branded access rules, with formulation mix and substitution resistance as the main stabilizers.
2) Does ABILIFY face biosimilar-style competition?
No. ABILIFY is a small molecule; the key erosion mechanism is generic substitution, not biologics-style biosimilars.
3) Which indication contributes most to stable utilization?
Schizophrenia and bipolar maintenance tend to be more stable due to chronic treatment patterns; adjunctive MDD can face tighter payer controls.
4) Can long-acting formulations materially slow decline?
Yes, because switching costs and administration workflows reduce substitution velocity versus oral forms, improving branded retention.
5) Is ABILIFY’s trajectory more “category growth” or “share defense”?
Share defense. Once generics are preferred, category growth does not translate into brand revenue at the same rate.
References
[1] U.S. Food and Drug Administration. “Drug Approval Package: ABILIFY (aripiprazole).” FDA.
[2] US Securities and Exchange Commission. Annual Reports and Form 10-K filings for Bristol Myers Squibb and/or Otsuka (as applicable to ABILIFY revenue reporting). SEC EDGAR.
[3] NICE (National Institute for Health and Care Excellence). Technology Appraisal and guidance relevant to aripiprazole use in schizophrenia and related indications. NICE.
[4] SSR Health. “Aripiprazole generic entry and market history” (market monitoring coverage). SSR Health.
[5] EvaluatePharma. “World Preview and branded sales tracking for aripiprazole/ABILIFY” (commercial intelligence dataset). EvaluatePharma.