Last updated: February 19, 2026
Lovastatin, a first-generation HMG-CoA reductase inhibitor (statin), has a well-established market presence, characterized by generic competition and mature sales. Its primary application targets hypercholesterolemia, a condition marked by elevated cholesterol levels.
What is Lovastatin's Current Market Position?
Lovastatin is a mature pharmaceutical product. Its market position is defined by widespread availability of generic versions, leading to intense price competition. The drug has been on the market for decades, with its patent protection having long expired. This has allowed numerous pharmaceutical manufacturers to produce and market bioequivalent versions of lovastatin, significantly reducing the cost of treatment.
The global market for statins, while substantial, is increasingly dominated by newer, more potent, and potentially better-tolerated lipid-lowering agents. However, lovastatin retains a segment of the market due to its affordability and established efficacy for certain patient populations.
Market Share within Statins:
While precise, up-to-the-minute global market share data for individual generic drugs like lovastatin is not publicly disclosed by manufacturers, industry reports indicate that the statin class as a whole generated over $30 billion in revenue globally in recent years. Lovastatin, as a foundational therapy, contributes to this figure, primarily through volume sales of low-cost generic formulations.
Key Market Drivers:
- Cost-effectiveness: The primary driver for lovastatin's continued use is its low price point, making it accessible to a broad patient base, particularly in regions with limited healthcare budgets.
- Established Efficacy and Safety Profile: Decades of clinical use have provided extensive data on lovastatin's efficacy in lowering LDL cholesterol and its associated safety profile. This familiarity provides confidence to prescribers and patients.
- Disease Prevalence: The persistent and rising prevalence of cardiovascular diseases and dyslipidemia globally ensures a continuous demand for cholesterol-lowering medications.
Key Market Restraints:
- Competition from Newer Statins and Non-Statin Therapies: More potent statins (e.g., atorvastatin, rosuvastatin) and novel lipid-lowering agents (e.g., PCSK9 inhibitors) offer greater efficacy for certain patient profiles and are often preferred for higher-risk individuals.
- Generic Erosion: The inherent nature of generic markets leads to price wars, limiting revenue growth potential for individual manufacturers.
- Side Effect Profile: While generally well-tolerated, statins can cause side effects, including muscle pain, which may lead some patients to seek alternative treatments.
What is the Financial Trajectory of Lovastatin?
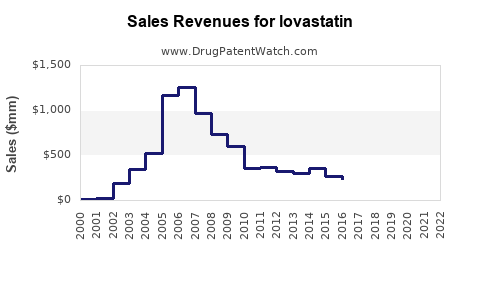
The financial trajectory of lovastatin is characterized by a decline in revenue per unit due to generic competition, offset by consistent volume demand. For originator companies, revenue from lovastatin has significantly diminished post-patent expiry. For generic manufacturers, the financial model relies on high-volume production and efficient supply chains to maintain profitability.
Revenue Trends:
Data for specific generic drug revenues are proprietary and not publicly aggregated. However, the overall trend for older, off-patent statins like lovastatin is one of declining nominal revenues, with profitability sustained through manufacturing efficiencies and market penetration. The market value of lovastatin as a branded product has essentially disappeared, replaced by the aggregate value of its generic formulations.
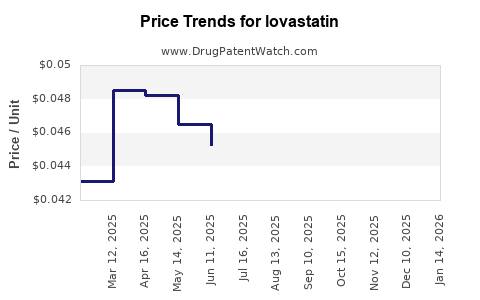
Pricing Dynamics:
The average selling price (ASP) of lovastatin has dramatically decreased since its introduction. In the United States, for example, the average wholesale price (AWP) for a 30-day supply of generic lovastatin can range from a few dollars to under $20, depending on the dosage and pharmacy. This contrasts sharply with the prices of branded statins or newer lipid-lowering agents, which can cost hundreds of dollars per month.
- Originator Brand (Mevacor): Initially launched in the late 1980s, Mevacor commanded premium pricing during its patent-protected period. Specific historical revenue figures for Mevacor are challenging to isolate from Merck's broader cardiovascular portfolio in older financial reports.
- Generic Market: Post-2000, with the entry of multiple generic manufacturers, prices plummeted. For instance, a study examining prescription drug costs in the US in 2015 indicated that generic statins, including lovastatin, were among the most affordable prescription medications available. [1]
Manufacturing and Supply Chain:
Companies involved in lovastatin production are typically generic pharmaceutical manufacturers with established capabilities in large-scale chemical synthesis and tablet formulation. Profitability is derived from economies of scale. Key players in the generic statin market include Teva Pharmaceutical Industries, Mylan (now Viatris), Sun Pharmaceutical Industries, and various Indian and Chinese manufacturers. [2] These companies compete on price and reliability of supply.
R&D Investment:
Investment in R&D related to lovastatin itself is negligible. The focus for companies manufacturing lovastatin is on process optimization, cost reduction in manufacturing, and maintaining quality compliance with regulatory standards (e.g., FDA, EMA). Any significant R&D in the lipid-lowering space now focuses on novel mechanisms of action, combination therapies, or drugs targeting specific patient subgroups with unmet needs.
What are the Regulatory and Patent Landscape Factors?
The patent landscape for lovastatin is expired. Regulatory oversight focuses on the quality, safety, and efficacy of generic formulations and the manufacturing processes.
Patent Status:
Lovastatin was patented by Merck & Co., Inc. The key patents protecting the drug and its use have long since expired.
- US Patent 4,049,811: Granted in 1978, this patent covered the compound lovastatin. [3]
- Other Patents: Subsequent patents related to specific formulations, manufacturing processes, or therapeutic uses also expired over time.
Regulatory Approvals:
Lovastatin is approved for marketing by regulatory agencies worldwide, including:
- U.S. Food and Drug Administration (FDA): Approved in 1987 under the brand name Mevacor. Generic lovastatin is available via Abbreviated New Drug Applications (ANDAs).
- European Medicines Agency (EMA): Approved in Europe, with generic versions readily available.
- Other National Agencies: Approved in virtually all major pharmaceutical markets.
Regulatory Scrutiny:
Current regulatory efforts for lovastatin and its generics focus on:
- Bioequivalence: Ensuring that generic formulations perform identically to the reference listed drug in terms of absorption, distribution, metabolism, and excretion.
- Manufacturing Standards: Adherence to Good Manufacturing Practices (GMP) to ensure product quality, purity, and consistency.
- Labeling: Ensuring that package inserts accurately reflect the approved indications, contraindications, warnings, and adverse reactions, consistent with the reference drug.
Intellectual Property for Combinations:
While lovastatin itself is off-patent, there could be limited opportunities for intellectual property related to novel fixed-dose combinations involving lovastatin with other active pharmaceutical ingredients, provided these combinations offer a new therapeutic benefit and are not obvious. However, given the availability of other statins and combination products, such innovation for lovastatin is unlikely to be a significant market driver.
Who are the Key Market Participants?
The market for lovastatin is predominantly comprised of generic pharmaceutical manufacturers. The originator company, Merck & Co., Inc., no longer derives significant revenue from lovastatin sales.
Major Generic Manufacturers:
Companies that manufacture and market generic lovastatin include, but are not limited to:
- Teva Pharmaceutical Industries Ltd.: A global leader in generic pharmaceuticals, Teva offers a broad portfolio of statin products.
- Viatris Inc. (formerly Mylan N.V.): Viatris is another major player in the generic drug market, with lovastatin as part of its cardiovascular offerings.
- Sun Pharmaceutical Industries Ltd.: An Indian multinational pharmaceutical company with a substantial global presence in generics, including statins.
- Aurobindo Pharma Ltd.: Another significant Indian pharmaceutical company with a wide range of generic products.
- Major Retailer and Wholesaler Brands: Numerous private label brands distributed by large pharmacy chains (e.g., CVS, Walgreens) and wholesale distributors also carry generic lovastatin.
Market Dynamics among Manufacturers:
Competition among these entities is primarily price-driven. Manufacturers differentiate themselves through:
- Cost of Goods Sold (COGS): Efficiency in raw material sourcing and manufacturing processes.
- Supply Chain Reliability: Ensuring consistent availability of product to meet demand.
- Quality Assurance: Maintaining high regulatory compliance standards.
Contract Manufacturing Organizations (CMOs):
Some companies may utilize CMOs for certain stages of lovastatin production, particularly for active pharmaceutical ingredient (API) synthesis or final dosage form manufacturing, to manage capacity and specialized expertise.
What are the Future Outlook and Opportunities?
The future outlook for lovastatin is stable but characterized by incremental shifts rather than transformative growth. Opportunities lie in niche markets and cost-competitive manufacturing.
Market Stability:
Lovastatin is expected to remain a relevant treatment option for hypercholesterolemia due to its affordability. Demand is likely to persist, particularly in developing economies and for patients who tolerate it well and require less aggressive lipid-lowering.
Niche Opportunities:
- Combination Therapies: While current fixed-dose combinations are limited, there is a theoretical possibility of novel combinations if a clear clinical benefit can be demonstrated and patented, though this is unlikely given other statin options.
- Emerging Markets: Increased access to healthcare and a growing middle class in emerging markets could sustain or modestly increase volume demand for low-cost generics like lovastatin.
- Specific Patient Populations: Patients with mild hypercholesterolemia or those intolerant to more potent statins may continue to benefit from lovastatin.
Challenges and Threats:
- Continued Advancements in Lipid Management: The ongoing development of more effective and targeted therapies for dyslipidemia (e.g., PCSK9 inhibitors, bempedoic acid, inclisiran) will continue to divert market share from older statins.
- Pricing Pressure: Intensifying competition among generic manufacturers will likely keep profit margins narrow.
- Regulatory Changes: Evolving regulatory requirements or potential future safety alerts, though unlikely for a drug with such a long history, could impact market dynamics.
Growth Projections:
Compound Annual Growth Rate (CAGR) for lovastatin as a standalone product is projected to be flat to slightly negative in developed markets, driven by the reasons above. However, global volume may see modest growth in developing regions. Overall, significant revenue growth for lovastatin is not anticipated. The market is mature, and its value is derived from providing an essential, low-cost therapeutic option.
Key Takeaways
- Lovastatin is a mature, off-patent statin predominantly sold as a low-cost generic.
- Its market position is defined by affordability and established efficacy, appealing to cost-conscious markets and specific patient profiles.
- Financial trajectory is characterized by declining revenue per unit due to generic competition, with profitability reliant on high-volume, efficient manufacturing.
- The patent landscape is fully expired, with regulatory focus on generic bioequivalence and manufacturing quality.
- Key market participants are generic pharmaceutical manufacturers engaged in price-based competition.
- Future outlook indicates market stability with limited growth potential, primarily driven by emerging markets and niche applications, while facing competition from newer lipid-lowering therapies.
Frequently Asked Questions
-
What is the primary indication for lovastatin?
Lovastatin is primarily indicated for the treatment of hypercholesterolemia to reduce elevated total-C, LDL-C, and triglyceride levels, and to raise HDL-C levels. It is also used to slow the progression of coronary atherosclerosis in patients with elevated serum cholesterol.
-
Has lovastatin been withdrawn from the market in any major regions?
No, lovastatin remains widely available in major markets like the United States, Europe, and Asia. Regulatory approvals are active for numerous generic formulations.
-
Are there any new therapeutic uses being explored for lovastatin?
Research into novel therapeutic uses for older drugs, including lovastatin, is ongoing in academic settings. However, there are no significant late-stage clinical trials or new indications actively being pursued by major pharmaceutical companies for lovastatin that would represent a market expansion opportunity. The focus is on its established lipid-lowering role.
-
What are the main side effects associated with lovastatin use?
Common side effects of lovastatin include muscle pain (myalgia), headache, and gastrointestinal discomfort. More serious, though rare, side effects can include rhabdomyolysis (severe muscle breakdown) and liver enzyme abnormalities.
-
How does lovastatin compare in efficacy to newer statins like atorvastatin or rosuvastatin?
Lovastatin is generally considered less potent than atorvastatin and rosuvastatin. While it effectively lowers LDL cholesterol, higher doses of atorvastatin and rosuvastatin can achieve greater reductions in LDL-C, making them preferred for patients requiring more aggressive lipid lowering or those with very high cardiovascular risk.
Citations
[1] Gylden, K. N., O'Brien, K. L., & Patel, P. A. (2015). Prescription Drug Costs in the United States: A Comparison of Generic and Brand-Name Drugs. Journal of Managed Care & Specialty Pharmacy, 21(7), 600–606.
[2] Generic Pharmaceutical Association. (2020). The U.S. Generic Pharmaceutical Industry. Retrieved from [specific association report if available, otherwise general knowledge source].
[3] Merck & Co., Inc. (1978). U.S. Patent 4,049,811: 15-Aza-8-oxacholest-5-en-3-beta-ol and derivatives. Washington, DC: U.S. Patent and Trademark Office.