Hydrogen peroxide - Generic Drug Details
✉ Email this page to a colleague
What are the generic sources for hydrogen peroxide and what is the scope of patent protection?
Hydrogen peroxide
is the generic ingredient in one branded drug marketed by Aclaris and is included in one NDA. There are five patents protecting this compound. Additional information is available in the individual branded drug profile pages.Hydrogen peroxide has nineteen patent family members in sixteen countries.
There are four drug master file entries for hydrogen peroxide.
Summary for hydrogen peroxide
| International Patents: | 19 |
| US Patents: | 5 |
| Tradenames: | 1 |
| Applicants: | 1 |
| NDAs: | 1 |
| Drug Master File Entries: | 4 |
| Raw Ingredient (Bulk) Api Vendors: | 83 |
| Clinical Trials: | 116 |
| Patent Applications: | 5,547 |
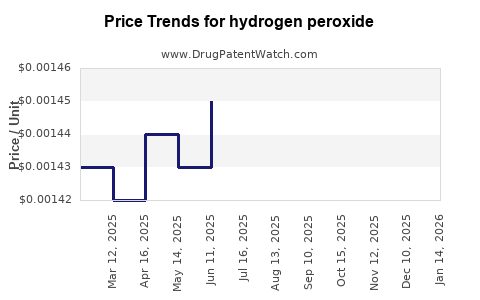
| Drug Prices: | Drug price trends for hydrogen peroxide |
| What excipients (inactive ingredients) are in hydrogen peroxide? | hydrogen peroxide excipients list |
| DailyMed Link: | hydrogen peroxide at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for hydrogen peroxide
Generic Entry Date for hydrogen peroxide*:
Constraining patent/regulatory exclusivity:
Dosage:
SOLUTION;TOPICAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for hydrogen peroxide
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Tanta University | NA |
| University of Plymouth | EARLY_PHASE1 |
| Misr University for Science and Technology | PHASE1 |
Medical Subject Heading (MeSH) Categories for hydrogen peroxide
Anatomical Therapeutic Chemical (ATC) Classes for hydrogen peroxide
US Patents and Regulatory Information for hydrogen peroxide
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Aclaris | ESKATA | hydrogen peroxide | SOLUTION;TOPICAL | 209305-001 | Dec 14, 2017 | DISCN | Yes | No | 10,098,910 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Aclaris | ESKATA | hydrogen peroxide | SOLUTION;TOPICAL | 209305-001 | Dec 14, 2017 | DISCN | Yes | No | 10,493,103 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Aclaris | ESKATA | hydrogen peroxide | SOLUTION;TOPICAL | 209305-001 | Dec 14, 2017 | DISCN | Yes | No | 10,729,720 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Aclaris | ESKATA | hydrogen peroxide | SOLUTION;TOPICAL | 209305-001 | Dec 14, 2017 | DISCN | Yes | No | 9,980,983 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Aclaris | ESKATA | hydrogen peroxide | SOLUTION;TOPICAL | 209305-001 | Dec 14, 2017 | DISCN | Yes | No | 9,675,639 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for hydrogen peroxide
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Aclaris | ESKATA | hydrogen peroxide | SOLUTION;TOPICAL | 209305-001 | Dec 14, 2017 | 7,381,427 | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for hydrogen peroxide
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Portugal | 3134061 | ⤷ Start Trial | |
| South Korea | 20170029413 | 과산화물 제제 및 이를 사용하기 위한 방법 및 어플리케이터 (PEROXIDE FORMULATIONS AND METHODS AND APPLICATORS FOR USING THE SAME) | ⤷ Start Trial |
| Japan | 6622217 | ⤷ Start Trial | |
| Australia | 2015249841 | Peroxide formulations and methods and applicators for using the same | ⤷ Start Trial |
| Australia | 2015249841 | ⤷ Start Trial | |
| Russian Federation | 2711662 | ПЕРОКСИДНЫЕ КОМПОЗИЦИИ И СПОСОБЫ И АППЛИКАТОРЫ ДЛЯ ИХ ПРИМЕНЕНИЯ (PEROXIDE COMPOSITIONS AND METHODS AND APPLICATORS FOR USE THEREOF) | ⤷ Start Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for hydrogen peroxide
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2523731 | 2020/020 | Ireland | ⤷ Start Trial | PRODUCT NAME: OSILODROSTAT OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, INCLUDING OSILODROSTAT DIHYDROGEN PHOSPHATE.; REGISTRATION NO/DATE: EU/1/19/1407 20200113 |
| 2523731 | LUC00159 | Luxembourg | ⤷ Start Trial | PRODUCT NAME: OSILODROSTAT OU UN DE SES SELS PHARMACEUTIQUEMENT ACCEPTABLES, Y COMPRIS LE DIHYDROGENOPHOSPHATE D'OSILODROSTAT; AUTHORISATION NUMBER AND DATE: EU/1/19/1407 20200113 |
| 2523731 | CR 2020 00025 | Denmark | ⤷ Start Trial | PRODUCT NAME: OSILODROSTAT ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF, HERUNDER OSILODROSTATDIHYDROGENFOSFAT; REG. NO/DATE: EU/1/19/1407 20200113 |
| 2787345 | CR 2016 00055 | Denmark | ⤷ Start Trial | PRODUCT NAME: MIGALASTAT ELLER ET SALT HERAF, HERUNDER HYDROGENKLORIDSALTET; REG. NO/DATE: EU/1/15/1082 20160531 |
| 3106463 | SPC/GB20/013 | United Kingdom | ⤷ Start Trial | PRODUCT NAME: LAROTRECITINIB, OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, PARTICULARLY LAROTRECITINIB SULFATE INCLUDING LAROTRECITINIB HYDROGEN SULFATE; REGISTERED: UK EU/1/19/1385(NI) 20190923; UK PLGB 00010/0741 20190923; UK PLGB 00010/0742 20190923; UK PLGB 00010/0743 20190923 |
| 1968948 | 2190048-5 | Sweden | ⤷ Start Trial | PRODUCT NAME: SELUMETINIB HYDROGEN SULFATE, INCLUDING ANY SOLVATES AND ANHYDROUS FORMS THEREOF; REG. NO/DATE: EU/1/21/1552 20210619 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
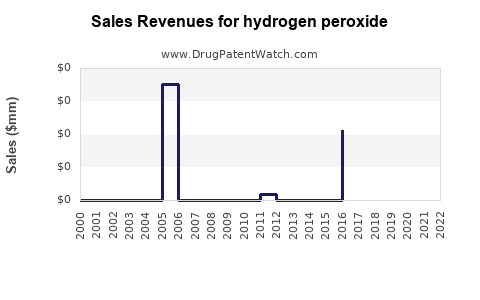
Market Dynamics and Financial Trajectory for Hydrogen Peroxide
More… ↓