Last updated: February 19, 2026
Mirtazapine, an atypical antidepressant, exhibits a mature market position characterized by widespread generic availability and stable demand driven by its established efficacy in treating major depressive disorder. While patent exclusivity has long expired, the drug’s financial trajectory is primarily influenced by generic competition, healthcare reimbursement policies, and the introduction of newer therapeutic alternatives. The global market for mirtazapine is projected to maintain steady growth, underpinned by its cost-effectiveness and continued clinical utility.
What is Mirtazapine and What are its Primary Indications?
Mirtazapine is a tetracyclic antidepressant that functions by enhancing noradrenergic and serotonergic neurotransmission. Its mechanism of action differs from selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs). Mirtazapine selectively antagonizes alpha-2 adrenergic autoreceptors and heteroreceptors, leading to increased norepinephrine and serotonin release. It also blocks postsynaptic 5-HT2 and 5-HT3 receptors, which may contribute to its antidepressant and anxiolytic effects while potentially reducing certain SSRI-related side effects like nausea and sexual dysfunction.
The primary indication for mirtazapine is the treatment of major depressive disorder (MDD) in adults. It is also utilized off-label for other conditions, including:
- Anxiety Disorders: Generalized anxiety disorder (GAD), social anxiety disorder.
- Insomnia: Particularly when associated with depression, due to its sedative properties at lower doses.
- Appetite Stimulation: Mirtazapine can increase appetite and lead to weight gain, making it useful in patients experiencing anorexia secondary to depression or other conditions.
- Post-Traumatic Stress Disorder (PTSD).
- Obsessive-Compulsive Disorder (OCD) (less commonly).
The drug is available in oral formulations, typically as tablets or orally disintegrating tablets. Dosing varies based on the condition being treated and patient response, with typical daily doses ranging from 15 mg to 45 mg.
What is the Patent Landscape for Mirtazapine?
The original patent protection for mirtazapine has long expired. Mirtazapine was first marketed by Organon (now part of Merck & Co.) under the brand name Remeron. The U.S. patent for mirtazapine was issued in the early 1980s, and its exclusivity period concluded by the early 2000s.
The expiration of primary patents opened the door for generic manufacturers to enter the market. Consequently, there are no active patents that prevent the widespread production and sale of generic mirtazapine. The patent landscape is now characterized by:
- Expired Composition of Matter Patents: The fundamental patent protecting the mirtazapine molecule itself has lapsed.
- Expired Formulation Patents: Patents covering specific tablet formulations or orally disintegrating tablets have also expired.
- Limited Remaining Patent Life for Specific Uses or Combinations: While less common for a drug of this age, niche patents related to specific novel delivery systems or combination therapies could theoretically exist but do not represent significant market barriers for the core drug.
The absence of strong patent protection means that the market is dominated by generic competition, significantly impacting pricing and profit margins for manufacturers.
How Has Genericization Affected Mirtazapine's Market Dynamics?
The genericization of mirtazapine has fundamentally altered its market dynamics, shifting the competitive landscape from brand exclusivity to price-based competition.
Key Impacts of Genericization:
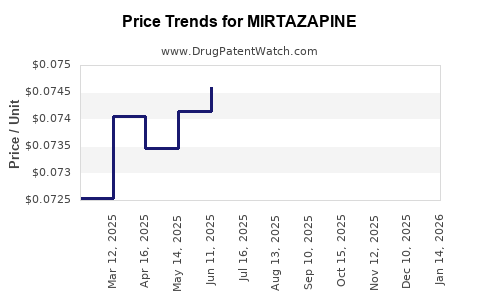
- Price Erosion: Following patent expiration, numerous generic manufacturers entered the market. This surge in competition led to a substantial decrease in the average selling price (ASP) of mirtazapine. The price of generic mirtazapine is a fraction of its original branded price.
- Increased Accessibility: Lower prices have made mirtazapine more affordable and accessible to a broader patient population and healthcare systems globally. This has likely contributed to its continued use.
- Dominance of Generic Market Share: Generic mirtazapine products now constitute the vast majority of the mirtazapine market. Brand-name Remeron has a negligible market share in most developed countries.
- Manufacturer Competition: Competition among generic manufacturers is intense, focusing on cost-efficiency in production, supply chain management, and securing favorable formulary placement with payers.
- Reduced R&D Investment: With the absence of patent protection and significant profit potential for the core molecule, there is minimal incentive for substantial R&D investment in novel mirtazapine formulations or new therapeutic applications by major pharmaceutical companies. The focus has shifted to generic production and optimization.
- Supply Chain Vulnerabilities: The reliance on a large number of generic manufacturers can, in some instances, lead to supply chain disruptions if a significant producer faces manufacturing issues or market withdrawal.
The market for mirtazapine is thus characterized by high-volume sales of a commoditized product where manufacturers compete primarily on cost and reliability of supply.
What is the Current Global Market Size and Projected Growth for Mirtazapine?
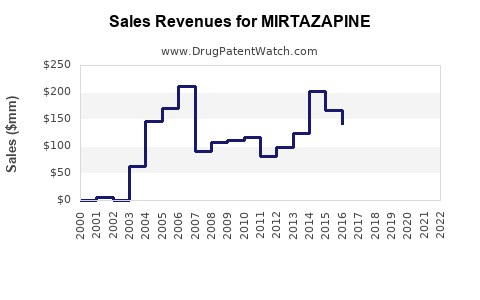
The global market for mirtazapine, driven by its generic availability, is substantial but exhibits moderate growth. Precise market figures are often embedded within broader antidepressant or CNS drug market reports, but industry estimates suggest a market size in the hundreds of millions of U.S. dollars annually.
Market Size and Growth Factors:
- Estimated Market Value: The global market for mirtazapine is estimated to be between $300 million and $500 million annually (as of recent industry reports, typically covering 2022-2023). This figure represents the wholesale value of both branded (minimal) and generic mirtazapine.
- Projected Compound Annual Growth Rate (CAGR): The market is projected to grow at a CAGR of 3% to 5% over the next five to seven years.
- Drivers of Growth:
- Increasing Prevalence of Depression: Global rates of depression and anxiety disorders continue to rise, creating sustained demand for effective treatments.
- Cost-Effectiveness: As a generic drug, mirtazapine offers a highly cost-effective treatment option compared to newer, patented antidepressants, making it a preferred choice in healthcare systems with budget constraints.
- Established Efficacy and Safety Profile: Mirtazapine has a well-documented track record of efficacy and a manageable safety profile, leading to continued physician confidence and prescription.
- Off-Label Use: Continued use for insomnia and anxiety disorders contributes to its overall demand.
- Emerging Markets: Growth in healthcare infrastructure and access to medicines in emerging economies can drive increased utilization of established generics like mirtazapine.
Factors Limiting Growth:
- Competition from Newer Antidepressants: The development of novel antidepressants with potentially improved efficacy, fewer side effects, or different mechanisms of action (e.g., esketamine, psilocybin research) can divert market share.
- Generic Saturation: The market is already highly saturated with generic options, limiting significant price increases or market share gains for individual manufacturers without significant cost advantages.
- Focus on Branded R&D: Pharmaceutical R&D investment is largely directed towards developing novel, patent-protected drugs, rather than reformulating or repositioning older generics.
The financial trajectory for mirtazapine is thus characterized by stable, albeit modest, revenue streams for generic manufacturers, driven by consistent demand rather than innovation-led expansion.
Who are the Key Manufacturers and Suppliers of Mirtazapine?
The mirtazapine market is populated by a large number of generic pharmaceutical manufacturers. The landscape is highly fragmented, with no single entity dominating global supply due to the nature of generic drug production.
Major Generic Manufacturers and Suppliers (examples, not exhaustive):
- Teva Pharmaceutical Industries: A significant global player in generics, Teva manufactures and supplies mirtazapine in various markets.
- Mylan N.V. (now Viatris): Viatris, formed from the merger of Mylan and Pfizer's Upjohn business, is a major producer of generic drugs, including mirtazapine.
- Sun Pharmaceutical Industries Ltd.: An Indian multinational pharmaceutical company with a strong presence in generics, Sun Pharma is a key supplier.
- Dr. Reddy's Laboratories: Another prominent Indian pharmaceutical company that manufactures and distributes generic mirtazapine globally.
- Lupin Limited: A diversified global pharmaceutical company involved in the production of a wide range of generic medications.
- Actavis (now part of AbbVie): While Actavis's generic business has undergone various changes through acquisitions, its legacy operations contribute to the generic mirtazapine supply.
- Various Smaller Regional Manufacturers: Numerous smaller and regional generic drug companies also produce and distribute mirtazapine within their respective territories.
Key Characteristics of Suppliers:
- Global Reach: Many of these manufacturers have a global supply chain, distributing their generic mirtazapine products to numerous countries.
- Cost Optimization: Their competitive advantage lies in efficient manufacturing processes, economies of scale, and aggressive procurement of raw materials.
- Regulatory Compliance: All manufacturers must adhere to stringent regulatory standards set by agencies like the FDA (U.S.), EMA (Europe), and others to ensure product quality and safety.
- Contract Manufacturing: Some larger pharmaceutical companies may also engage contract manufacturing organizations (CMOs) for specific production needs.
The competitive environment among these suppliers is primarily driven by pricing and the ability to secure favorable contracts with wholesalers, distributors, and pharmacy benefit managers.
What are the Regulatory and Reimbursement Considerations for Mirtazapine?
Regulatory and reimbursement policies significantly influence the accessibility and financial viability of mirtazapine. As a well-established generic drug, it faces specific considerations in these domains.
Regulatory Considerations:
- ANDA Approval: Generic versions of mirtazapine require Abbreviated New Drug Application (ANDA) approval from regulatory bodies such as the U.S. Food and Drug Administration (FDA). This process demonstrates bioequivalence to the reference listed drug (Remeron).
- Good Manufacturing Practices (GMP): Manufacturers must consistently meet GMP standards to ensure product quality, purity, and potency. Regular inspections and audits by regulatory agencies are standard.
- Labeling Requirements: Generic labels must accurately reflect the indications, contraindications, warnings, and dosage information of the reference product, with appropriate disclaimers about the manufacturer.
- Pharmacovigilance: Post-market surveillance and reporting of adverse events are mandatory for all approved drug products, including generics.
Reimbursement Considerations:
- Formulary Placement: Mirtazapine is widely available on insurance formularies, typically classified as a preferred or tier-1 generic option due to its low cost. This broad formulary access is a key driver of its consistent prescription volume.
- Cost-Effectiveness Analysis: Healthcare payers and pharmacy benefit managers (PBMs) frequently evaluate drugs based on cost-effectiveness. Mirtazapine generally scores highly in this regard, making it a preferred treatment for major depressive disorder.
- Rebate and Discount Negotiations: Generic manufacturers engage in complex negotiations with PBMs and wholesalers to offer rebates and discounts, influencing their product's inclusion on formularies and its competitive pricing.
- Government Healthcare Programs: Mirtazapine is covered by government healthcare programs (e.g., Medicare, Medicaid in the U.S.; national health services in other countries), further solidifying its market access.
- Prior Authorization: While less common for mirtazapine due to its generic status and broad acceptance, some specific payer plans or treatment guidelines might require prior authorization for higher doses or in specific patient populations, although this is rare for standard indications.
The stable reimbursement landscape for generic mirtazapine ensures its continued place in standard treatment protocols, supporting its ongoing market presence.
What are the Financial Implications for Investors and Pharmaceutical Companies?
For investors and pharmaceutical companies involved with mirtazapine, the financial implications are tied to the dynamics of the generic drug market.
Financial Implications:
- Low Profit Margins: The primary financial characteristic is low per-unit profit margins due to intense generic competition and price erosion. Companies do not achieve the high-margin returns seen with patented novel drugs.
- Volume-Based Revenue: Revenue generation relies on high sales volumes rather than premium pricing. Manufacturers with efficient production, strong distribution networks, and favorable contracts can achieve significant revenue, but profitability is closely managed.
- Stable, Predictable Revenue Streams: For generic manufacturers, mirtazapine represents a stable, predictable revenue stream. Demand is not subject to the rapid obsolescence that can plague innovative drugs if new therapies emerge.
- Investment Focus: Operational Efficiency: Investment for companies focused on mirtazapine is directed towards optimizing manufacturing processes, supply chain logistics, and minimizing production costs to maintain competitiveness.
- Strategic Role in a Diversified Portfolio: For larger generic companies, mirtazapine is often one product among hundreds. Its value lies in contributing to overall revenue and market presence rather than being a standalone growth engine.
- Limited M&A Activity for the Drug Itself: There is limited opportunity for significant mergers or acquisitions specifically for mirtazapine patents or brands, as these are largely expired and the market is commoditized. Acquisitions would focus on acquiring manufacturing capabilities or distribution networks.
- No Significant R&D Return Potential: Investors should not expect significant returns from R&D related to mirtazapine itself. Investment in this area is minimal for established generics.
- Risk Factors: Financial risks include potential supply chain disruptions, unexpected increases in raw material costs, intense price wars among generic competitors, and regulatory changes impacting drug pricing or reimbursement.
Pharmaceutical companies and investors primarily view mirtazapine as a reliable contributor to a diversified generic portfolio, offering steady, albeit modest, financial returns.
Key Takeaways
- Mirtazapine is a well-established generic antidepressant with stable global demand driven by its cost-effectiveness and clinical utility for major depressive disorder.
- The drug's patent exclusivity has expired, leading to a market dominated by numerous generic manufacturers and significant price erosion.
- The global mirtazapine market is valued between $300 million and $500 million annually, projected to grow at a CAGR of 3-5%, fueled by increasing mental health prevalence and affordability.
- Key manufacturers are large generic pharmaceutical companies focused on operational efficiency and competitive pricing, including Teva, Viatris, Sun Pharma, and Dr. Reddy's.
- Regulatory approval is through ANDA, and reimbursement is generally favorable due to its classification as a cost-effective generic option on most health formularies.
- Financial implications for companies and investors are characterized by low profit margins, volume-based revenue, and predictable, stable income streams rather than high-growth potential.
Frequently Asked Questions
-
What is the average wholesale price (AWP) range for a standard mirtazapine prescription (e.g., 30-day supply)?
The AWP for a 30-day supply of generic mirtazapine can range from approximately $10 to $50, depending on the dosage, specific generic manufacturer, and geographic market. This is significantly lower than the original branded price, which could exceed $200 per month.
-
Are there any significant emerging clinical trials or research focused on novel applications of mirtazapine?
While there is ongoing research into mental health treatments, significant R&D focused on novel applications or new formulations of mirtazapine itself is minimal. The drug is considered a mature product, and research efforts are largely directed towards newer therapeutic classes or approaches.
-
What are the primary supply chain risks associated with generic mirtazapine?
Primary risks include reliance on a limited number of active pharmaceutical ingredient (API) suppliers, potential manufacturing disruptions by key generic producers, geopolitical instability affecting raw material sourcing, and unexpected quality control issues leading to product recalls.
-
How does mirtazapine's market performance compare to other widely prescribed generic antidepressants like fluoxetine or sertraline?
Mirtazapine's market performance is similar to other established generic antidepressants in that it faces intense price competition and relies on volume. Fluoxetine and sertraline, also widely available generics, operate within comparable market dynamics, with their respective market sizes and growth rates influenced by similar factors of prevalence, cost-effectiveness, and physician preference.
-
What is the typical patent expiration timeline for new antidepressant drugs, and how does this inform the long-term financial outlook for newer medications compared to mirtazapine?
The standard patent protection for new drugs is typically 20 years from the filing date, though effective market exclusivity can be shorter due to R&D and regulatory approval periods. Newer antidepressants, upon patent expiry, will follow a similar trajectory to mirtazapine, experiencing generic entry and subsequent price erosion. However, their initial launch prices and potential for premium revenue during their patent-protected period are significantly higher.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases/orange-book-approved-drug-products-therapeutic-equivalence-evaluations
[2] IQVIA. (various years). Global Pharmaceutical Market Reports. (Proprietary market data, accessed via subscription services).
[3] Various Pharmaceutical Industry Analyst Reports. (various years). Generic Drug Market Analysis and Antidepressant Market Overviews. (Confidential and proprietary market intelligence reports).