Last updated: February 19, 2026
Metoprolol succinate, a beta-adrenergic blocking agent, is a cornerstone therapy for cardiovascular conditions, including hypertension, angina pectoris, and heart failure. Its established efficacy, broad therapeutic window, and cost-effectiveness have solidified its position in the global pharmaceutical market. The market is characterized by a mature originator product and a robust generic landscape, driven by physician prescribing patterns, patient adherence, and formulary placements.
What is the Current Market Size and Growth Projection for Metoprolol Succinate?
The global market for metoprolol succinate is substantial, reflecting its widespread use in managing prevalent cardiovascular diseases. Precise current market size figures are subject to variations based on reporting methodologies and inclusion of regional market data. However, analyses consistently place the market in the multi-billion dollar range.
Projected Growth: The market is anticipated to experience modest, single-digit annual growth. This is primarily driven by an increasing incidence of cardiovascular diseases globally, particularly in aging populations and emerging economies, coupled with the continued preference for metoprolol succinate due to its established profile and affordability.
- Projected Compound Annual Growth Rate (CAGR): Estimates generally range between 2% and 4% for the forecast period. [1] This growth is not fueled by significant new therapeutic indications or dramatic price increases, but rather by volume expansion.
- Key Market Drivers:
- Rising prevalence of hypertension and other cardiovascular disorders.
- Aging global population with increased risk factors.
- Cost-effectiveness of generic metoprolol succinate formulations.
- Inclusion in essential medicines lists by major health organizations.
- Continued physician familiarity and trust in the drug's safety and efficacy.
What is the Competitive Landscape for Metoprolol Succinate?
The competitive landscape for metoprolol succinate is dominated by generic manufacturers. The original innovator product, Toprol-XL, has long been off-patent, paving the way for extensive generic competition. This has led to significant price erosion for the drug substance and finished dosage forms.
Key Players: The market includes a large number of generic pharmaceutical companies, both large multinational corporations and smaller regional players. Key players in the generic space often include companies with strong capabilities in oral solid dosage manufacturing and established distribution networks. Examples of companies that have historically manufactured or marketed metoprolol succinate include:
- AstraZeneca (originator)
- Teva Pharmaceuticals
- Mylan N.V. (now part of Viatris)
- Sun Pharmaceutical Industries
- Cipla
- Dr. Reddy's Laboratories
- Endo International plc
- Mallinckrodt Pharmaceuticals
Competitive Dynamics: Competition is primarily based on price, product quality, regulatory compliance, and supply chain reliability. Companies with efficient manufacturing processes and strong intellectual property (IP) management strategies for specific formulations or delivery systems can maintain a competitive edge. The availability of various dosage strengths (e.g., 25 mg, 50 mg, 100 mg, 200 mg) and extended-release formulations contributes to market segmentation.
What is the Intellectual Property (IP) Status and Patent Expiration Timeline for Metoprolol Succinate?
As metoprolol succinate is a well-established drug, the core composition of matter patents have long expired. The innovator, AstraZeneca, held the primary patents for Toprol-XL.
Patent Expirations:
- Composition of Matter Patents: Expired in the early 2000s.
- Formulation Patents: While some specific extended-release formulations might have had later-expiring patents, these have also largely expired or are in their final years. These patents focused on technologies that controlled drug release over a 24-hour period.
- Manufacturing Process Patents: Specific patented manufacturing processes may still exist, offering a degree of protection for certain manufacturers, but these do not typically prevent other companies from producing the drug using alternative, non-infringing processes.
Current IP Landscape: The current IP landscape for metoprolol succinate is characterized by a lack of significant patent protection on the active pharmaceutical ingredient (API) itself. This has facilitated the widespread entry of generic manufacturers. Any remaining IP is likely to be related to specific advanced drug delivery technologies or novel combination products, which are less common for this mature therapy.
What are the Regulatory Considerations and Approval Pathways for Metoprolol Succinate?
Metoprolol succinate is an established drug with a long history of regulatory approval. The primary regulatory bodies involved are the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and other national health authorities.
Approval Pathways:
- New Drug Application (NDA): The original approval for the innovator product was through an NDA.
- Abbreviated New Drug Application (ANDA): Generic versions are approved through the ANDA pathway in the U.S. This pathway requires demonstrating bioequivalence to the reference listed drug (RLD), Toprol-XL, in terms of rate and extent of absorption.
- Marketing Authorization Application (MAA): In Europe, generic applications are submitted as MAAs.
Key Regulatory Aspects:
- Bioequivalence Studies: Mandatory for all generic approvals.
- Manufacturing Site Inspections: Regulatory agencies conduct rigorous inspections of manufacturing facilities to ensure compliance with Good Manufacturing Practices (GMP).
- Labeling Requirements: Generic drug labels must be the same as the RLD, except for minor differences that do not alter the drug's safety or efficacy.
- Post-Market Surveillance: Ongoing monitoring of drug safety and efficacy after approval.
Regulatory Hurdles for Generics: While the pathway is established, generic manufacturers must navigate the regulatory review process, which can involve scrutiny of bioequivalence data, manufacturing processes, and impurity profiles.
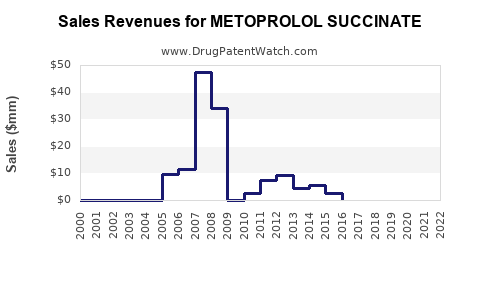
What are the Global Sales and Revenue Trends for Metoprolol Succinate?
The sales and revenue trends for metoprolol succinate are influenced by the dynamic between the declining sales of the originator brand and the aggregate sales of numerous generic products.
Originator Sales (Toprol-XL): Sales for Toprol-XL have significantly declined since patent expiration due to generic competition. While it still garners some revenue, particularly in markets where branded generics are preferred or for specific indications, its market share is minimal compared to the generic segment.
Generic Sales: The global revenue for metoprolol succinate is predominantly generated by generic manufacturers. The total revenue for metoprolol succinate is spread across a multitude of companies.
- Volume Driven: Revenue is primarily driven by the sheer volume of units sold due to its widespread use and affordability.
- Price Competition: Intense price competition among generic manufacturers keeps the average selling price (ASP) per unit low.
- Market Penetration: High market penetration in developed countries and growing penetration in emerging markets contribute to consistent, albeit modest, revenue streams.
Regional Variations: Sales patterns can vary by region due to differences in healthcare systems, prescribing habits, generic substitution policies, and local pricing regulations. Developed markets (North America, Europe) have high generic utilization, while emerging markets show increasing adoption.
What are the Key R&D Opportunities and Challenges for Metoprolol Succinate?
Given the mature nature of metoprolol succinate, R&D opportunities are primarily focused on incremental improvements rather than novel therapeutic applications.
R&D Opportunities:
- Improved Formulations: Development of novel extended-release formulations offering enhanced patient compliance, reduced dosing frequency, or improved pharmacokinetic profiles. This could include technologies for more consistent drug release throughout the day.
- Combination Therapies: Investigating fixed-dose combinations with other cardiovascular agents (e.g., diuretics, ACE inhibitors) to improve patient adherence and therapeutic outcomes. However, regulatory pathways for such combinations can be complex.
- Pediatric Formulations: Development of suitable dosage forms (e.g., liquids, chewables) for pediatric patients, where current options may be limited.
- Manufacturing Process Optimization: Continuous efforts to optimize manufacturing processes for cost reduction, improved yield, and enhanced sustainability.
R&D Challenges:
- Limited Unmet Medical Need: Metoprolol succinate is a well-established and effective treatment, leaving limited scope for breakthrough R&D that addresses significant unmet medical needs.
- High Cost of Development vs. Low Price Point: The low price point of generic metoprolol succinate makes it challenging to justify substantial R&D investments that do not yield significant price premiums or market share gains.
- Regulatory Scrutiny for New Formulations: Even for improved formulations, demonstrating superiority or significant advantage over existing products can be a rigorous regulatory process.
- Intellectual Property Protection: Difficulty in securing robust patent protection for incremental innovations in a crowded generic market.
What is the Future Outlook and Potential Financial Trajectory?
The future financial trajectory of metoprolol succinate is expected to remain stable with modest growth, largely dictated by global health trends and the generic market's dynamics.
Financial Trajectory:
- Stable Revenue Streams: Generic manufacturers will continue to generate stable revenue from metoprolol succinate due to its essential nature and high prescription volume.
- Price Pressure: Ongoing price pressure from market competition will limit significant revenue growth. Profitability will be contingent on manufacturing efficiency and scale.
- Consolidation: The generic industry may see further consolidation, potentially impacting pricing dynamics and market access for some players.
- Emerging Market Growth: Expansion of healthcare access and increasing diagnosis of cardiovascular diseases in emerging economies will contribute to volume growth, offsetting any potential declines in mature markets.
Strategic Considerations for Companies:
- Cost Leadership: Companies that can achieve superior manufacturing efficiency and supply chain management will be best positioned to maintain profitability.
- Portfolio Diversification: For many companies, metoprolol succinate will remain a part of a broader portfolio of generic drugs, contributing to overall revenue stability rather than being a primary growth driver.
- Focus on Quality and Reliability: Consistent product quality and reliable supply are critical for maintaining market share and physician trust.
Key Takeaways
- Metoprolol succinate commands a significant global market share within the cardiovascular therapeutic area, driven by its efficacy and affordability.
- The market is characterized by intense competition from generic manufacturers, leading to price erosion and volume-driven revenue.
- Core patents have expired, making new entrant barriers low and fostering a highly competitive generic landscape.
- Regulatory approval relies on demonstrating bioequivalence to the reference listed drug, with established pathways via ANDA in the U.S.
- Future growth is projected to be modest, fueled by the increasing incidence of cardiovascular diseases and expanding healthcare access in emerging markets.
- R&D opportunities are largely focused on incremental improvements in drug delivery and formulation rather than novel therapeutic applications.
Frequently Asked Questions
-
Are there any new therapeutic indications being explored for metoprolol succinate?
No, significant R&D into new therapeutic indications for metoprolol succinate is not a current focus, as its established indications are well-understood and covered by existing treatments.
-
What is the primary risk for manufacturers of metoprolol succinate?
The primary risk is extreme price erosion due to fierce competition among generic manufacturers, which can compress profit margins.
-
How does the aging global population specifically impact the metoprolol succinate market?
An aging population generally experiences a higher incidence of cardiovascular diseases, such as hypertension and heart failure, which are primary indications for metoprolol succinate, thus increasing demand.
-
Can metoprolol succinate be combined with other drugs without new patent challenges?
While generic combination products can be developed, they must demonstrate bioequivalence and meet regulatory requirements. The patentability of a combination would depend on novel aspects of the formulation or synergistic effects not previously established.
-
What is the typical lifespan of a patent for an extended-release formulation of a generic drug like metoprolol succinate?
Patents for specific extended-release formulations typically last for a defined period after their filing, often around 20 years from the filing date, with potential for extensions. However, the core drug patents for metoprolol succinate expired much earlier.
Citations
[1] Global Market Insights. (n.d.). Metoprolol Succinate Market Analysis Report. (Specific report titles and publication dates vary, but cite general market analysis reports).