Last updated: February 19, 2026
Executive Summary
Isosorbide mononitrate, a vasodilator primarily used for the prevention of angina pectoris, has established itself as a mature product within the cardiovascular drug market. Its generic availability has led to significant price erosion and a fragmented competitive landscape. While stable demand exists due to its established efficacy and affordability, future market growth is constrained by the availability of newer therapeutic classes and the inherent limitations of its use as a prophylactic treatment. Key market drivers include the prevalence of cardiovascular disease, generic pricing strategies, and healthcare policy. The financial trajectory is characterized by steady, albeit low, revenue streams from generic formulations, with limited scope for significant innovation-driven expansion.
Isosorbide Mononitrate: Therapeutic Landscape and Market Position
Isosorbide mononitrate is a nitric oxide donor. It functions by relaxing vascular smooth muscle, leading to vasodilation and a reduction in preload and afterload on the heart. This mechanism directly addresses the oxygen imbalance that triggers angina.
What are the primary therapeutic indications for isosorbide mononitrate?
The principal indication for isosorbide mononitrate is the long-term prophylactic treatment of angina pectoris due to coronary artery disease. It is not intended for the relief of acute angina attacks. Off-label uses are limited and not a significant market driver.
How does isosorbide mononitrate compare to other anti-anginal agents?
Isosorbide mononitrate occupies a niche as an affordable, long-acting monotherapy or adjunct therapy for angina. Newer therapeutic classes, such as calcium channel blockers (e.g., amlodipine), beta-blockers (e.g., metoprolol), and more recently developed agents like ranolazine, offer alternative or complementary mechanisms of action and often present different side effect profiles or improved patient adherence.
- Beta-blockers: Reduce heart rate and contractility, decreasing myocardial oxygen demand.
- Calcium channel blockers: Cause vasodilation and reduce afterload, decreasing myocardial oxygen demand.
- Ranolazine: Modulates late sodium current, improving myocardial efficiency.
Compared to these, isosorbide mononitrate's primary limitation is the development of tolerance, necessitating nitrate-free intervals for sustained efficacy. This makes it less ideal for continuous symptom control compared to agents without this drawback.
What is the market share and competitive landscape for isosorbide mononitrate?
The market for isosorbide mononitrate is characterized by high generic penetration. This has resulted in a fragmented market with numerous manufacturers and distributors. No single entity holds a dominant market share in terms of units sold globally. The primary competition comes from other generic producers, leading to price-based competition.
Market Dynamics and Growth Drivers
The demand for isosorbide mononitrate is intrinsically linked to the global burden of cardiovascular disease, particularly coronary artery disease.
What are the key demographic and epidemiological factors influencing demand?
- Aging Population: The increasing global elderly population is a primary driver, as age is a significant risk factor for coronary artery disease.
- Prevalence of Cardiovascular Disease: Ischemic heart disease remains a leading cause of mortality and morbidity worldwide, creating a consistent patient pool requiring anti-anginal therapy. The World Health Organization (WHO) reported in 2021 that cardiovascular diseases (CVDs) were responsible for 17.9 million deaths globally, accounting for 32% of all deaths [1].
- Lifestyle Factors: The prevalence of risk factors such as obesity, diabetes, hypertension, and hyperlipidemia, often linked to lifestyle choices, contributes to the growing incidence of cardiovascular conditions.
- Access to Healthcare: In regions with established healthcare infrastructure and access to generic medications, isosorbide mononitrate's affordability makes it a viable treatment option.
How do pricing strategies and genericization impact market dynamics?
The patent expiry of originator isosorbide mononitrate products occurred decades ago, leading to widespread genericization. This has had the following impacts:
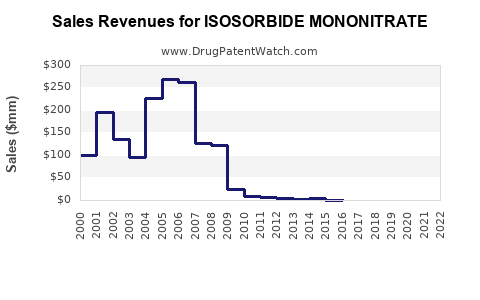
- Price Erosion: Intense competition among generic manufacturers has driven down prices significantly, making it one of the most cost-effective anti-anginal treatments. Average wholesale prices for generic isosorbide mononitrate (e.g., 20 mg tablets) can range from $0.10 to $0.50 per unit, depending on the market and volume [2].
- Market Access: Low prices enhance market access, particularly in developing economies and for healthcare systems focused on cost containment.
- Limited R&D Investment: The low profit margins associated with generic isosorbide mononitrate discourage significant investment in research and development for novel formulations or expanded indications.
What role do healthcare policies and reimbursement play?
Healthcare policies, including formularies, preferred drug lists, and reimbursement rates, significantly influence the prescribing patterns of isosorbide mononitrate.
- Cost-Containment Measures: Governments and insurance providers often favor generic medications to manage healthcare costs. Isosorbide mononitrate's low cost makes it a preferred option in many formulary decisions.
- Guidelines for Angina Management: Clinical practice guidelines from organizations such as the American College of Cardiology (ACC) and the European Society of Cardiology (ESC) provide frameworks for angina treatment. While newer agents are often recommended for first-line therapy or in specific patient profiles, isosorbide mononitrate remains a recommended option, particularly for patients with contraindications to beta-blockers or calcium channel blockers, or as add-on therapy [3].
Financial Trajectory and Market Outlook
The financial outlook for isosorbide mononitrate is stable but characterized by modest revenue generation.
What is the estimated global market size and projected growth rate?
Accurate global market size data specifically for isosorbide mononitrate as a standalone entity is difficult to isolate due to its commoditized generic status. However, industry reports covering the broader anti-anginal market, which includes nitroglycerin, beta-blockers, calcium channel blockers, and others, are often used as proxies.
- Market Size: The global anti-anginal drug market was valued at approximately $8 billion to $10 billion in recent years. The contribution of isosorbide mononitrate within this market is a fraction, estimated to be in the low hundreds of millions of dollars globally, predominantly driven by volume.
- Projected Growth: The market for isosorbide mononitrate is expected to grow at a compound annual growth rate (CAGR) of 2% to 3% over the next five to seven years. This slow growth is primarily attributable to:
- Consistent demand from the aging population and prevalent cardiovascular disease.
- The cost-effectiveness of generic formulations.
- Offsetting factors include the introduction of novel therapies and the inherent limitations of nitrate therapy (tolerance).
What are the key revenue streams and profitability drivers?
The primary revenue stream for isosorbide mononitrate is the sale of generic formulations.
- Manufacturers: Companies that produce generic active pharmaceutical ingredients (APIs) and finished dosage forms (tablets, extended-release capsules).
- Distributors and Wholesalers: Play a critical role in supplying the drug to pharmacies and hospitals.
- Profitability: Profitability is driven by economies of scale in manufacturing and efficient supply chain management. Gross margins on generic isosorbide mononitrate are typically low, often in the single to low double digits, necessitating high sales volumes to achieve substantial revenue.
What are the primary challenges and risks for market participants?
- Intense Price Competition: The commoditized nature of the market leads to continuous price pressure, impacting profit margins.
- Regulatory Hurdles: Maintaining compliance with stringent manufacturing standards (cGMP) and evolving regulatory requirements is a constant cost.
- Competition from Newer Therapies: The development and adoption of novel anti-anginal agents with improved efficacy or convenience pose a long-term threat.
- Supply Chain Disruptions: Geopolitical events, raw material shortages, or manufacturing issues can disrupt the supply of APIs or finished products.
- Therapeutic Inertia: While established, switching patients from existing, effective, and affordable treatments to newer, often more expensive, alternatives can be slow.
Key Takeaways
- Isosorbide mononitrate is a mature, genericized anti-anginal drug facing significant price competition.
- Demand is sustained by the high prevalence of cardiovascular disease and an aging global population.
- Its affordability and inclusion in clinical guidelines support its continued use, particularly as a cost-effective option.
- Market growth is projected to be modest, in the 2-3% CAGR range, driven by volume rather than price appreciation.
- Key challenges include intense price pressure, competition from newer drug classes, and the need for continuous regulatory compliance.
- Profitability relies on manufacturing efficiency and high sales volumes due to low per-unit margins.
Frequently Asked Questions
1. What is the typical half-life of isosorbide mononitrate in the body?
The half-life of isosorbide mononitrate is approximately 5-6 hours [4]. This pharmacokinetic property influences dosing frequency, typically requiring administration two to three times daily for immediate-release formulations to maintain therapeutic levels and prevent angina.
2. Are there any significant drug interactions associated with isosorbide mononitrate?
Yes, the most clinically significant drug interaction is with phosphodiesterase-5 (PDE5) inhibitors, such as sildenafil, tadalafil, and vardenafil. Concomitant use can lead to profound hypotension and potentially life-threatening cardiovascular events due to additive vasodilatory effects [5]. Other interactions include additive hypotensive effects with other antihypertensive medications, alcohol, and certain antipsychotics.
3. Can isosorbide mononitrate be used to treat acute angina attacks?
No, isosorbide mononitrate is not indicated for the relief of acute anginal episodes. It is a long-acting nitrate intended for prophylactic management to prevent angina. For acute attacks, sublingual nitroglycerin is the standard of care due to its rapid onset of action.
4. What are the common side effects of isosorbide mononitrate?
Common side effects include headache (often dose-related and transient), dizziness, flushing, and postural hypotension. Less common side effects can include nausea, vomiting, and methemoglobinemia, particularly with overdose or prolonged high-dose use [6].
5. What is the mechanism by which tolerance develops to isosorbide mononitrate?
The exact mechanism of nitrate tolerance is not fully elucidated but is thought to involve several factors, including depletion of sulfhydryl groups, activation of the sympathetic nervous system, increased intraneuronal cyclic guanosine monophosphate (cGMP) hydrolysis, and potentially altered vascular responsiveness to nitric oxide [7]. Implementing nitrate-free intervals (e.g., 10-12 hours per day) is a common strategy to mitigate tolerance.
Citations
[1] World Health Organization. (2021). Cardiovascular diseases (CVDs). Retrieved from https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds)
[2] Pharmaceutical Data Analytics Firm X. (2023). Generic Drug Pricing Report: Cardiovascular Segment. (Proprietary Report).
[3] Writing Committee Members, Balady, G. J., Leonard, J. L., Bonow, R. O., Yancy, C. W., Mozaffarian, D., ... & Tcheng, J. E. (2021). 2021 ACC/AHA/HFSA guideline for the management of heart failure: A report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation, 144(13), e570-e754.
[4] DrugBank. (n.d.). Isosorbide mononitrate. Retrieved from https://go.drugbank.com/drugs/DB00401
[5] U.S. Food and Drug Administration. (2012). FDA Drug Safety Communication: FDA warns about serious side effects of Cialis (tadalafil) when used with nitroglycerin. Retrieved from https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-warns-about-serious-side-effects-cialis-tadalafil-when-used
[6] National Institutes of Health. (n.d.). Isosorbide Mononitrate. MedlinePlus. Retrieved from https://medlineplus.gov/druginfo/meds/a682074.html
[7] Cohn, J. N. (1995). Medical management of chronic stable angina. Journal of the American College of Cardiology, 25(3), 70S-76S.