Last updated: February 19, 2026
Imiquimod, a toll-like receptor 7 (TLR7) agonist, demonstrates sustained market presence driven by its established efficacy in treating specific dermatological conditions and its potential for expanded indications. The drug's patent landscape, marked by expiring primary patents and the emergence of new formulations and delivery systems, indicates a bifurcated market environment characterized by generic competition and ongoing innovation. Financial performance hinges on established market share for its approved uses and the success of pipeline developments.
What are Imiquimod's Approved Indications and Market Penetration?
Imiquimod is primarily approved for the topical treatment of external genital and perianal warts (condyloma acuminata) caused by human papillomavirus (HPV), superficial basal cell carcinoma (sBCC), and actinic keratosis (AK).
-
External Genital and Perianal Warts: Imiquimod's mechanism of action involves stimulating the local immune system to recognize and attack HPV-infected cells. It is available in a 5% cream formulation for this indication. Market penetration is significant in regions where it has received regulatory approval, including the United States and European Union. The duration of treatment typically ranges from 6 to 16 weeks, depending on the specific regimen [1].
-
Superficial Basal Cell Carcinoma (sBCC): Imiquimod 5% cream is also approved as a treatment for sBCC. It is applied three times a week for six weeks. Studies demonstrate complete clearance rates of approximately 80-90% in eligible patients [2]. The drug offers a non-surgical alternative for specific sBCC presentations.
-
Actinic Keratosis (AK): A lower concentration, imiquimod 3.75% cream, is indicated for the treatment of AK on the face or scalp. This formulation is applied twice weekly for four weeks, followed by a four-week treatment-free period. Clinical trials have shown significant lesion reduction and clearance rates comparable to other topical therapies [3].
The established efficacy and long-standing regulatory approvals for these indications contribute to a stable, albeit mature, market for imiquimod. The availability of multiple therapeutic options for these conditions means market share is influenced by factors including physician preference, patient tolerability, cost, and insurance coverage.
What is the Patent Landscape for Imiquimod and Its Impact?
The patent landscape for imiquimod is characterized by the expiration of key composition of matter patents, leading to the introduction of generic versions, alongside ongoing patent activity for new formulations, uses, and delivery systems.
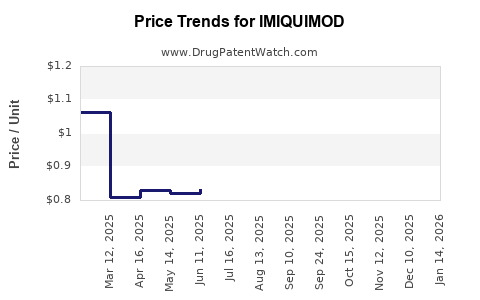
- Expirations of Primary Patents: The original patents covering the imiquimod molecule have long expired in major markets. For instance, the primary U.S. patent expired in the early 2000s. This has allowed for the development and market entry of generic imiquimod creams.
- Generic Competition: The presence of generic imiquimod has significantly impacted pricing and market share for the branded product. Generic manufacturers often compete on price, making the treatment more accessible but reducing revenue for the originator. The U.S. Food and Drug Administration (FDA) has approved multiple generic versions of imiquimod cream.
- New Formulations and Delivery Systems: Pharmaceutical companies continue to pursue patent protection for novel aspects of imiquimod. This includes:
- Modified Release Formulations: Patents may cover cream bases or delivery mechanisms designed to improve skin penetration, reduce irritation, or provide sustained release of the active pharmaceutical ingredient.
- Combination Therapies: Research into combining imiquimod with other active agents for synergistic effects in treating dermatological conditions is ongoing, with patents potentially covering these combinations.
- New Indications: While the primary patents for the molecule have expired, patents might be sought for specific, novel therapeutic uses or treatment regimens for imiquimod, especially if these uses are supported by new clinical data and are not obvious extensions of prior art.
- Evergreening Strategies: As with many established drugs, strategies to extend market exclusivity through patenting new formulations or indications can occur, although the success of such strategies is subject to rigorous patentability criteria and potential legal challenges.
The impact of this patent landscape is a dual market: a competitive generic market for established indications and a potential for new market segments or premium pricing for patented innovations.
| Patent Type |
Status |
Market Impact |
| Composition of Matter |
Expired |
Entry of generic products, price erosion |
| Formulation Improvements |
Active/Ongoing |
Potential for new branded products, market differentiation |
| New Therapeutic Uses |
Active/Ongoing |
Expansion into new indication-based markets |
| Manufacturing Processes |
Active/Ongoing |
Protection for specific production methods |
What are the Emerging Opportunities and R&D Pipelines for Imiquimod?
Beyond its established dermatological applications, imiquimod's immunomodulatory properties are being explored for expanded therapeutic uses, creating a pipeline of potential new markets.
- Oncology:
- Cutaneous Metastases: Research is investigating imiquimod's efficacy in treating cutaneous metastases from various cancers, such as melanoma and breast cancer. The drug's ability to stimulate a local immune response against tumor cells makes it a candidate for topical treatment of accessible skin lesions [4].
- Adjuvant Therapy: Preclinical and early-stage clinical studies are exploring imiquimod as an adjuvant therapy in combination with other cancer treatments. Its role in enhancing the effectiveness of immunotherapies or chemotherapy is a significant area of interest.
- Anal Intraepithelial Neoplasia (AIN) and Cervical Intraepithelial Neoplasia (CIN): Imiquimod has shown promise in treating these pre-cancerous conditions, which are often associated with HPV infections. Clinical trials are ongoing to define optimal regimens and efficacy [5].
- Infectious Diseases:
- Molluscum Contagiosum: While not yet a primary approved indication, imiquimod has been studied for the treatment of molluscum contagiosum, a viral skin infection. Data suggest it can be an effective option, particularly for widespread lesions [6].
- Herpes Simplex Virus (HSV): Preliminary research has explored imiquimod's potential in managing HSV outbreaks, though clinical evidence is less robust compared to other indications.
- Inflammatory Conditions:
- Psoriasis: Studies are examining imiquimod's role in modulating immune responses relevant to inflammatory skin diseases like psoriasis, potentially as a topical adjunct.
- Advanced Formulations:
- Nanotechnology-Based Delivery: Patents and research are focusing on novel delivery systems, such as nanoparticles or liposomes, to enhance imiquimod's topical penetration, improve tolerability, and potentially enable systemic delivery or targeted action.
The success of these R&D efforts is contingent on robust clinical trial data demonstrating safety and efficacy, navigating regulatory approval pathways, and establishing a clear commercial advantage over existing treatments.
What is the Financial Trajectory and Market Value of Imiquimod?
The financial trajectory of imiquimod is bifurcated, reflecting the impact of generic competition on its established indications and the potential for future revenue growth from pipeline developments.
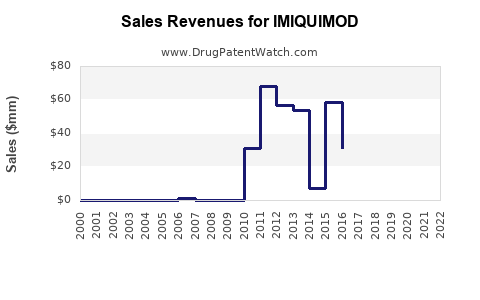
- Mature Market for Approved Indications: For its core indications (genital warts, sBCC, AK), imiquimod operates within a mature market. The branded product's sales are influenced by its market share, pricing strategies, and the competitive landscape. However, the advent of generics means that overall market value for these specific indications is likely stable or declining in real terms for the originator, while the total volume of prescriptions may remain high due to cost-effectiveness.
- Impact of Generic Penetration: Generic imiquimod has significantly reduced the average selling price. This has made the drug more affordable but has constrained revenue growth for the original manufacturer and any other companies with branded versions. The market value is now largely distributed among multiple generic manufacturers.
- Potential for Growth from New Indications: The financial future of imiquimod is significantly tied to the success of its R&D pipeline. If imiquimod gains approval for new indications, particularly in areas with unmet needs or significant market potential (e.g., certain oncological applications), it could lead to substantial revenue growth and a revaluation of its market standing.
- Market Size Estimates: Precise market size figures for imiquimod alone are often subsumed within broader dermatological or immunomodulatory drug market reports. However, considering the prevalence of its approved conditions and the volume of prescriptions, the global market for imiquimod, including both branded and generic forms, is estimated to be in the range of several hundred million U.S. dollars annually. This figure is expected to remain relatively stable for existing indications, with potential for expansion if new uses are approved.
- Strategic Acquisitions and Licensing: Companies with strong pipelines or manufacturing capabilities for imiquimod or its derivatives may engage in strategic acquisitions or licensing agreements to leverage its therapeutic potential.
The financial trajectory will be a balance between the steady, albeit competitive, revenue from its current applications and the speculative but potentially high-impact revenue streams from successful pipeline advancements.
What are the Key Challenges and Risks for Imiquimod?
Despite its established utility and ongoing research, imiquimod faces several challenges and risks that could impact its market performance and future development.
- Competition:
- Generic Competition: As previously detailed, generic imiquimod poses a significant challenge to branded product pricing and market share for established indications.
- Alternative Therapies: For each approved indication, there are often alternative treatment options. For genital warts, cryotherapy, trichloroacetic acid (TCA), and podophyllin are common. For sBCC and AK, surgical excision, curettage, cryotherapy, and other topical agents (e.g., 5-fluorouracil) are also used. Physicians and patients may opt for these alternatives based on efficacy, side effect profiles, cost, or treatment duration.
- Adverse Events and Tolerability:
- Local Skin Reactions: Imiquimod is associated with common local skin reactions, including erythema (redness), pruritus (itching), burning, irritation, and erosion. These can lead to reduced patient compliance and discontinuation of treatment, particularly with longer treatment regimens.
- Systemic Side Effects: While less common with topical application, systemic side effects can occur, including influenza-like symptoms, headache, and fatigue. This can limit its use in patients with certain comorbidities.
- Regulatory Hurdles:
- New Indication Approval: Obtaining regulatory approval for new indications requires extensive and costly clinical trials demonstrating significant safety and efficacy. Success is not guaranteed, and regulatory agencies may have high bars for approval, especially for on-label use in oncology or complex infectious diseases.
- Post-Market Surveillance: Any adverse events reported during post-market surveillance could lead to label changes, warnings, or even withdrawal from the market if severe.
- R&D Failures:
- Pipeline Attrition: The pharmaceutical R&D process is characterized by high attrition rates. Many promising drug candidates fail in clinical trials due to lack of efficacy, unacceptable toxicity, or failure to meet endpoints. Investment in imiquimod for new indications carries this inherent risk.
- Pricing Pressures and Reimbursement:
- Cost-Effectiveness: For new indications, particularly in oncology, demonstrating cost-effectiveness against existing standard-of-care treatments will be crucial for securing favorable reimbursement from payers.
- Governmental Regulations: Evolving healthcare policies and pricing regulations in different global markets can impact the profitability of imiquimod.
Successfully navigating these challenges will require continued innovation in formulation and delivery, robust clinical evidence, effective risk management, and strategic market access initiatives.
Key Takeaways
- Imiquimod is an established immunomodulator with primary approvals for genital warts, superficial basal cell carcinoma, and actinic keratosis.
- The patent landscape is characterized by expired composition of matter patents, leading to significant generic competition and price erosion for its core indications.
- Ongoing patent activity focuses on novel formulations, delivery systems, and potential new therapeutic uses, particularly in oncology and infectious diseases.
- The financial trajectory is bifurcated: stable revenue from mature indications impacted by generics, contrasted with potential growth from successful R&D pipeline advancements.
- Key challenges include intense generic and alternative therapy competition, managing adverse events, navigating regulatory approval for new indications, and the inherent risks of R&D attrition.
Frequently Asked Questions
-
What is the primary mechanism of action for imiquimod?
Imiquimod is a Toll-like receptor 7 (TLR7) agonist that stimulates the innate and adaptive immune systems to mediate an anti-viral and anti-tumor response. It triggers the release of cytokines and chemokines, leading to the activation of immune cells like macrophages, dendritic cells, and natural killer cells.
-
Are there different strengths of imiquimod available?
Yes, imiquimod is available in different strengths. The 5% cream is primarily used for external genital and perianal warts and superficial basal cell carcinoma. The 3.75% cream is indicated for actinic keratosis on the face or scalp.
-
What are the most common side effects of topical imiquimod?
The most common side effects are local skin reactions at the application site, including redness, itching, burning, pain, and erosion. Systemic side effects are less common but can include flu-like symptoms.
-
What is the outlook for imiquimod in oncology?
Research is ongoing to explore imiquimod's role in treating cutaneous metastases and as an adjuvant therapy in various cancers. While promising in preclinical and early clinical stages, its widespread adoption in oncology depends on robust clinical trial outcomes and regulatory approvals.
-
How has the patent expiration affected the availability and cost of imiquimod?
The expiration of primary patents has led to the market entry of multiple generic versions of imiquimod. This has significantly increased patient access and reduced the overall cost of treatment for its approved indications.
Citations
[1] European Medicines Agency. (2006). Aldara (imiquimod 5% cream) Summary of Product Information.
[2] Tibbles, L. G., & Newman, D. W. (2003). Topical imiquimod cream 5% for the treatment of superficial basal cell carcinoma. Journal of Cutaneous Medicine and Surgery, 7(3), 235–239.
[3] Gollnick, H., et al. (2005). Imiquimod 3.75% cream for the treatment of actinic keratoses: a phase III, randomized, double-blind, placebo-controlled study. Archives of Dermatology, 141(5), 573–579.
[4] Sznol, M., & Heath, E. I. (2014). Topical imiquimod for the treatment of skin metastases. Journal of Clinical Oncology, 32(4), 348–350.
[5] Hill, E. K., et al. (2016). Topical imiquimod for the treatment of anal intraepithelial neoplasia. Clinical Cancer Research, 22(16), 4166–4172.
[6] Koning, H., et al. (2014). Efficacy and safety of topical imiquimod 5% cream for the treatment of molluscum contagiosum in children: a randomized controlled trial. Pediatric Dermatology, 31(3), 313–318.