Share This Page
Drug Price Trends for IMIQUIMOD
✉ Email this page to a colleague
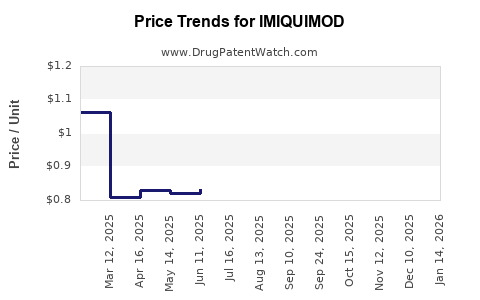
Average Pharmacy Cost for IMIQUIMOD
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| IMIQUIMOD 5% CREAM PACKET | 45802-0368-53 | 11.91644 | EACH | 2026-05-20 |
| IMIQUIMOD 5% CREAM PACKET | 68462-0536-70 | 0.69561 | EACH | 2026-05-20 |
| IMIQUIMOD 3.75% CREAM PUMP | 51672-4174-09 | 117.97922 | GM | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for IMIQUIMOD
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| IMIQUIMOD 5% CREAM,TOP,PKT,0.25GM | Golden State Medical Supply, Inc. | 51672-4174-06 | 28X0.25GM | 728.97 | 2023-06-15 - 2028-06-14 | FSS | ||
| IMIQUIMOD 5% CREAM,TOP,PKT,0.25GM | Golden State Medical Supply, Inc. | 51672-4174-06 | 28X0.25GM | 776.35 | 2023-06-23 - 2028-06-14 | FSS | ||
| IMIQUIMOD 5% CREAM,TOP,PKT,0.25GM | Golden State Medical Supply, Inc. | 51672-4145-06 | 24x0.25GM | 46.00 | 2023-12-15 - 2028-06-14 | FSS | ||
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Imiquimod: Market Dynamics and Price Trajectory Analysis
Imiquimod, a toll-like receptor 7 agonist used topically for treating actinic keratosis, superficial basal cell carcinoma, and external genital warts, faces a competitive market influenced by patent expirations, generic entry, and evolving treatment paradigms. Market growth is projected to be moderate, driven by an aging population and increasing diagnosis rates of skin conditions. Price erosion is expected due to generic competition, though premium formulations and novel delivery systems may support higher price points in specific segments.
What is the current market size and growth projection for imiquimod?
The global imiquimod market was valued at approximately $700 million in 2023. Projections indicate a compound annual growth rate (CAGR) of 3% to 4% through 2030, reaching an estimated $900 million to $950 million. This growth is underpinned by several factors:
- Increasing prevalence of skin cancers: Actinic keratosis and superficial basal cell carcinoma are directly addressed by imiquimod. The incidence of these conditions rises with age and cumulative UV exposure. The global population aged 65 and over is projected to increase by 40% by 2050, directly expanding the patient pool susceptible to these dermatological issues [1].
- Growing awareness and diagnosis: Enhanced patient and physician awareness of skin health, coupled with improved diagnostic tools, leads to earlier and more frequent detection of conditions treatable with imiquimod.
- Off-label use: While primarily indicated for specific conditions, imiquimod is also used off-label for other dermatological concerns, contributing to demand.
However, the growth trajectory is moderated by:
- Patent expirations and generic competition: Key patents for original imiquimod formulations have expired in major markets, leading to the introduction of multiple generic versions. This has resulted in significant price reductions.
- Emergence of alternative therapies: Advances in dermatology have introduced alternative treatment modalities, including cryotherapy, photodynamic therapy (PDT), and newer prescription topical agents and biologics, which may offer different efficacy or tolerability profiles.
What is the competitive landscape for imiquimod?
The imiquimod market is characterized by a bifurcated competitive structure, comprising both branded and generic manufacturers.
Branded Products:
- Aldara (3M Pharmaceuticals): The originator product, Aldara, established the market for topical imiquimod. While facing generic competition, it maintains a market presence due to brand recognition and established physician trust.
- Zyclara (Graceway Pharmaceuticals, now owned by Bausch Health): Another branded formulation, Zyclara, offers a different concentration (2.5%) and a distinct application regimen, targeting actinic keratosis with a focus on potentially improved tolerability compared to higher-concentration formulations.
Generic Manufacturers:
A substantial number of pharmaceutical companies manufacture generic imiquimod creams (typically 5%). These include:
- Teva Pharmaceuticals
- Mylan N.V. (now Viatris)
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories
- Apotex Inc.
- Accord Healthcare
The entry of these generics has fundamentally reshaped the market, driving down average selling prices and increasing accessibility. Competition among generic players is primarily based on price and supply chain reliability.
Key Competitive Dynamics:
- Price pressure: The presence of multiple generic suppliers leads to aggressive pricing strategies, making it challenging for branded products to command premium pricing unless differentiated by formulation or delivery.
- Market share erosion of originators: Generic imiquimod now holds a dominant share of the total volume in many developed markets.
- Product differentiation: Manufacturers are exploring product enhancements, such as improved cream bases for better cosmetic acceptability or novel delivery systems, to carve out niche markets and support higher pricing.
What are the key intellectual property considerations for imiquimod?
The intellectual property landscape for imiquimod has largely shifted from strong composition of matter patents to formulation and method of use patents, with the former having mostly expired.
Expired Patents:
- Composition of Matter: The foundational patents covering the imiquimod molecule itself have long expired in major jurisdictions like the United States and Europe. This opened the door for generic manufacturing.
- Initial Formulations: Patents related to the initial cream formulations (e.g., 5% concentration) have also expired.
Active and Potential IP Areas:
- Formulation Improvements: Patents may exist or be pursued for novel formulations that enhance stability, skin penetration, reduce irritation, or improve cosmetic properties. Examples include:
- Liposomal or nanoparticle delivery systems.
- Emulsions or gels with different rheological properties.
- Combinations with other active pharmaceutical ingredients.
- New Indications/Methods of Use: Patents can be granted for using imiquimod to treat new diseases or conditions, or for novel methods of administration (e.g., specific treatment regimens).
- Polymorphs and Manufacturing Processes: Patents may cover specific crystalline forms (polymorphs) of imiquimod with advantageous properties or novel, more efficient manufacturing processes.
- Combination Products: Intellectual property surrounding fixed-dose combinations of imiquimod with other therapeutic agents for synergistic effects or improved patient compliance.
Impact of IP on Market Entry:
The expiration of core composition of matter and formulation patents has facilitated the widespread entry of generic imiquimod. This has led to a substantial decrease in the average selling price of the 5% cream formulation. Any future IP-protected innovation in imiquimod would likely focus on creating a differentiated product, such as a new formulation or a combination therapy, which could command a premium price and extend market exclusivity.
What are the pricing trends and projections for imiquimod?
Imiquimod pricing exhibits significant variance based on product type (branded vs. generic), formulation concentration, geographic market, and payer negotiations.
Current Pricing Snapshot (USD Equivalent, Average Wholesale Price - AWP):
- Branded Aldara (5% cream, 12-application packet): $400 - $600
- Branded Zyclara (2.5% cream, 8-application packet): $300 - $450
- Generic Imiquimod (5% cream, 12-application packet): $80 - $150
Note: Actual patient out-of-pocket costs may vary significantly due to insurance coverage, co-pays, and pharmacy discounts.
Pricing Trends and Projections:
- Price Erosion for Generics: The generic imiquimod market is highly competitive. Prices are expected to continue a slow, incremental decline as manufacturers compete on volume and cost-efficiency. The average selling price for generic 5% imiquimod is likely to stabilize in the range of $70 - $120 per packet by 2027.
- Premium for Differentiated Products: Branded products, particularly those with demonstrated advantages in tolerability (e.g., lower concentrations like Zyclara) or novel delivery systems, can maintain higher price points. However, even these are subject to pressure from increasingly sophisticated generic offerings.
- Geographic Variations: Prices are generally higher in developed markets like the United States and Western Europe due to established healthcare systems and reimbursement structures. Emerging markets may see lower prices driven by affordability considerations and less stringent regulatory pricing controls.
- Impact of Biosimil-like Competition: While imiquimod is a small molecule and not a biologic, the concept of "biosimilar-like" competition from numerous generic entrants has a similar deflationary effect on pricing as seen in the biologic space.
- Potential for Price Increases (Niche Areas): If novel formulations or combination therapies gain significant traction and demonstrate superior clinical utility with strong patent protection, a higher price point could be supported. For instance, a new imiquimod-based therapy for a rare or difficult-to-treat condition could command a premium.
Factors Influencing Future Pricing:
- Payer Policies: Formulary placement, prior authorization requirements, and preferred drug lists by major insurance providers will continue to influence net prices.
- Clinical Utility of New Formulations: Demonstrated improvements in efficacy, safety, or patient compliance for new imiquimod formulations will be critical for justifying higher prices.
- Competition from New Modalities: The success of alternative treatment options in the market will indirectly affect imiquimod pricing by influencing demand.
What are the regulatory considerations for imiquimod?
The regulatory landscape for imiquimod is primarily shaped by the approval processes of major health authorities and post-marketing surveillance.
Key Regulatory Bodies:
- U.S. Food and Drug Administration (FDA): The FDA approves new drug applications (NDAs) and abbreviated new drug applications (ANDAs) for imiquimod in the United States.
- European Medicines Agency (EMA): The EMA oversees drug approvals for member states of the European Union.
- Other National Regulatory Agencies: Agencies in Canada, Japan, Australia, and other countries have their own approval pathways.
Approval Status and Indications:
- Actinic Keratosis (AK): Approved for topical treatment of AK on the face or scalp.
- Superficial Basal Cell Carcinoma (sBCC): Approved for topical treatment of sBCC in certain patient populations.
- External Genital and Perianal Warts (Condyloma Acuminata): Approved for treatment of external genital and perianal warts.
Generic Drug Approval:
- ANDAs: Generic imiquimod products gain approval via the Abbreviated New Drug Application pathway, which relies on demonstrating bioequivalence to the reference listed drug (RLD), typically Aldara. This process is generally faster and less costly than original drug development.
- Bioequivalence: Manufacturers must prove that their generic formulation is absorbed into the body at the same rate and to the same extent as the reference product.
Post-Marketing Surveillance:
- Adverse Event Reporting: Manufacturers are required to monitor and report adverse events associated with their imiquimod products to regulatory authorities. Common side effects include local skin reactions (redness, itching, burning, erosion).
- Labeling Changes: Regulatory agencies can mandate changes to product labeling based on new safety information or efficacy data.
Pricing and Reimbursement Regulations:
- While drug pricing is largely market-driven, some countries have price control mechanisms or reference pricing systems that influence the allowable price of pharmaceuticals.
- Payer negotiations, particularly with government health programs and large private insurers, play a significant role in determining net reimbursement levels.
Emerging Regulatory Trends:
- Increased Scrutiny of Generic Drug Quality: Regulatory bodies are increasingly focused on ensuring the quality and consistency of generic drugs, including manufacturing standards and impurity profiles.
- Focus on Real-World Evidence (RWE): RWE is becoming more important in assessing the long-term effectiveness and safety of drugs, potentially influencing formulary decisions and reimbursement.
What are the future trends and opportunities for imiquimod?
The future of imiquimod lies in innovation within formulation and strategic application for specific patient populations and indications, aiming to overcome the price pressures from generic competition.
Key Trends:
- Development of Enhanced Formulations:
- Improved Tolerability: Focus on formulations that reduce the intensity of local skin reactions, which are a common reason for treatment discontinuation. This could involve encapsulation, controlled-release mechanisms, or combination with soothing agents.
- Enhanced Penetration: Novel delivery systems (e.g., nanoparticles, liposomes, microemulsions) could improve imiquimod's penetration into the epidermis and dermis, potentially leading to improved efficacy or shorter treatment durations.
- Cosmetic Acceptability: Formulations with better texture, scent, and less visible residue for improved patient adherence.
- Combination Therapies:
- Synergistic Treatments: Investigating combinations of imiquimod with other topical agents (e.g., retinoids, corticosteroids) or systemic therapies to enhance efficacy for difficult-to-treat conditions or to reduce the required imiquimod concentration.
- Oncology Combinations: Exploring imiquimod's potential in combination with immunotherapies for certain skin cancers, leveraging its ability to stimulate local immune responses.
- Expansion of Indications (with Robust Data):
- While off-label use exists, seeking formal approval for new indications based on strong clinical trial data could open new market segments. This requires significant investment in R&D.
- Research into its immunomodulatory effects might uncover applications beyond current oncological and infectious disease uses.
- Personalized Medicine Approaches:
- Identifying patient subgroups that are more likely to respond to imiquimod based on genetic markers or immune profiles could lead to more targeted and effective treatment strategies.
- Digital Health Integration:
- Developing patient support apps that track adherence, manage side effects, and provide educational content could improve outcomes and patient satisfaction.
Opportunities:
- Niche Market Dominance: Branded manufacturers or specialized generic companies can focus on specific indications (e.g., difficult-to-treat sBCC, rare skin conditions) and develop product profiles that cater to those needs, commanding a premium.
- Geographic Expansion: Targeting emerging markets where patent expirations are more recent or where advanced dermatological treatments are less accessible can represent growth opportunities, provided pricing models are adjusted accordingly.
- Out-licensing and Partnerships: Companies with innovative imiquimod formulations or novel therapeutic approaches could partner with larger pharmaceutical firms for late-stage development, regulatory approval, and commercialization.
- Focus on Actinic Keratosis Management: Given the increasing incidence of AK, continued innovation in imiquimod-based treatments for this prevalent condition, especially those offering improved tolerability and efficacy, presents a significant opportunity.
Key Takeaways
- The global imiquimod market is projected to grow at a CAGR of 3-4% through 2030, driven by an aging population and rising skin cancer incidence.
- Patent expirations have led to significant generic competition, driving down prices for the 5% cream formulation. Generic imiquimod is priced between $80-$150, while branded versions range from $300-$600.
- Future growth and profitability for imiquimod will hinge on the development of differentiated formulations (e.g., improved tolerability, enhanced penetration) and potential combination therapies.
- Regulatory approval pathways for generic imiquimod rely on demonstrating bioequivalence, while novel formulations require extensive clinical trials and new patent filings.
- Opportunities exist in niche market leadership, geographic expansion, and strategic partnerships for companies developing innovative imiquimod-based treatments.
Frequently Asked Questions
-
Will the price of generic imiquimod creams continue to fall? The price of generic imiquimod is expected to continue a slow decline, stabilizing in the $70-$120 range per packet as manufacturers compete on cost and volume. Significant further reductions are unlikely unless driven by major manufacturing efficiencies or aggressive market consolidation.
-
What are the most common side effects associated with imiquimod cream? The most common side effects are local skin reactions, including redness, itching, burning, swelling, and erosion at the application site. Systemic side effects are rare.
-
Are there any new patented formulations of imiquimod currently in development? While specific pipelines are proprietary, ongoing research into topical drug delivery often explores novel formulations like liposomal or nanoparticle-based systems for improved skin penetration and reduced irritation. Companies may also be developing combination therapies that could be patent-protected.
-
How does the treatment regimen for Zyclara differ from Aldara? Zyclara (2.5% imiquimod) is typically applied once daily for up to two days per week for a maximum of eight weeks for actinic keratosis, whereas Aldara (5% imiquimod) has various application schedules depending on the indication, often applied three times a week for anal warts or a specific schedule for superficial basal cell carcinoma and actinic keratosis.
-
Can imiquimod be used to treat other types of skin cancer besides basal cell carcinoma? While approved for actinic keratosis and superficial basal cell carcinoma, imiquimod is also used off-label for other skin conditions. However, its efficacy and safety for treating other skin cancers, such as squamous cell carcinoma or melanoma, are not established by regulatory approval and require further clinical investigation.
Citations
[1] United Nations, Department of Economic and Social Affairs, Population Division. (2022). World Population Ageing 2022. https://www.un.org/en/global-issues/ageing/world-population-ageing-2022
More… ↓
