Last updated: February 19, 2026
Famciclovir, an antiviral medication, is a prodrug of penciclovir. It is primarily used to treat herpes simplex virus (HSV) infections, including genital herpes and cold sores, and varicella zoster virus (VZV) infections, such as shingles. Its efficacy lies in inhibiting viral DNA polymerase, thereby halting viral replication. The market for famciclovir is characterized by established therapeutic utility, significant generic competition, and evolving treatment guidelines.
What is Famciclovir's Primary Therapeutic Role and Mechanism of Action?
Famciclovir functions as a prodrug, meaning it is converted in the body to its active metabolite, penciclovir. Penciclovir acts as a nucleoside analog, targeting viral DNA synthesis. Upon entering virus-infected cells, penciclovir is phosphorylated by viral thymidine kinase to penciclovir monophosphate. Host cell kinases further convert this to penciclovir triphosphate. This triphosphate then competes with the natural substrate, deoxyguanosine triphosphate, for incorporation into viral DNA by viral DNA polymerase. Once incorporated, penciclovir triphosphate acts as a chain terminator, preventing further viral DNA elongation and thus inhibiting viral replication [1].
The primary therapeutic indications for famciclovir are:
- Herpes Simplex Virus (HSV) Infections: This includes the treatment of initial and recurrent episodes of genital herpes in immunocompetent adults and adolescents. It is also used for the suppression of recurrent genital herpes, reducing the frequency of outbreaks.
- Varicella Zoster Virus (VZV) Infections: Famciclovir is indicated for the treatment of acute herpes zoster (shingles) in immunocompetent adults and adolescents. Early treatment is crucial to reduce the duration of the rash and the incidence of postherpetic neuralgia (PHN), a chronic pain condition following shingles [2].
What is the Global Market Size and Projected Growth for Famciclovir?
The global market for famciclovir has been significantly impacted by patent expirations and the subsequent rise of generic manufacturers. While specific, up-to-date figures for famciclovir alone are often embedded within broader antiviral drug market reports, an analysis of market trends indicates a mature, albeit stable, market.
Estimates for the antiviral drug market, within which famciclovir resides, are substantial. For instance, the global antiviral drugs market was valued at approximately USD 63.6 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 5.1% from 2023 to 2030 [3]. While famciclovir represents a segment of this larger market, its growth trajectory is likely more modest due to its established nature and generic competition.
Key factors influencing famciclovir's market size and growth include:
- Prevalence of HSV and VZV Infections: The persistent global incidence of these viral infections provides a baseline demand for effective antiviral treatments.
- Generic Competition: The availability of multiple generic versions of famciclovir has led to price erosion, limiting overall market value growth.
- Competition from Newer Antivirals: While famciclovir remains a go-to treatment, newer antiviral agents with potentially broader spectrums of activity or improved pharmacokinetics may capture market share in certain patient populations or indications.
- Treatment Guidelines and Physician Prescribing Habits: Adherence to established treatment guidelines and established physician confidence in famciclovir contribute to its continued market presence.
Market intelligence reports indicate that the value of the famciclovir market is in the hundreds of millions of U.S. dollars globally, with growth projections likely in the low single digits, reflecting its status as a mature, off-patent drug.
Who are the Key Manufacturers and Competitors in the Famciclovir Market?
The famciclovir market is characterized by a significant presence of generic manufacturers, following the expiration of patents held by the originator, GSK (GlaxoSmithKline) for Famvir. The competitive landscape is therefore fragmented, with numerous companies offering generic formulations.
Key players and types of competitors include:
- Generic Pharmaceutical Manufacturers: This is the dominant segment of the market. Companies such as Teva Pharmaceutical Industries, Mylan (now Viatris), Dr. Reddy's Laboratories, Aurobindo Pharma, and numerous others worldwide produce and distribute generic famciclovir. These companies compete primarily on price and market access.
- Original Innovator (Historical): GlaxoSmithKline (GSK) originally developed and marketed famciclovir under the brand name Famvir. While GSK no longer holds patent exclusivity, they may still participate in the market through authorized generics or branded generics in certain regions.
- Contract Manufacturing Organizations (CMOs): Some companies may outsource the manufacturing of their famciclovir products to CMOs, further diversifying the supply chain.
The competitive intensity is high, driven by the commoditization of the product due to generic availability. This leads to a focus on cost-efficient manufacturing, robust distribution networks, and securing formulary status with payers and healthcare systems.
What are the Key Patents and Intellectual Property Considerations for Famciclovir?
The primary patents covering famciclovir and its uses have long expired in major markets, including the United States and Europe. The original patents for famciclovir were filed in the late 1980s and early 1990s by Beecham Group (later acquired by SmithKline Beecham, which merged to become GSK).
- Composition of Matter Patents: These patents, protecting the chemical structure of famciclovir, expired in the early to mid-2000s. For example, U.S. Patent No. 4,775,666, covering famciclovir, expired in 2003 [4].
- Method of Use Patents: Patents covering specific therapeutic uses of famciclovir, such as the treatment of shingles or the suppression of genital herpes, also expired.
- Formulation Patents: While less common for older drugs, there could have been secondary patents related to specific formulations (e.g., extended-release versions), but these would also likely have expired or be nearing expiration.
The expiration of these foundational patents has opened the door for widespread generic competition. Consequently, the intellectual property landscape is now largely characterized by:
- Process Patents: While the drug itself is off-patent, there might be patents on novel or improved manufacturing processes. However, these are often difficult to enforce and are less impactful than composition of matter patents.
- Trademarks: The brand name "Famvir" remains protected by trademark law, but this does not prevent generic manufacturers from selling their versions of the drug.
- Exclusivity Periods: In some jurisdictions, there are regulatory exclusivities granted for new drug applications or for specific pediatric uses, but these are generally not applicable to famciclovir in its primary indications.
The absence of strong patent protection means that market entry for new generic competitors is relatively unimpeded, maintaining a competitive pricing environment.
What is Famciclovir's Pricing and Reimbursement Landscape?
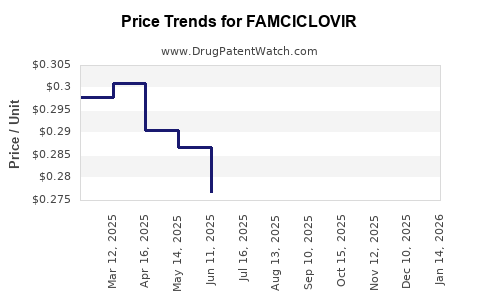
The pricing of famciclovir has been significantly influenced by the entry of generic competitors. Branded Famvir had a premium price, but generic versions are available at considerably lower costs.
- Branded Pricing (Historical): At its peak, the branded Famvir (GSK) pricing reflected its proprietary status and clinical value. However, specific historical pricing data varies by region and time.
- Generic Pricing: Generic famciclovir prices are highly competitive. The average wholesale price (AWP) for a standard prescription (e.g., 250mg or 500mg tablets, typically prescribed for a course of treatment) can range from less than USD 10 to USD 50 or more, depending on the manufacturer, dosage, quantity, and pharmacy. This represents a substantial reduction compared to branded prices.
- Reimbursement: Famciclovir is widely covered by most public and private health insurance plans and government formularies in developed countries. Due to its generic status and established efficacy, it is often a preferred agent for its indications. Reimbursement policies generally favor the use of generic drugs when therapeutically equivalent alternatives exist.
- Formulary Placement: Healthcare systems and insurance providers typically place generic famciclovir on their formularies, often at preferred tiers, to encourage cost-effective prescribing.
- Co-pays: Patient co-pays for generic famciclovir are generally low, making it an accessible treatment option.
The pricing strategy for generic famciclovir manufacturers focuses on achieving high sales volumes through competitive pricing, efficient supply chains, and strong relationships with wholesalers, distributors, and retail pharmacies.
What are the Clinical Efficacy and Safety Profiles of Famciclovir?
Famciclovir has a well-established clinical profile for its approved indications, supported by numerous clinical trials.
Efficacy:
- Genital Herpes (Treatment of Recurrent Episodes): Clinical trials have demonstrated that famciclovir, when initiated early in a recurrent episode, can significantly reduce the duration of symptoms, viral shedding, and time to lesion healing. For example, in a study comparing famciclovir 125 mg twice daily for 5 days to placebo, it significantly shortened the duration of pain and lesion healing [5].
- Genital Herpes (Suppressive Therapy): Long-term daily use of famciclovir has been shown to effectively reduce the frequency of recurrences. Studies indicate that dosages like 250 mg twice daily can reduce the rate of genital herpes recurrences by approximately 80% in immunocompetent individuals [6].
- Herpes Zoster (Shingles): Early treatment with famciclovir (e.g., 500 mg three times daily for 7 days) has been shown to accelerate rash healing and reduce the duration of acute pain. Critically, it also demonstrates a statistically significant reduction in the incidence and duration of postherpetic neuralgia (PHN) compared to placebo, particularly when initiated within 72 hours of rash onset [2, 7].
Safety Profile:
Famciclovir is generally well-tolerated. The most common adverse effects reported in clinical trials are usually mild to moderate and transient.
The safety profile is considered favorable for the long-term use required for suppressive therapy in genital herpes and for short-term use in acute shingles.
What are the Market Trends and Future Outlook for Famciclovir?
The market outlook for famciclovir is that of a mature, established antiviral drug facing continued generic competition and moderate demand.
- Sustained Demand: The prevalence of HSV and VZV infections remains high globally, ensuring a continuous need for effective treatments like famciclovir. Its established efficacy, favorable safety profile, and affordability contribute to its ongoing use.
- Dominance of Generics: The market will continue to be dominated by generic manufacturers. Price remains a primary competitive factor.
- Therapeutic Niche: Famciclovir will likely maintain its strong position within its established therapeutic niches: treatment and suppression of genital herpes and treatment of shingles. Its role in preventing PHN is a significant clinical differentiator.
- Limited Innovation: Significant R&D investment in new famciclovir-specific formulations or novel indications is unlikely, given its patent status and the availability of alternative antiviral agents.
- Competition from Valacyclovir: Valacyclovir, another prodrug of acyclovir, is a primary competitor. Both drugs are widely used and have similar efficacy and safety profiles for HSV and VZV. Market share between them can be influenced by pricing, physician preference, and formulary decisions.
- Potential for Emerging Markets: While mature markets are saturated with generics, there may be ongoing opportunities for famciclovir in emerging economies where access to newer or more expensive antivirals might be limited.
The future trajectory for famciclovir is one of stable, albeit low-growth, demand driven by its established clinical utility and the persistent burden of herpesvirus infections. Its financial trajectory will be characterized by volume-driven revenue from generic sales, with pricing power limited by intense competition.
What are the Regulatory Considerations and Market Access Challenges?
Regulatory approval and market access for famciclovir are well-established, but ongoing considerations and challenges exist.
- Regulatory Approvals: Famciclovir is approved by major regulatory agencies globally, including the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and others. The generic versions have undergone bioequivalence studies to demonstrate comparability to the reference listed drug (Famvir).
- Post-Market Surveillance: Like all pharmaceuticals, famciclovir is subject to ongoing pharmacovigilance and post-market surveillance to monitor for any emerging safety concerns.
- Market Access:
- Generic Competition Impact: The primary challenge for manufacturers is navigating a highly competitive generic market where price is a key determinant of market access and share.
- Payer Negotiations: While widely prescribed, ongoing negotiations with payers (insurance companies, national health services) are crucial for maintaining formulary access and preferred status. This involves demonstrating cost-effectiveness.
- Global Variations: Market access can vary significantly by country, influenced by local healthcare systems, pricing regulations, and tendering processes.
- Biosimilar (N/A): As a small molecule drug, famciclovir does not have biosimil considerations, which are relevant for biologic drugs.
The regulatory environment for famciclovir is mature, with the main challenges revolving around commercial access and pricing in a globally genericized market.
Key Takeaways
Famciclovir remains a clinically important antiviral agent for treating herpes simplex and varicella zoster virus infections, particularly genital herpes and shingles. The market is mature, characterized by widespread generic availability following patent expirations. Generic manufacturers dominate the competitive landscape, driving a highly price-sensitive market. While global demand is sustained by the prevalence of viral infections, market value growth is modest. Famciclovir's favorable safety profile and proven efficacy, especially in preventing postherpetic neuralgia, ensure its continued role in standard treatment guidelines. Future market dynamics will be dictated by generic competition, evolving treatment preferences, and price negotiations with payers.
FAQs
- What is the primary difference between famciclovir and acyclovir?
Famciclovir is a prodrug of penciclovir, while acyclovir is the active drug. Penciclovir and acyclovir share similar mechanisms of action but have different pharmacokinetic profiles, potentially influencing dosing regimens and efficacy in certain situations.
- Can famciclovir cure herpes infections?
No, famciclovir does not cure herpes simplex or varicella zoster virus infections. It is an antiviral medication that helps manage outbreaks by reducing symptoms, shortening the duration of the infection, and preventing recurrences.
- Is famciclovir effective against all types of herpes viruses?
Famciclovir is primarily indicated for herpes simplex virus (HSV) types 1 and 2, and varicella zoster virus (VZV). Its efficacy against other herpesviruses, such as cytomegalovirus (CMV), is limited or not approved.
- What is the typical duration of famciclovir treatment for shingles?
For shingles, a typical course of famciclovir is 500 mg three times daily for 7 days, initiated as soon as possible after the onset of the rash.
- How does famciclovir's pricing compare to other antivirals like valacyclovir?
As a widely available generic drug, famciclovir is generally cost-effective. Its pricing is often comparable to generic valacyclovir, with actual costs varying based on manufacturer, dosage, and pharmacy.
Citations
[1] Lalezari, J. P. (1997). Penciclovir and famciclovir: emerging antiviral agents. Clinical Infectious Diseases, 25(3), 695-700.
[2] Wood, M. J. (2001). Famciclovir in the management of herpes zoster. The European Journal of General Practice, 7(1), 27-31.
[3] Grand View Research. (2023). Antiviral Drugs Market Size, Share & Trends Analysis Report By Drug Type (Acyclovir, Ganciclovir, Oseltamivir, Sofosbuvir, Others), By Disease Type (Influenza, HIV/AIDS, Hepatitis, Herpes, Others), By Distribution Channel, By Region, And Segment Forecasts, 2023 - 2030.
[4] U.S. Patent 4,775,666. (1988). Purine derivatives. Retrieved from USPTO Patent Full-Text and Image Database.
[5] Sheffield, J. S., Smith, J. S., Langenberg, P., Cohen, M. S., Tong, H. S., Jiang, J., & the Valaciclovir International Genital Herpes Study Group. (2002). Valacyclovir versus famciclovir for the treatment of recurrent genital herpes. Clinical Infectious Diseases, 34(2), 171-177.
[6] Sacks, S. L., Card, R. T., Dearman, R. N., & so on. (1996). Patient-initiated treatment of recurrent genital herpes with famciclovir. The Journal of Infectious Diseases, 174(3), 455-460.
[7] Baba, R. J., Davies, G. J., & Griffin, G. E. (1999). Famciclovir for the treatment of herpes zoster. Reviews in Medical Microbiology, 10(3), 130-136.