DOCOSANOL - Generic Drug Details
✉ Email this page to a colleague
What are the generic sources for docosanol and what is the scope of freedom to operate?
Docosanol
is the generic ingredient in two branded drugs marketed by Haleon Us Holdings, Alembic, Aurobindo Pharma Ltd, Dr Reddys, P And L, P And L Development, and Sun Pharma Canada, and is included in eight NDAs. Additional information is available in the individual branded drug profile pages.There are five drug master file entries for docosanol. Thirty-nine suppliers are listed for this compound.
Summary for DOCOSANOL
| US Patents: | 0 |
| Tradenames: | 2 |
| Applicants: | 7 |
| NDAs: | 8 |
| Drug Master File Entries: | 5 |
| Finished Product Suppliers / Packagers: | 39 |
| Raw Ingredient (Bulk) Api Vendors: | 122 |
| Clinical Trials: | 1 |
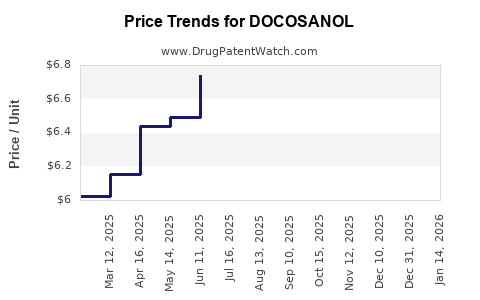
| Drug Prices: | Drug price trends for DOCOSANOL |
| What excipients (inactive ingredients) are in DOCOSANOL? | DOCOSANOL excipients list |
| DailyMed Link: | DOCOSANOL at DailyMed |
Recent Clinical Trials for DOCOSANOL
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Laboratoire Boreaderme Inc. | Phase 2 |
| Ecogene 21 | Phase 2 |
Anatomical Therapeutic Chemical (ATC) Classes for DOCOSANOL
US Patents and Regulatory Information for DOCOSANOL
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sun Pharma Canada | DOCOSANOL | docosanol | CREAM;TOPICAL | 214454-001 | Oct 26, 2023 | OTC | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Haleon Us Holdings | ABREVA | docosanol | CREAM;TOPICAL | 020941-001 | Jul 25, 2000 | OTC | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Dr Reddys | DOCOSANOL | docosanol | CREAM;TOPICAL | 214613-001 | Oct 3, 2025 | OTC | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| P And L | DOCOSANOL | docosanol | CREAM;TOPICAL | 208754-001 | Nov 19, 2018 | OTC | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Dr Reddys | DOCOSANOL | docosanol | CREAM;TOPICAL | 215505-001 | Oct 23, 2025 | OTC | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for DOCOSANOL
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Haleon Us Holdings | ABREVA | docosanol | CREAM;TOPICAL | 020941-001 | Jul 25, 2000 | ⤷ Start Trial | ⤷ Start Trial |
| Haleon Us Holdings | ABREVA | docosanol | CREAM;TOPICAL | 020941-001 | Jul 25, 2000 | ⤷ Start Trial | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Market Dynamics and Financial Trajectory for Docosanol
More… ↓