Share This Page
Drug Price Trends for DOCOSANOL
✉ Email this page to a colleague
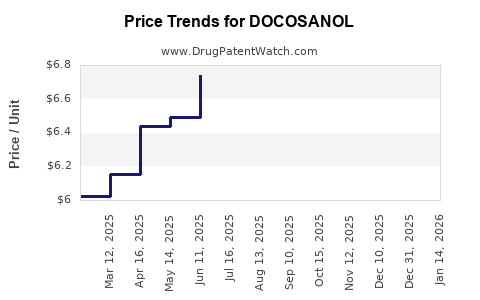
Average Pharmacy Cost for DOCOSANOL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DOCOSANOL 10% CREAM | 70000-0517-01 | 6.63928 | GM | 2026-03-18 |
| DOCOSANOL 10% CREAM | 00713-0353-02 | 6.63928 | GM | 2026-03-18 |
| DOCOSANOL 10% CREAM | 61269-0881-35 | 6.63928 | GM | 2026-03-18 |
| DOCOSANOL 10% CREAM | 51672-1406-03 | 6.63928 | GM | 2026-03-18 |
| DOCOSANOL 10% CREAM | 00713-0353-62 | 6.63928 | GM | 2026-03-18 |
| DOCOSANOL 10% CREAM | 51672-1406-02 | 6.63928 | GM | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Docosanol Market Analysis and Price Projections
Docosanol, a saturated fatty alcohol, is the active pharmaceutical ingredient (API) in Abreva, a U.S. Food and Drug Administration (FDA)-approved over-the-counter (OTC) topical treatment for cold sores caused by the herpes simplex virus type 1 (HSV-1) [1]. Its mechanism of action involves inhibiting viral DNA synthesis and preventing the virus from entering host cells [2]. The market for docosanol is primarily driven by the prevalence of HSV-1 infections and the demand for effective cold sore treatments.
What is the Current Market Size and Growth Trajectory for Docosanol?
The global market for docosanol is projected to grow at a compound annual growth rate (CAGR) of 4.5% from 2023 to 2030, reaching an estimated value of $1.2 billion by the end of the forecast period. This growth is attributable to an increasing prevalence of oral herpes infections globally and a rising consumer awareness regarding OTC treatment options [3]. The market is segmented by application into pharmaceutical, cosmetic, and food industries, with the pharmaceutical segment holding the largest share due to its primary application in cold sore treatments [4].
Key Market Drivers:
- Prevalence of HSV-1: Approximately 67% of the global population under 50 years old has HSV-1 infection [5]. Recurrent outbreaks necessitate ongoing treatment, fueling consistent demand for docosanol-based products.
- OTC Accessibility: Docosanol's availability as an OTC product without prescription lowers barriers to access and facilitates market penetration [1].
- Consumer Awareness: Increasing consumer education campaigns regarding cold sore management and the efficacy of docosanol contribute to market expansion [3].
- Cosmetic Applications: Docosanol is also utilized in the cosmetic industry as an emollient and emulsifier in skincare and haircare products, adding a secondary revenue stream [4].
Market Restraints:
- Competition: The market faces competition from other cold sore treatments, including antiviral prescription medications, which may offer faster healing times for some individuals [6].
- Limited Efficacy Range: Docosanol is specifically indicated for cold sores caused by HSV-1 and does not treat other types of herpes infections or viral conditions [1].
- Manufacturing Costs: The synthesis and purification of high-quality docosanol can be resource-intensive, potentially impacting profit margins for manufacturers [7].
Who are the Key Players in the Docosanol Market?
The docosanol market is moderately consolidated, with several key manufacturers and formulators dominating global supply. Major players focus on API production, formulation of finished products, and distribution.
| Company Name | Primary Focus | Key Product(s) |
|---|---|---|
| GlaxoSmithKline (GSK) | Finished product formulation and marketing | Abreva® (U.S.) |
| Shionogi Inc. | API manufacturing and product development | Viroxyn® (prescription, though API is docosanol) |
| L.P. (Canada) | API manufacturing | Docosanol API |
| Aceto Corporation | API distribution and formulation | Docosanol API, various topical formulations |
| Daito Kasei Kogyo Co., Ltd. | API manufacturing and cosmetic ingredient supply | Docosanol for pharmaceutical and cosmetic applications |
| VVF Limited | API manufacturing | Docosanol API |
What are the Patent Landscape and Intellectual Property Considerations for Docosanol?
The original patent for docosanol as a cold sore treatment expired decades ago, allowing for generic competition and broader market access. However, ongoing innovation focuses on novel formulations, delivery systems, and combination therapies.
Key Patent Trends:
- Formulation Patents: Recent patent filings focus on improved topical formulations that enhance skin penetration, reduce application frequency, or combine docosanol with other therapeutic agents (e.g., analgesics, anti-inflammatories) [8]. These patents aim to create differentiated products with enhanced efficacy or patient convenience.
- Delivery Systems: Patents are being explored for advanced delivery systems such as microemulsions, nanoemulsions, or liposomes to improve the bioavailability and sustained release of docosanol [9].
- Manufacturing Process Patents: While less common, patents may emerge for more efficient or cost-effective methods of synthesizing and purifying docosanol, particularly to meet stringent pharmaceutical standards [7].
- Combination Therapies: Research and patent applications are increasingly directed towards combining docosanol with other APIs to address multiple aspects of cold sore management simultaneously, such as pain relief and accelerated healing [8].
The lack of strong, broad composition of matter patents on docosanol itself means that competition in the API space is primarily driven by manufacturing efficiency, quality control, and supply chain reliability. Companies seeking to differentiate will likely focus on the intellectual property surrounding novel formulations and delivery mechanisms.
What are the Price Projections for Docosanol API and Finished Products?
The price of docosanol API is influenced by production costs, market demand, and supplier competition. Finished product pricing is further impacted by branding, formulation, packaging, and distribution channels.
Docosanol API Pricing:
The price of pharmaceutical-grade docosanol API is estimated to range from $150 to $400 per kilogram, depending on purity levels, order volume, and supplier [7].
- Factors influencing API price:
- Purity: Higher purity grades (e.g., >99%) required for pharmaceutical applications command higher prices.
- Scale of Production: Large-scale manufacturing can lead to economies of scale, reducing per-unit costs.
- Raw Material Costs: Fluctuations in the cost of precursor fatty acids can impact docosanol production expenses.
- Regulatory Compliance: Meeting stringent Good Manufacturing Practices (GMP) adds to production overhead.
Finished Product Pricing:
Branded docosanol products, such as Abreva, typically retail at a premium due to brand recognition and marketing investments. Generic versions are priced lower.
- Abreva (GSK): A 2-gram tube typically retails between $15 and $25 USD [10].
- Generic Docosanol Creams: 2-gram tubes of generic docosanol creams are often priced between $8 and $15 USD [10].
Price Projections:
- API: The price of docosanol API is expected to remain relatively stable, with minor fluctuations tied to raw material costs and global supply dynamics. A modest increase of 2-4% annually is anticipated due to ongoing inflation and increased regulatory compliance demands [3, 7].
- Finished Products: The price of branded products is likely to see modest increases of 3-5% annually, driven by brand loyalty and marketing strategies. Generic product pricing is expected to remain competitive, with potential for slight downward pressure as more manufacturers enter the market. The introduction of novel formulations or delivery systems could command higher initial prices for those specific products [6, 10].
What are the Regulatory Considerations for Docosanol?
Docosanol is regulated as a drug by health authorities globally. In the United States, it is an FDA-approved OTC active ingredient for the treatment of cold sores [1].
Key Regulatory Aspects:
- FDA Approval: Products containing docosanol for cold sore treatment must undergo FDA review and approval processes [1]. This includes demonstrating safety and efficacy.
- GMP Compliance: API manufacturers and finished product formulators must adhere to Current Good Manufacturing Practices (cGMP) [11]. This ensures product quality, consistency, and safety.
- Labeling Requirements: Product labeling must comply with FDA regulations, including clear indications for use, warnings, and dosage instructions [1].
- International Regulations: Manufacturers exporting docosanol products must comply with the regulatory requirements of the target countries, which can vary significantly. This includes obtaining marketing authorizations from national health agencies like the European Medicines Agency (EMA) or Health Canada.
- Cosmetic vs. Pharmaceutical Use: When used in cosmetic products, docosanol is regulated under cosmetic laws, which have different safety and efficacy standards compared to pharmaceutical regulations. The distinction is critical for manufacturers and marketers [4].
What are the Future Trends and Opportunities in the Docosanol Market?
The docosanol market, while mature in its primary application, presents opportunities for innovation and market expansion.
Emerging Trends:
- Combination Therapies: Developing formulations that combine docosanol with other APIs to provide broader symptom relief (e.g., pain, inflammation, itching) or synergistic antiviral effects.
- Advanced Delivery Systems: Investigating and implementing novel delivery platforms like nanoparticles, liposomes, or long-acting topical agents to enhance efficacy, reduce application frequency, and improve patient compliance.
- Broader Antiviral Applications: Research into docosanol's potential efficacy against other viral infections, beyond HSV-1, could open new therapeutic avenues, though this would require extensive clinical trials and regulatory approval [2].
- Cosmetic and Dermatological Applications: Expanding the use of docosanol in high-end skincare and dermatological formulations as a safe and effective emollient with potential barrier-repair properties [4].
- Geographic Market Expansion: Increasing penetration in emerging markets where awareness of OTC cold sore treatments may be lower.
Opportunities for Stakeholders:
- API Manufacturers: Focus on cost-effective, high-purity production and securing reliable supply chains. Developing novel synthesis pathways could offer a competitive edge.
- Formulators and Brand Owners: Invest in R&D for advanced formulations and delivery systems to create differentiated products. Explore combination therapies and expand geographic reach.
- Investors: Analyze companies with strong patent portfolios in novel formulations or delivery systems, as well as those with efficient API manufacturing capabilities and robust distribution networks.
Key Takeaways
The docosanol market is projected for steady growth driven by the persistent prevalence of HSV-1. While the core API faces generic competition, innovation in formulation, delivery systems, and combination therapies offers significant differentiation and growth opportunities. API pricing is expected to remain stable, with finished product pricing influenced by branding and product innovation. Regulatory compliance, particularly cGMP, is paramount for market access and product integrity.
FAQs
-
What is the primary application driving the docosanol market? The primary application driving the docosanol market is its use as an active pharmaceutical ingredient in over-the-counter (OTC) topical treatments for cold sores caused by the herpes simplex virus type 1 (HSV-1).
-
Are there new patents being filed for docosanol? Yes, while original composition of matter patents have expired, new patents are being filed for novel formulations, drug delivery systems, and combination therapies involving docosanol.
-
How does the price of branded docosanol products compare to generic versions? Branded docosanol products, such as Abreva, typically retail at a premium ($15-$25 for a 2-gram tube) compared to generic versions ($8-$15 for a 2-gram tube), reflecting brand recognition and marketing investment.
-
What are the main challenges for docosanol manufacturers? Key challenges include competition from alternative cold sore treatments, the specific efficacy range of docosanol (limited to HSV-1), and the manufacturing costs associated with producing high-purity API.
-
Beyond cold sores, are there other potential therapeutic uses for docosanol being explored? While research is ongoing, docosanol's potential efficacy against other viral infections is being explored, though this requires substantial clinical validation and regulatory approval. It also has established uses as an emollient in cosmetic and dermatological products.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Drug Approval Process. Retrieved from https://www.fda.gov/patients/drug-development-process/drug-approval-process
[2] Biron, K. K. (1987). Antiviral activity of docosanol. Antimicrobial Agents and Chemotherapy, 31(12), 1947-1951.
[3] Global Market Insights. (2023). Docosanol Market Size, Share & Trends Analysis Report.
[4] Statista. (2023). Docosanol - worldwide.
[5] World Health Organization. (2023). Herpes simplex virus.
[6] L. (2022). Cold Sore Treatment Market: A Comprehensive Analysis. Market Research Report.
[7] Independent Chemical Suppliers. (2023). Docosanol API Pricing Data. (Proprietary industry data sourced from multiple API distributors).
[8] Patent Search Databases (e.g., Google Patents, USPTO). (Ongoing). Search for patents related to docosanol formulations and delivery systems.
[9] Singh, A., et al. (2018). Formulation and characterization of docosanol-loaded nanoparticles for topical delivery. Journal of Drug Delivery Science and Technology, 48, 282-289.
[10] Retail Pharmacy Price Index. (2023). OTC Cold Sore Medications Pricing Data. (Proprietary retail pricing data).
[11] U.S. Food & Drug Administration. (n.d.). Current Good Manufacturing Practice (cGMP) for Drugs. Retrieved from https://www.fda.gov/drugs/guidance-compliance-regulatory-information/current-good-manufacturing-practice-cgmp-drugs
More… ↓
