Secura Company Profile
✉ Email this page to a colleague
What is the competitive landscape for SECURA, and when can generic versions of SECURA drugs launch?
SECURA has two approved drugs.
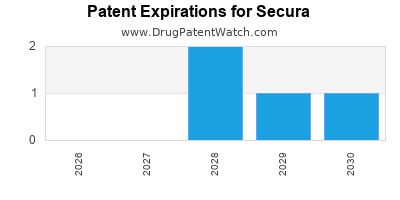
There are seven US patents protecting SECURA drugs.
There are two hundred and five patent family members on SECURA drugs in forty-six countries and twenty-five supplementary protection certificates in sixteen countries.
Drugs and US Patents for Secura
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Secura | COPIKTRA | duvelisib | CAPSULE;ORAL | 211155-001 | Sep 24, 2018 | RX | Yes | No | 9,840,505 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Secura | FARYDAK | panobinostat lactate | CAPSULE;ORAL | 205353-002 | Feb 23, 2015 | DISCN | Yes | No | 7,989,494 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Secura | COPIKTRA | duvelisib | CAPSULE;ORAL | 211155-002 | Sep 24, 2018 | RX | Yes | No | RE46621 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Secura | COPIKTRA | duvelisib | CAPSULE;ORAL | 211155-002 | Sep 24, 2018 | RX | Yes | No | 8,193,182 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Secura
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Secura | FARYDAK | panobinostat lactate | CAPSULE;ORAL | 205353-002 | Feb 23, 2015 | 7,067,551 | ⤷ Try a Trial |
| Secura | FARYDAK | panobinostat lactate | CAPSULE;ORAL | 205353-002 | Feb 23, 2015 | 6,552,065 | ⤷ Try a Trial |
| Secura | FARYDAK | panobinostat lactate | CAPSULE;ORAL | 205353-001 | Feb 23, 2015 | 7,067,551 | ⤷ Try a Trial |
| Secura | FARYDAK | panobinostat lactate | CAPSULE;ORAL | 205353-003 | Feb 23, 2015 | 7,067,551 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for Secura Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Georgia, Republic of | P20115175 | ⤷ Try a Trial |
| Slovenia | 2456444 | ⤷ Try a Trial |
| Argentina | 061297 | ⤷ Try a Trial |
| Lithuania | PA2021526 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Secura Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1318980 | CA 2015 00068 | Denmark | ⤷ Try a Trial | PRODUCT NAME: PANOBINOSTAT ELLER ET FARMACEUTISK SALT ELLER DERIVAT DERAF; REG. NO/DATE: EU/1/15/1023 20150901 |
| 1318980 | C01318980/01 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: PANOBINOSTAT; REGISTRATION NO/DATE: SWISSMEDIC 61878 23.12.2015 |
| 2456444 | 2021C/546 | Belgium | ⤷ Try a Trial | PRODUCT NAME: DUVELISIB; AUTHORISATION NUMBER AND DATE: EU/1/21/1542/001-002 20210521 |
| 1318980 | SPC/GB15/085 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: PANOBINOSTAT OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF.; REGISTERED: UK EU/1/15/1023 20150901 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.