RECORDATI RARE Company Profile
✉ Email this page to a colleague
What is the competitive landscape for RECORDATI RARE, and what generic alternatives to RECORDATI RARE drugs are available?
RECORDATI RARE has twelve approved drugs.
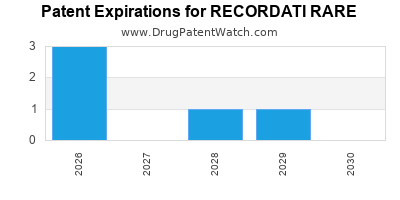
There are eleven US patents protecting RECORDATI RARE drugs.
There are two hundred and eighty-three patent family members on RECORDATI RARE drugs in sixty countries and fifty-one supplementary protection certificates in nineteen countries.
Summary for RECORDATI RARE
| International Patents: | 283 |
| US Patents: | 11 |
| Tradenames: | 12 |
| Ingredients: | 12 |
| NDAs: | 12 |
Drugs and US Patents for RECORDATI RARE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Recordati Rare | ISTURISA | osilodrostat phosphate | TABLET;ORAL | 212801-002 | Mar 6, 2020 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Recordati Rare | SIGNIFOR | pasireotide diaspartate | SOLUTION;SUBCUTANEOUS | 200677-001 | Dec 14, 2012 | RX | Yes | No | 8,299,209 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Recordati Rare | ISTURISA | osilodrostat phosphate | TABLET;ORAL | 212801-002 | Mar 6, 2020 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Recordati Rare | ISTURISA | osilodrostat phosphate | TABLET;ORAL | 212801-001 | Mar 6, 2020 | RX | Yes | No | 8,835,646 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Recordati Rare | ISTURISA | osilodrostat phosphate | TABLET;ORAL | 212801-002 | Mar 6, 2020 | RX | Yes | No | 8,835,646 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Recordati Rare | ISTURISA | osilodrostat phosphate | TABLET;ORAL | 212801-001 | Mar 6, 2020 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Recordati Rare | CHEMET | succimer | CAPSULE;ORAL | 019998-002 | Jan 30, 1991 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for RECORDATI RARE
Paragraph IV (Patent) Challenges for RECORDATI RARE drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Injection | 10 mg/mL, 2 mL vial | ➤ Subscribe | 2010-10-01 |
International Patents for RECORDATI RARE Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Tunisia | SN08082 | ⤷ Try a Trial |
| Poland | 1648934 | ⤷ Try a Trial |
| Ukraine | 96737 | ⤷ Try a Trial |
| Russian Federation | 2006120484 | ⤷ Try a Trial |
| South Korea | 20170029491 | ⤷ Try a Trial |
| Norway | 340854 | ⤷ Try a Trial |
| Croatia | P20130797 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for RECORDATI RARE Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2523731 | CR 2020 00025 | Denmark | ⤷ Try a Trial | PRODUCT NAME: OSILODROSTAT ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF, HERUNDER OSILODROSTATDIHYDROGENFOSFAT; REG. NO/DATE: EU/1/19/1407 20200113 |
| 1919458 | 2014C/018 | Belgium | ⤷ Try a Trial | PRODUCT NAME: CYSTEAMINE; AUTHORISATION NUMBER AND DATE: EU/1/13/861 20130910 |
| 2523731 | SPC/GB20/034 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: OSILODROSTAT, OR OSILODROSTAT PHOSPHATE; REGISTERED: UK EU/1/19/1407/001(NI) 20200113; UK EU/1/19/1407/002(NI) 20200113; UK EU/1/19/1407/003(NI) 20200113; UK PLGB 15266/0029-0001 20200113; UK PLGB 15266/0030-0001 20200113; UK PLGB 15266/0031-0001 20200113 |
| 2523731 | 24/2020 | Austria | ⤷ Try a Trial | PRODUCT NAME: OSILODROSTAT ODER PHARMAZEUTISCH AKZEPTABLES SALZ DAVON, EINSCHLIESSLICH OSILODROSTATDIHYDROGENPHOSPHAT; REGISTRATION NO/DATE: EU/1/19/1407 (MITTEILUNG) 20200113 |
| 2523731 | 20C1023 | France | ⤷ Try a Trial | PRODUCT NAME: OSILODROSTAT OU UN SEL PHARMACEUTIQUEMENT ACCEPTABLE DE CELUI-CI, Y COMPRIS LE DIHYDROGENOPHOSPHATE D'OSILODROSTAT; REGISTRATION NO/DATE: EU/1/19/1407 20200113 |
| 2523731 | 2090024-7 | Sweden | ⤷ Try a Trial | PRODUCT NAME: OSILODROSTAT OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, INCLUDING OSILODROSTAT DIHYDROGEN PHOSPHATE; REG. NO/DATE: EU/1/19/1407 20200113 |
| 1919458 | C300649 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: CYSTEAMINE; REGISTRATION NO/DATE: EU/1/13/861/001-002 20130906 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.