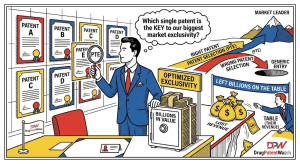
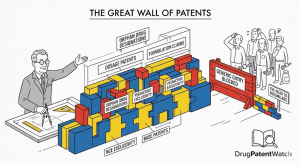
Pick the Right Patent Or Leave Billions on the Table: The Definitive Guide to the One-Patent-Per-Product Rule and PTE Selection Strategy
The Decision That Defines Decades of Revenue Somewhere between the FDA approval notice hitting a pharmaceutical company’s inbox and the […]