PAR PHARM INC Company Profile
✉ Email this page to a colleague
What is the competitive landscape for PAR PHARM INC, and what generic alternatives to PAR PHARM INC drugs are available?
PAR PHARM INC has thirty-eight approved drugs.
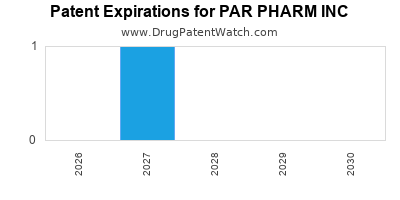
There is one US patent protecting PAR PHARM INC drugs. There are seven tentative approvals on PAR PHARM INC drugs.
There are nine patent family members on PAR PHARM INC drugs in eight countries and sixty-one supplementary protection certificates in fourteen countries.
Drugs and US Patents for PAR PHARM INC
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Par Pharm Inc | ETHACRYNIC ACID | ethacrynic acid | TABLET;ORAL | 208501-001 | Jul 21, 2017 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Par Pharm Inc | NUBAIN | nalbuphine hydrochloride | INJECTABLE;INJECTION | 018024-002 | May 27, 1982 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Par Pharm Inc | CHLORZOXAZONE | chlorzoxazone | TABLET;ORAL | 212743-002 | Apr 29, 2021 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Par Pharm Inc | ASPIRIN AND DIPYRIDAMOLE | aspirin; dipyridamole | CAPSULE, EXTENDED RELEASE;ORAL | 207944-001 | Jan 18, 2017 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Par Pharm Inc | TOLVAPTAN | tolvaptan | TABLET;ORAL | 206119-001 | Feb 15, 2022 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for PAR PHARM INC Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Japan | 5378788 | ⤷ Try a Trial |
| Canada | 2605413 | ⤷ Try a Trial |
| South Korea | 20080000660 | ⤷ Try a Trial |
| World Intellectual Property Organization (WIPO) | 2006115626 | ⤷ Try a Trial |
| China | 101163679 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for PAR PHARM INC Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2330892 | 2016/064 | Ireland | ⤷ Try a Trial | PRODUCT NAME: GLYCEROL PHENYLBUTYRATE; REGISTRATION NO/DATE: EU/1/15/1062 20151201 |
| 0526708 | 0290017-3 | Sweden | ⤷ Try a Trial | PRODUCT NAME: BOSENTAN ELLER ETT SALT DAERAV; NAT. REGISTZRATION NO/DATE: EU/1/02/220/001 20020515; FIRST REGISTRATION: CH 55841 01 20020228 |
| 2101777 | 122016000039 | Germany | ⤷ Try a Trial | PRODUCT NAME: AMBRISENTAN IN VERWENDUNG IN DER KOMBINATIONSTHERAPIE MIT TADALAFIL; REGISTRATION NO/DATE: EU/1/08/451 20151120 |
| 0237929 | 68/1997 | Austria | ⤷ Try a Trial | PRODUCT NAME: TOLCAPONE; REGISTRATION NO/DATE: EU/1/97/044/001 - EU/1/97/044/006 19970827 |
| 2101777 | 93081 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: AMBRISENTAN UTILISE EN TRAITEMENT COMBINE AVEC TADALAFIL; FRIST REGISTRATION: 20151125 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.