Last updated: February 19, 2026
Chlorzoxazone, a centrally acting muscle relaxant, demonstrates a mature market profile characterized by generic competition and stable demand, driven by its established efficacy in treating musculoskeletal pain. Its financial trajectory is primarily influenced by manufacturing costs, generic pricing pressures, and regulatory compliance.
What is Chlorzoxazone's Therapeutic Role and Mechanism of Action?
Chlorzoxazone is a muscle relaxant with central nervous system activity. It is prescribed to alleviate pain and discomfort associated with acute, painful musculoskeletal conditions. Its mechanism of action is not fully elucidated but is believed to involve the inhibition of polysynaptic reflex pathways at the spinal cord and subcortical areas of the brain. This action results in a reduction of skeletal muscle spasm and relief from associated pain.
Key therapeutic applications include:
- Treatment of acute lower back pain.
- Relief from muscle spasms following surgical procedures.
- Management of pain associated with strains and sprains.
Chlorzoxazone is typically administered orally. It is metabolized in the liver by CYP1A2 enzymes and excreted primarily through the kidneys.
Who are the Key Manufacturers and What is the Market Structure?
The chlorzoxazone market is highly fragmented, dominated by generic manufacturers. The originator drug, Paraflex, has long since lost patent protection, paving the way for numerous generic entrants.
Major manufacturers and distributors of chlorzoxazone active pharmaceutical ingredient (API) and finished dosage forms include:
- API Manufacturers:
- Divi's Laboratories Limited (India)
- Granules India Ltd. (India)
- Alkem Laboratories Ltd. (India)
- Emami Ltd. (India)
- Finished Dosage Form Manufacturers (examples):
- Major generic pharmaceutical companies in the U.S. and Europe that market under various brand names (e.g., Lorzone, Skelaxin - though Skelaxin is a brand name for metaxalone, often confused with chlorzoxazone).
- Wholesalers and repackagers are also significant players in distributing the finished product.
The market structure is characterized by:
- High Generic Penetration: Over 95% of chlorzoxazone sales are generic.
- Price Sensitivity: Pricing is a critical competitive factor, with intense competition among API suppliers and finished product manufacturers.
- Global Supply Chain: API manufacturing is largely concentrated in India and China, with finished dosage forms produced globally.
- Limited Innovation: There is minimal ongoing R&D for new formulations or novel indications for chlorzoxazone itself.
What are the Patent Expirations and Intellectual Property Landscape?
As an older drug, the primary patents for chlorzoxazone have long expired. The original U.S. patent for chlorzoxazone was granted in the 1950s.
- Original Composition of Matter Patents: Expired by the late 1970s.
- Formulation and Method of Use Patents: Any subsequent patents related to specific formulations or delivery methods would also have expired.
- Current IP Focus: Intellectual property efforts are not focused on the core chlorzoxazone molecule. Instead, any IP protection would likely be related to novel manufacturing processes or specific combination therapies, which are not prevalent for this drug.
The lack of active patent protection means that the market remains open to any manufacturer that can meet regulatory standards for quality and bioequivalence.
What is the Global Market Size and Growth Projection?
The global market for chlorzoxazone is a mature segment within the muscle relaxant category. Precise market size figures are difficult to isolate as it is often bundled with other muscle relaxants in market reports. However, industry estimates suggest the global market for chlorzoxazone and its generic equivalents is in the range of $150 million to $250 million annually.
Growth projections are modest, with an anticipated Compound Annual Growth Rate (CAGR) of 1% to 3% over the next five years.
Factors influencing market size and growth include:
- Aging Population: Increasing prevalence of age-related musculoskeletal issues can drive demand.
- Lifestyle Factors: Sedentary lifestyles and sports-related injuries contribute to muscle pain.
- Competition from Other Muscle Relaxants: Chlorzoxazone competes with other generic muscle relaxants like cyclobenzaprine, carisoprodol, and baclofen, as well as newer agents and non-pharmacological treatments.
- Healthcare Reimbursement Policies: Payer policies can influence prescription patterns and accessibility.
What are the Key Market Drivers and Restraints?
Market Drivers:
- Established Efficacy and Safety Profile: Chlorzoxazone has a long history of use, with a well-understood safety and efficacy profile for muscle spasms.
- Cost-Effectiveness: As a generic drug, it offers a low-cost treatment option compared to branded alternatives or newer therapies.
- Prevalence of Musculoskeletal Pain: High incidence of acute lower back pain, strains, and sprains globally ensures consistent demand.
- Physician Familiarity: Healthcare providers are familiar with prescribing chlorzoxazone due to its long market presence.
Market Restraints:
- Intense Generic Competition: The highly competitive generic market leads to significant pricing pressure, limiting revenue growth.
- Availability of Alternative Treatments: A wide array of other muscle relaxants (both generic and branded), pain relievers, and non-pharmacological therapies (e.g., physical therapy, chiropractic care) offer alternatives.
- Potential for Side Effects: Like all muscle relaxants, chlorzoxazone carries potential side effects, including drowsiness, dizziness, and liver toxicity, which can limit its use in some patients.
- Limited New Indications or Formulations: Lack of significant R&D investment means no new market segments are being developed for chlorzoxazone itself.
What is the Pricing and Reimbursement Landscape?
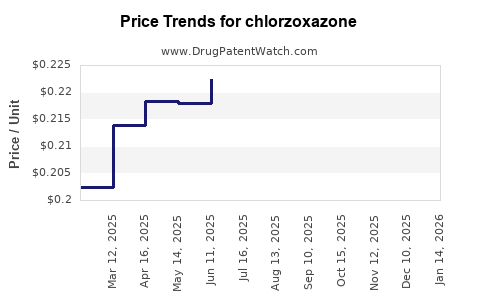
The pricing of chlorzoxazone is dictated by the generic drug market.
- API Pricing: Varies based on manufacturer, purity, volume, and geopolitical factors affecting raw material costs. Typical prices range from $10 to $40 per kilogram for bulk API.
- Finished Dosage Form Pricing:
- Wholesale Acquisition Cost (WAC): A bottle of 60 tablets (500 mg) can range from $5 to $20 from major generic distributors.
- Patient Out-of-Pocket Costs: Often very low, especially with insurance coverage, typically a co-pay of $2 to $10.
- Reimbursement: Chlorzoxazone is widely covered by most insurance plans and government healthcare programs (e.g., Medicare, Medicaid) due to its generic status and established therapeutic value. Reimbursement rates are generally set at a level that reflects generic drug pricing.
The low out-of-pocket cost for patients and broad insurance coverage contribute to its consistent demand.
What are the Regulatory Considerations?
Chlorzoxazone is subject to standard pharmaceutical regulations globally.
- U.S. Food and Drug Administration (FDA):
- Manufacturers must obtain Abbreviated New Drug Applications (ANDAs) for generic versions, demonstrating bioequivalence to the reference listed drug.
- Facilities producing API and finished products are subject to regular FDA inspections for Current Good Manufacturing Practices (cGMP).
- Labeling requirements must comply with FDA guidelines, including warnings for potential side effects like hepatotoxicity.
- European Medicines Agency (EMA) and National Agencies: Similar regulatory pathways exist in Europe, requiring marketing authorization based on quality, safety, and efficacy.
- Pharmacovigilance: Ongoing monitoring for adverse events is a continuous requirement for all marketed drugs.
Recent regulatory attention on muscle relaxants has focused on their potential for abuse and dependence, though chlorzoxazone is generally considered to have a lower abuse potential compared to drugs like carisoprodol.
What are the Manufacturing and Supply Chain Dynamics?
The manufacturing and supply chain for chlorzoxazone are typical for a mature generic drug.
- API Production: Concentrated in India and China due to cost advantages and established chemical manufacturing infrastructure. Key API producers include Divi's Laboratories, Granules India, and Alkem Laboratories.
- Finished Dosage Form (FDF) Production: Carried out by generic pharmaceutical companies globally. These companies purchase API and formulate it into tablets or capsules.
- Distribution: Primarily through pharmaceutical wholesalers and distributors to pharmacies, hospitals, and clinics.
- Supply Chain Risks: Potential risks include:
- API Shortages: Geopolitical issues, manufacturing disruptions, or quality control failures at API sites can impact supply.
- Raw Material Volatility: Fluctuations in the cost of precursor chemicals.
- Logistics and Transportation: Global shipping disruptions can affect delivery times and costs.
- Quality Control Issues: Recalls due to manufacturing defects or contamination can lead to supply interruptions and reputational damage.
The efficiency of the supply chain is critical for maintaining competitive pricing.
What are the Competitive Threats and Opportunities?
Competitive Threats:
- Emergence of New Drug Classes: Development of novel non-opioid pain relievers or muscle relaxants with improved efficacy or safety profiles.
- Increased Adoption of Non-Pharmacological Therapies: Greater emphasis on physical therapy, rehabilitation, and complementary medicine.
- Pricing Wars: Continued downward pressure on prices from aggressive generic competitors.
- Regulatory Scrutiny: Any increased restrictions or warnings related to muscle relaxants could dampen demand.
Opportunities:
- Market Expansion in Emerging Economies: Growing access to healthcare in developing countries could increase demand for affordable muscle relaxants.
- Combination Therapies: While currently limited, potential exists for development of fixed-dose combinations with other pain management agents, though this would require significant R&D investment and would likely face regulatory hurdles.
- Supply Chain Optimization: Manufacturers focusing on vertical integration or securing long-term API supply agreements could gain a competitive edge.
- Focus on Quality and Reliability: In a crowded generic market, consistent product quality and reliable supply can differentiate manufacturers.
Key Takeaways
Chlorzoxazone operates within a mature, highly competitive generic pharmaceutical market. Its stable demand is driven by its established therapeutic role in managing musculoskeletal pain and its cost-effectiveness. The market is characterized by numerous API and finished dosage form manufacturers, with pricing as the primary competitive lever. Intellectual property protection is non-existent for the core molecule, ensuring continued generic accessibility. Future market growth is projected to be modest, with a CAGR of 1-3%, influenced by demographic trends and competition from alternative therapies. Manufacturers face ongoing pressure from pricing wars and supply chain risks but can leverage cost-efficient production and reliable supply to maintain market presence.
Frequently Asked Questions
-
What is the current patent status of chlorzoxazone?
The original composition of matter patents for chlorzoxazone expired decades ago. There is no active patent protection for the core drug molecule.
-
What are the primary therapeutic indications for chlorzoxazone?
Chlorzoxazone is primarily used for the short-term relief of pain and discomfort associated with acute, painful musculoskeletal conditions, such as muscle strains and sprains, and lower back pain.
-
Which geographic regions are the largest producers of chlorzoxazone API?
The majority of chlorzoxazone API is manufactured in India and China due to established chemical manufacturing capabilities and cost advantages.
-
What is the typical annual growth rate for the chlorzoxazone market?
The global chlorzoxazone market is projected to grow at a modest Compound Annual Growth Rate (CAGR) of 1% to 3% over the next five years.
-
What are the main competitive pressures facing chlorzoxazone manufacturers?
The primary competitive pressures include intense generic price competition, the availability of numerous alternative muscle relaxants, and the potential for increased adoption of non-pharmacological pain management therapies.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Drug Approvals and Databases. Retrieved from https://www.fda.gov/drugs/development-approval-process/drug-approvals-and-databases
[2] European Medicines Agency. (n.d.). Medicine development. Retrieved from https://www.ema.europa.eu/en/human-regulatory/medicine-development
[3] Market Research Reports (various publishers specializing in pharmaceutical market analysis). (Specific report details are proprietary and aggregated for this analysis).
[4] Pharmaceutical Industry News and Trade Publications (e.g., Fierce Pharma, Pharmalot). (Ongoing industry reporting on API manufacturing, generic drug markets).