ninlaro Drug Patent Profile
✉ Email this page to a colleague
When do Ninlaro patents expire, and what generic alternatives are available?
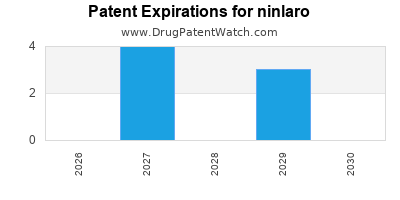
Ninlaro is a drug marketed by Takeda Pharms Usa and is included in one NDA. There are seven patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and eighty-seven patent family members in forty-four countries.
The generic ingredient in NINLARO is ixazomib citrate. One supplier is listed for this compound. Additional details are available on the ixazomib citrate profile page.
DrugPatentWatch® Generic Entry Outlook for Ninlaro
Ninlaro was eligible for patent challenges on November 20, 2019.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be June 16, 2029. This may change due to patent challenges or generic licensing.
There have been five patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for ninlaro?
- What are the global sales for ninlaro?
- What is Average Wholesale Price for ninlaro?
Summary for ninlaro
| International Patents: | 187 |
| US Patents: | 7 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 42 |
| Clinical Trials: | 48 |
| Drug Prices: | Drug price information for ninlaro |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for ninlaro |
| What excipients (inactive ingredients) are in ninlaro? | ninlaro excipients list |
| DailyMed Link: | ninlaro at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for ninlaro
Generic Entry Date for ninlaro*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
CAPSULE;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for ninlaro
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Genentech, Inc. | Phase 1/Phase 2 |
| Rajshekhar Chakraborty, MD | Phase 1/Phase 2 |
| Takeda | Phase 4 |
Pharmacology for ninlaro
| Drug Class | Proteasome Inhibitor |
| Mechanism of Action | Proteasome Inhibitors |
Paragraph IV (Patent) Challenges for NINLARO
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| NINLARO | Capsules | ixazomib citrate | 2.3 mg, 3 mg and 4 mg | 208462 | 1 | 2019-11-20 |
US Patents and Regulatory Information for ninlaro
ninlaro is protected by seven US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of ninlaro is ⤷ Start Trial.
This potential generic entry date is based on patent 8,859,504.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Takeda Pharms Usa | NINLARO | ixazomib citrate | CAPSULE;ORAL | 208462-001 | Nov 20, 2015 | RX | Yes | No | 8,871,745 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Takeda Pharms Usa | NINLARO | ixazomib citrate | CAPSULE;ORAL | 208462-003 | Nov 20, 2015 | RX | Yes | Yes | 7,442,830 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Takeda Pharms Usa | NINLARO | ixazomib citrate | CAPSULE;ORAL | 208462-001 | Nov 20, 2015 | RX | Yes | No | 7,687,662 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for ninlaro
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Takeda Pharms Usa | NINLARO | ixazomib citrate | CAPSULE;ORAL | 208462-003 | Nov 20, 2015 | 8,546,608 | ⤷ Start Trial |
| Takeda Pharms Usa | NINLARO | ixazomib citrate | CAPSULE;ORAL | 208462-002 | Nov 20, 2015 | 8,546,608 | ⤷ Start Trial |
| Takeda Pharms Usa | NINLARO | ixazomib citrate | CAPSULE;ORAL | 208462-002 | Nov 20, 2015 | 9,233,115 | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for ninlaro
When does loss-of-exclusivity occur for ninlaro?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 2162
Estimated Expiration: ⤷ Start Trial
Patent: 5554
Estimated Expiration: ⤷ Start Trial
Patent: 5566
Estimated Expiration: ⤷ Start Trial
Patent: 0011
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 09260778
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2014008750
Estimated Expiration: ⤷ Start Trial
Patent: 2014008753
Estimated Expiration: ⤷ Start Trial
Patent: 2014008754
Estimated Expiration: ⤷ Start Trial
Patent: 0915300
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 27862
Estimated Expiration: ⤷ Start Trial
Patent: 21946
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 10001475
Estimated Expiration: ⤷ Start Trial
China
Patent: 2066386
Estimated Expiration: ⤷ Start Trial
Patent: 3435638
Estimated Expiration: ⤷ Start Trial
Patent: 3450241
Estimated Expiration: ⤷ Start Trial
Patent: 3467565
Estimated Expiration: ⤷ Start Trial
Patent: 3497210
Estimated Expiration: ⤷ Start Trial
Patent: 3497232
Estimated Expiration: ⤷ Start Trial
Patent: 7253966
Estimated Expiration: ⤷ Start Trial
Patent: 7253975
Estimated Expiration: ⤷ Start Trial
Patent: 7266480
Estimated Expiration: ⤷ Start Trial
Patent: 7266482
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 21289
Estimated Expiration: ⤷ Start Trial
Costa Rica
Patent: 850
Estimated Expiration: ⤷ Start Trial
Patent: 160014
Estimated Expiration: ⤷ Start Trial
Patent: 160015
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0150592
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 16435
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 18419
Estimated Expiration: ⤷ Start Trial
Patent: 30581
Estimated Expiration: ⤷ Start Trial
Dominican Republic
Patent: 010000384
Estimated Expiration: ⤷ Start Trial
Ecuador
Patent: 11010763
Estimated Expiration: ⤷ Start Trial
Patent: 14010763
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 7346
Estimated Expiration: ⤷ Start Trial
Patent: 0685
Estimated Expiration: ⤷ Start Trial
Patent: 1170036
Estimated Expiration: ⤷ Start Trial
Patent: 1500430
Estimated Expiration: ⤷ Start Trial
Patent: 1500431
Estimated Expiration: ⤷ Start Trial
Patent: 1500432
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 18419
Estimated Expiration: ⤷ Start Trial
Patent: 30579
Estimated Expiration: ⤷ Start Trial
Patent: 30580
Estimated Expiration: ⤷ Start Trial
Patent: 30581
Estimated Expiration: ⤷ Start Trial
Patent: 33147
Estimated Expiration: ⤷ Start Trial
Patent: 36693
Estimated Expiration: ⤷ Start Trial
Finland
Patent: 18419
Estimated Expiration: ⤷ Start Trial
Georgia, Republic of
Patent: 0135847
Estimated Expiration: ⤷ Start Trial
Patent: 01706703
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 57786
Estimated Expiration: ⤷ Start Trial
Patent: 98252
Estimated Expiration: ⤷ Start Trial
Patent: 98253
Estimated Expiration: ⤷ Start Trial
Patent: 98254
Estimated Expiration: ⤷ Start Trial
Patent: 99260
Estimated Expiration: ⤷ Start Trial
Patent: 44809
Estimated Expiration: ⤷ Start Trial
Patent: 44810
Estimated Expiration: ⤷ Start Trial
Patent: 44811
Estimated Expiration: ⤷ Start Trial
Patent: 44827
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 0056
Estimated Expiration: ⤷ Start Trial
Patent: 4285
Estimated Expiration: ⤷ Start Trial
Patent: 4286
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 66380
Estimated Expiration: ⤷ Start Trial
Patent: 26319
Estimated Expiration: ⤷ Start Trial
Patent: 10066
Estimated Expiration: ⤷ Start Trial
Patent: 10813
Estimated Expiration: ⤷ Start Trial
Patent: 11524903
Estimated Expiration: ⤷ Start Trial
Patent: 14144981
Estimated Expiration: ⤷ Start Trial
Patent: 14169316
Estimated Expiration: ⤷ Start Trial
Patent: 14169317
Estimated Expiration: ⤷ Start Trial
Patent: 14196310
Estimated Expiration: ⤷ Start Trial
Patent: 17214396
Estimated Expiration: ⤷ Start Trial
Patent: 20055829
Estimated Expiration: ⤷ Start Trial
Patent: 21165288
Estimated Expiration: ⤷ Start Trial
Patent: 23158150
Estimated Expiration: ⤷ Start Trial
Patent: 25172854
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 5592
Estimated Expiration: ⤷ Start Trial
Patent: 1021
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 0186
Estimated Expiration: ⤷ Start Trial
Patent: 9769
Estimated Expiration: ⤷ Start Trial
Patent: 8222
Estimated Expiration: ⤷ Start Trial
Patent: 10013642
Estimated Expiration: ⤷ Start Trial
Morocco
Patent: 471
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 3236
Estimated Expiration: ⤷ Start Trial
Patent: 4121
Estimated Expiration: ⤷ Start Trial
Patent: 4122
Estimated Expiration: ⤷ Start Trial
Patent: 4123
Estimated Expiration: ⤷ Start Trial
Norway
Patent: 17016
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 100256
Estimated Expiration: ⤷ Start Trial
Patent: 141064
Estimated Expiration: ⤷ Start Trial
Patent: 141065
Estimated Expiration: ⤷ Start Trial
Patent: 141076
Estimated Expiration: ⤷ Start Trial
Patent: 141078
Estimated Expiration: ⤷ Start Trial
Philippines
Patent: 015501191
Estimated Expiration: ⤷ Start Trial
Patent: 015501192
Estimated Expiration: ⤷ Start Trial
Patent: 015501193
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 18419
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 18419
Estimated Expiration: ⤷ Start Trial
Patent: 30581
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 020
Patent: ORGANOBORNA ESTARSKA JEDINJENJA I NJIHOVE FARMACEUTSKE KOMPOZICIJE (BORONATE ESTER COMPOUNDS AND PHARMACEUTICAL COMPOSITIONS THEREOF)
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 201701524X
Patent: BORONATE ESTER COMPOUNDS AND PHARMACEUTICAL COMPOSITIONS THEREOF
Estimated Expiration: ⤷ Start Trial
Patent: 4349
Patent: BORONATE ESTER COMPOUNDS AND PHARMACEUTICAL COMPOSITIONS THEREOF
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 18419
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1009177
Patent: BORONATE ESTER COMPOUNDS AND PHARMACEUTICAL COMPOSITIONS THEREOF
Estimated Expiration: ⤷ Start Trial
Patent: 1109368
Patent: BORONATE ESTER COMPOUNDS AND PHARMACEUTICAL COMPOSITIONS THEREOF
Estimated Expiration: ⤷ Start Trial
Patent: 1304728
Patent: BORONATE ESTER CONPOUNDDS AND PHARMACEUTICAL COMPOSITIONS THEREOF
Estimated Expiration: ⤷ Start Trial
Patent: 1504133
Patent: BORONATE ESTER COMPOUNDS AND PHARMACEUTICAL COMPOSITIONS THEREOF
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1690571
Estimated Expiration: ⤷ Start Trial
Patent: 1691093
Estimated Expiration: ⤷ Start Trial
Patent: 1704694
Estimated Expiration: ⤷ Start Trial
Patent: 1741142
Estimated Expiration: ⤷ Start Trial
Patent: 1860743
Estimated Expiration: ⤷ Start Trial
Patent: 110043599
Estimated Expiration: ⤷ Start Trial
Patent: 140094663
Estimated Expiration: ⤷ Start Trial
Patent: 140107482
Estimated Expiration: ⤷ Start Trial
Patent: 140107483
Estimated Expiration: ⤷ Start Trial
Patent: 170068599
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 41467
Estimated Expiration: ⤷ Start Trial
Patent: 85114
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1002727
Patent: Boronate ester compounds and pharmaceutical compositions thereof
Estimated Expiration: ⤷ Start Trial
Patent: 1433317
Patent: Boronate ester compounds and pharmaceutical compositions thereof
Estimated Expiration: ⤷ Start Trial
Patent: 1433569
Patent: Boronate ester compounds and pharmaceutical compositions thereof
Estimated Expiration: ⤷ Start Trial
Patent: 98333
Estimated Expiration: ⤷ Start Trial
Patent: 42351
Estimated Expiration: ⤷ Start Trial
Patent: 43985
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 6205
Patent: СОЕДИНЕНИЯ БОРОНАТНОГО ЭФИРА, ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ (ВАРИАНТЫ), КОТОРАЯ СОДЕРЖИТ ИХ, И СПОСОБ ИХ ПОЛУЧЕНИЯ (ВАРИАНТЫ);СПОЛУКИ БОРОНАТНОГО ЕФІРУ, ФАРМАЦЕВТИЧНА КОМПОЗИЦІЯ (ВАРІАНТИ), ЯКА МІСТИТЬ ЇХ, І СПОСІБ ЇХ ОДЕРЖАННЯ (ВАРІАНТИ) (BORONATE ESTER COMPOUNDS, PHARMACEUTICAL COMPOSITIONS THEREOFAND PROCESS FOR THE PREPARATION)
Estimated Expiration: ⤷ Start Trial
Patent: 5131
Patent: СПОЛУКИ БОРОНАТНОГО ЕФІРУ І ЙОГО ФАРМАЦЕВТИЧНІ РЕЦЕПТУРИ
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering ninlaro around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Luxembourg | C00013 | ⤷ Start Trial | |
| Ukraine | 106205 | СОЕДИНЕНИЯ БОРОНАТНОГО ЭФИРА, ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ (ВАРИАНТЫ), КОТОРАЯ СОДЕРЖИТ ИХ, И СПОСОБ ИХ ПОЛУЧЕНИЯ (ВАРИАНТЫ);СПОЛУКИ БОРОНАТНОГО ЕФІРУ, ФАРМАЦЕВТИЧНА КОМПОЗИЦІЯ (ВАРІАНТИ), ЯКА МІСТИТЬ ЇХ, І СПОСІБ ЇХ ОДЕРЖАННЯ (ВАРІАНТИ) (BORONATE ESTER COMPOUNDS, PHARMACEUTICAL COMPOSITIONS THEREOFAND PROCESS FOR THE PREPARATION) | ⤷ Start Trial |
| Norway | 2017016 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for ninlaro
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2178888 | 17C1011 | France | ⤷ Start Trial | PRODUCT NAME: IXAZOMIB ET SES SELS PHARMACEUTIQUEMENT ACCEPTABLES ET SES ESTERS TELS QUE LE CITRATE D'IXAZOMIB; REGISTRATION NO/DATE: EU/1/16/1094 20161123 |
| 2178888 | PA2017010,C2178888 | Lithuania | ⤷ Start Trial | PRODUCT NAME: IKSAZOMIBAS ARBA JO FARMACINIU POZIURIU PRIIMTINA DRUSKA IR ESTERIS, TOKS KAIP IKSAZOMIBO CITRATAS; REGISTRATION NO/DATE: EU/1/16/1094 20161121 |
| 2178888 | CA 2017 00014 | Denmark | ⤷ Start Trial | PRODUCT NAME: IXAZOMIB OG FARMACEUTISK ACCEPTABLE SALTE DERAF, HERUNDER IXAZOMIBCITRAT; REG. NO/DATE: EU/1/16/1094 20161123 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
NINLARO: Market Dynamics and Financial Trajectory
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.