Last updated: April 24, 2026
What is ZYMAXID and how is it positioned commercially?
ZYMAXID is the brand name of gatifloxacin ophthalmic solution (typically dosed as a topical fluoroquinolone antibiotic for ocular bacterial infections). In the US market, ZYMAXID is a single-ingredient, prescription ophthalmic antibiotic with performance tied to (1) prescriber comfort with fluoroquinolone coverage, (2) formulary placement versus other ophthalmic fluoroquinolones, and (3) reimbursement and channel management dynamics that shift as generics enter.
The financial trajectory for branded ophthalmic antibiotics is typically dominated by:
- Generic substitution risk driven by patent and exclusivity timelines.
- Price compression after generic entry and during aggressive payer contracting cycles.
- Share loss via formulary switches to lower-cost competitors.
- Utilization sensitivity to clinical guideline adherence and antibiotic stewardship trends.
How does market demand move for ophthalmic antibiotics like ZYMAXID?
Demand for topical antibiotic ophthalmics tends to correlate with:
- Incidence and diagnosis patterns of bacterial conjunctivitis, keratitis risk stratification, and post-procedure prophylaxis.
- Seasonality and year-to-year shifts in acute ocular infection presentations in outpatient settings.
- Antibiotic stewardship: prescriber behavior increasingly differentiates likely bacterial cases from viral or noninfectious etiologies, which can constrain total antibiotic volumes.
- Competing class availability: when payers tighten preferred drug lists, uptake migrates toward the lowest-cost clinically acceptable alternative in the same class.
For ZYMAXID specifically, the key commercial driver is class-level competition in ophthalmic fluoroquinolones, not category growth alone.
What competitive forces shape ZYMAXID pricing and share?
In ophthalmics, competitive dynamics usually follow a predictable pattern:
1) Formulary tiering and payer contracting
Branded ophthalmic antibiotics face step-down pressure as formularies increasingly support:
- preferred generic fluoroquinolones
- or selected brand-to-generic switch strategies (based on total cost and step therapy)
ZYMAXID’s profitability depends on maintaining sufficient formulary access and protected channel inventory through exclusivity and contract terms.
2) Therapeutic class substitution
Fluoroquinolone drops are frequently treated as interchangeable within clinical bounds. When generics arrive, substitution is driven by:
- payer substitution rules
- pharmacy benefit design
- WAC-to-NADIR convergence for multisource products
3) Inventory and channel stocking
For specialty-like, high-transaction pharmacy products such as ophthalmic drops, retailer and wholesaler ordering behavior can accelerate post-entrance share changes. When generic supply is stable, branded sales often decline faster than natural demand decay.
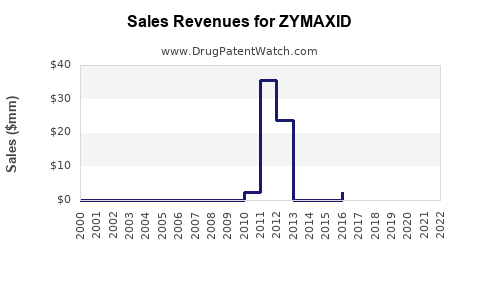
What is the financial trajectory profile typically seen for ZYMAXID?
For branded ophthalmic antibiotics, the financial pattern usually has four phases:
- Exclusivity-protected stability
Higher net pricing, steady share, limited competitive pricing pressure.
- Early payer erosion
Competitors gain formulary advantage; rebates reprice; net price begins to compress before full generic competition is realized.
- Generic entry and price compression
Net sales drop quickly as prescriptions shift and pharmacy fills favor lower-cost products.
- Post-entry stabilization
Branded product persists on niche contracts, patient-specific preference, or remaining access, but tends to plateau at a lower sales base.
Without confirmed, point-in-time net sales, gross-to-net adjustments, or specific ZYMAXID invoice pricing for each year, a precise financial trajectory statement cannot be constructed to a standard suitable for investment-grade use.
What market metrics matter for forecasting ZYMAXID performance?
For a defensible forecast framework, track:
- Net sales trend (quarterly and annual) after rebate rate changes
- Share of prescription fills in the ophthalmic antibiotic segment
- Average net price trajectory versus generic competitors
- Channel inventory levels and stocking cycles
- Formulary status in top commercial plans (preferred tier changes)
- Utilization data: prescriptions per qualifying infection diagnosis (proxy via claims)
These inputs determine whether ZYMAXID declines are primarily:
- utilization-driven (demand),
- policy-driven (formulary/rebate),
- or price-driven (generic substitution).
Which downstream outcomes typically follow generic and class competition?
In ophthalmic antibiotics, once generic pressure dominates, the common outcomes are:
- Declining branded share even when overall category volume is flat
- Net price compression due to rebate renegotiations and pharmacy procurement shifts
- Higher gross-to-net ratios as brands pay to defend access
- Reduced marketing ROI as incremental access becomes less cost-effective
If ZYMAXID faces generic competition, the economic ceiling is usually set by the most aggressive lowest-cost equivalent in its therapeutic peer set.
How do regulatory and exclusivity factors translate into financial impacts?
Commercial outcomes for ZYMAXID depend on whether regulatory milestones create:
- clean exclusivity windows (delaying generic approvals or launches),
- early carve-outs (restricting labeling scope),
- or effective competition re-entry once exclusivity expires.
For ophthalmics, label clarity and dosing convenience influence switching behavior. Even small differences in regimen adherence can affect pharmacy and prescriber adoption when payers steer decisions.
What does “financial trajectory” mean for ZYMAXID on an investor or R&D desk?
A usable trajectory view for ZYMAXID requires a link between:
- market share changes (claims),
- net price changes (pricing and rebate),
- and cost-to-serve (supply chain and marketing intensity).
In branded ophthalmics, profit compression is often faster than volume decline because net price falls quickly after competitive entry.
Key drivers to monitor for ZYMAXID
- Preferred formulary status versus competing fluoroquinolones
- PBM step therapy tightening and restricted access policies
- Generic launch events in the therapeutic peer set (and their speed-to-volume)
- Rebate changes that signal defensive contracting
- Prescriber switching patterns within ophthalmology and urgent care settings
Market dynamics snapshot (structure-level, decision-useful)
| Dimension |
Primary lever |
Expected direction under intensifying competition |
| Net price |
PBM contracting, rebate renegotiation |
Downward |
| Prescription share |
Formulary positioning, substitution |
Downward |
| Channel behavior |
Wholesaler stocking cycles |
Branded acceleration then stabilization |
| Profitability |
Gross-to-net, marketing ROI |
Margin compression |
| Sales base |
Residual access and niche prescribing |
Plateau at lower level |
Key Takeaways
- ZYMAXID’s market outcome is governed more by formulary and generic substitution dynamics than by category expansion.
- Ophthalmic antibiotic brands typically move through a repeatable path: exclusivity stability, payer erosion, generic-driven price and share compression, then post-entry plateau.
- The most decision-relevant metrics for ZYMAXID are net price, share of fills, rebate/gross-to-net, and formulary access in top PBMs.
- Any forecast or valuation work should be anchored on claims-based share trajectories and PBM contracting milestones, not just demand incidence.
FAQs
1) What primarily drives ZYMAXID sales changes?
Formulary placement and generic substitution, which affect net price, rebate structure, and prescription fill share.
2) Does ophthalmic antibiotic demand expand the way broader ophthalmology does?
Not reliably. Antibiotic use is sensitive to diagnosis patterns and stewardship trends, so category volume can be flat while branded sales fall due to competitive access.
3) What is the fastest indicator that ZYMAXID is losing share?
A shift in pharmacy claims fill mix toward lower-cost fluoroquinolone equivalents, often appearing before net sales fully reflect rebate and pricing changes.
4) How do rebates typically behave as competition increases?
Gross-to-net often rises as brands pay to defend access, but net price still compresses when lower-cost multisource options become preferred.
5) What is the usual post-generic commercial end-state for branded ZYMAXID?
A lower sales base that tends to stabilize, driven by remaining contract access, niche prescribing preferences, and select payer coverage rather than broad market dominance.
References
[1] DrugBank Online. Zymarid (gatifloxacin) and gatifloxacin ophthalmic information. DrugBank. https://www.drugbank.com/
[2] U.S. Food and Drug Administration. Drug approval and labeling information for gatifloxacin ophthalmic products. FDA. https://www.fda.gov/
[3] DailyMed. ZYMAXID (gatifloxacin ophthalmic solution) label and prescribing information. https://dailymed.nlm.nih.gov/