Last updated: February 19, 2026
Zolmitriptan, a selective serotonin receptor agonist, demonstrates a stable market position primarily driven by its established efficacy in acute migraine treatment. Patent expirations have led to generic competition, significantly impacting pricing and market share dynamics. The financial trajectory is characterized by a mature product lifecycle, with revenue streams reliant on volume rather than premium pricing.
What is Zolmitriptan's Current Market Position?
Zolmitriptan is a well-established therapeutic agent for the acute treatment of migraine with or without aura. Its mechanism of action involves selective activation of serotonin 5-HT1B and 5-HT1D receptors, leading to cranial vasoconstriction and inhibition of neuropeptide release, thereby alleviating migraine symptoms. As a triptan, it represents a significant class of antimigraine medications, maintaining a consistent demand due to its efficacy and established safety profile.
The market for zolmitriptan is segmented by formulation, with oral tablets, orally disintegrating tablets, and nasal sprays available. The oral tablet remains the most prevalent form due to cost-effectiveness and ease of administration. The orally disintegrating tablet and nasal spray formulations cater to patients experiencing nausea or vomiting, offering faster onset of action.
Key market players include original innovator companies and a substantial number of generic manufacturers. The presence of multiple generic alternatives has intensified price competition, making it a highly competitive market segment.
How Have Patents Impacted Zolmitriptan's Market Access?
The patent landscape for zolmitriptan has significantly shaped its market access and financial performance. The original U.S. patent for zolmitriptan was granted to AstraZeneca, with primary patent protection expiring around the early 2010s. Subsequent patents related to specific formulations (e.g., orally disintegrating tablets) and manufacturing processes provided secondary exclusivity for a period.
Upon patent expiration, the market experienced an influx of generic versions. This transition from brand-name exclusivity to generic availability typically results in:
- Increased Competition: Multiple manufacturers enter the market with bioequivalent products.
- Price Erosion: Generic pricing is substantially lower than the innovator brand, driven by manufacturing efficiencies and competitive pressures.
- Shift in Market Share: The innovator brand's market share declines as healthcare providers and payers opt for more cost-effective generic alternatives.
The ability of generic manufacturers to enter the market is contingent upon successful Abbreviated New Drug Applications (ANDAs) approved by regulatory bodies like the U.S. Food and Drug Administration (FDA). The bioequivalence of generic zolmitriptan to the branded product is a critical factor in its market acceptance and widespread prescription.
What is Zolmitriptan's Competitive Landscape?
Zolmitriptan operates within the broader antimigraine drug market, which includes other triptans, CGRP inhibitors, and older symptomatic treatments. Its primary competitive set consists of other triptans:
- Sumatriptan: The first triptan to market, it has the largest market share in the class and is available in multiple formulations and dosages.
- Rizatriptan: Known for its rapid onset of action and efficacy.
- Eletriptan: Also offers rapid relief and is often considered for moderate to severe migraines.
- Zolmitriptan: Differentiates itself through its oral disintegrating tablet formulation and nasal spray option, offering alternatives for patients with specific needs.
More recently, CGRP (calcitonin gene-related peptide) inhibitors have emerged as a significant competitive force, particularly for migraine prevention and in some cases, acute treatment. These newer agents, such as ubrogepant and rimegepant, offer a different mechanism of action and may be preferred for patients who do not respond to or tolerate triptans.
The competitive positioning of zolmitriptan is largely defined by its cost-effectiveness relative to newer therapies and its established track record for acute migraine management. Generic pricing makes it an attractive option for formulary inclusion and patient out-of-pocket expenses.
What Are the Key Revenue Drivers and Financial Trends for Zolmitriptan?
The financial trajectory of zolmitriptan is characteristic of a mature pharmaceutical product. Post-patent expiration, revenue drivers shift from premium pricing to volume-driven sales of generic products.
Key Revenue Drivers:
- Volume of Prescriptions: The primary driver is the number of prescriptions filled for zolmitriptan products.
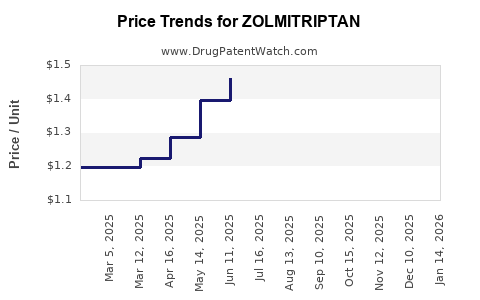
- Generic Pricing: The average selling price per unit of generic zolmitriptan is significantly lower than the brand-name product. Price negotiations between generic manufacturers, wholesalers, and pharmacy benefit managers (PBMs) influence this.
- Market Penetration: The extent to which zolmitriptan is adopted by physicians and patients for acute migraine treatment.
- Formulation Mix: While oral tablets dominate, the availability and uptake of orally disintegrating tablets and nasal sprays contribute to revenue diversification, albeit at a smaller scale.
- Geographic Market Access: Sales performance varies across different countries based on regulatory approvals, market access policies, and healthcare reimbursement systems.
Financial Trends:
- Declining Revenue for Innovator: The original brand manufacturer experienced a significant decline in revenue following patent expiry due to generic competition.
- Growth for Generic Manufacturers: Generic companies have seen revenue generation from zolmitriptan, but this is subject to intense price pressure. Profitability for generic manufacturers is often tied to efficient manufacturing and supply chain management.
- Market Stabilization: While innovator revenue declines, the overall market volume for zolmitriptan remains relatively stable, reflecting its continued utility in migraine treatment.
- Price Volatility: Generic drug prices can be subject to fluctuations due to competition, supply shortages, and changes in payer policies.
Specific financial figures for zolmitriptan as a standalone generic drug are often aggregated within broader therapeutic class reports and are not always publicly disclosed by individual generic manufacturers. However, the overall triptan market, inclusive of zolmitriptan, demonstrates sustained demand for acute migraine treatment.
What Regulatory Factors Influence Zolmitriptan's Market?
Regulatory approval and post-market surveillance are critical for zolmitriptan. Key regulatory factors include:
- FDA Approval: The initial New Drug Application (NDA) for branded zolmitriptan and subsequent ANDAs for generic versions require rigorous review by the FDA to ensure safety, efficacy, and bioequivalence.
- Manufacturing Standards: All manufacturing facilities producing zolmitriptan, whether for branded or generic products, must comply with current Good Manufacturing Practices (cGMP) to ensure product quality and consistency.
- Labeling Requirements: Prescribing information, patient information leaflets, and packaging must adhere to regulatory standards, including indications, contraindications, warnings, and adverse reactions.
- Post-Market Surveillance: Companies are required to report adverse events and potential safety issues to regulatory authorities. This ongoing monitoring can lead to label changes or, in rare cases, market withdrawal.
- International Regulations: Approval and market access in other countries (e.g., EMA in Europe, PMDA in Japan) follow their respective regulatory pathways, which can differ from FDA requirements.
- Generic Competition Pathway: The Hatch-Waxman Act in the U.S. provides a framework for the approval of generic drugs, balancing patent protection with public access to affordable medicines.
What are the Future Market Prospects for Zolmitriptan?
The future market prospects for zolmitriptan are characterized by continued stability in demand but limited growth potential, primarily due to its status as a mature generic product.
Factors Influencing Future Prospects:
- Sustained Migraine Prevalence: The ongoing prevalence of migraine as a neurological disorder ensures a persistent demand for effective acute treatments like zolmitriptan.
- Cost-Effectiveness: As a generic medication, zolmitriptan offers a cost-effective solution, making it a favored choice for payers, healthcare providers, and patients seeking affordable treatment options, especially in the face of rising healthcare costs.
- Competition from Newer Therapies: The increasing development and adoption of CGRP inhibitors and other novel antimigraine drugs represent the most significant competitive challenge. These newer agents may offer differentiated benefits, such as improved efficacy in certain patient populations or a different side effect profile, potentially diverting market share.
- Therapeutic Guidelines: Inclusion and recommendations within clinical practice guidelines for migraine management will continue to influence prescription patterns.
- Generic Market Dynamics: The generic market itself is competitive. Factors such as the number of generic manufacturers, supply chain reliability, and ongoing price negotiations will shape the profitability of zolmitriptan generics.
- Formulation Innovations (Limited): While significant new formulation innovation is unlikely for a product of this age, incremental improvements or niche marketing of existing formulations could maintain specific market segments.
Zolmitriptan is unlikely to experience significant market growth. Its future role will be as a reliable, affordable option within the acute migraine treatment armamentarium, coexisting with newer, potentially more expensive, therapeutic classes. The market share will be largely determined by its ability to maintain a competitive price point and the continued reliance on its established efficacy by a substantial patient population.
Key Takeaways
- Zolmitriptan holds a stable market position as an established acute migraine treatment, primarily accessed through generic formulations.
- Patent expirations have resulted in significant price erosion and intense competition from multiple generic manufacturers.
- The financial trajectory is defined by volume-driven revenue in a mature product lifecycle, with limited growth prospects.
- The competitive landscape includes other triptans and, increasingly, newer CGRP inhibitors, positioning zolmitriptan as a cost-effective alternative.
- Regulatory factors, including FDA approval and cGMP compliance, are critical for market access and product integrity.
- Future market prospects are stable but constrained by generic pricing pressures and competition from novel therapies, with zolmitriptan remaining a foundational cost-effective option.
Frequently Asked Questions
-
What are the primary indications for zolmitriptan?
Zolmitriptan is indicated for the acute treatment of migraine with or without aura in adults.
-
Which formulations of zolmitriptan are available on the market?
Available formulations include oral tablets, orally disintegrating tablets, and nasal sprays.
-
What is the typical pricing trend for zolmitriptan following patent expiry?
Following patent expiry, zolmitriptan prices typically experience substantial declines due to the introduction of generic competition.
-
How does zolmitriptan compare in efficacy to other triptans?
Zolmitriptan is generally considered comparable in efficacy to other triptans, with some variations in onset of action and patient response based on individual characteristics and formulation.
-
Are there any significant safety concerns associated with zolmitriptan use?
Zolmitriptan carries a boxed warning regarding cardiovascular risks, and contraindications include uncontrolled hypertension and ischemic heart disease. It should be used cautiously in patients with risk factors for cardiovascular disease.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases/approved-drug-products-therapeutic-equivalence-evaluations-orange-book
[2] AstraZeneca. (2023). Zomig (zolmitriptan) Prescribing Information. Retrieved from manufacturer's official product information or reputable drug database.
[3] Multiple Generic Pharmaceutical Manufacturers. (Various Dates). Zolmitriptan Product Information and ANDA Filings. Retrieved from various product inserts and regulatory agency databases (e.g., FDA).
[4] Market Research Reports on Migraine Therapeutics. (Various Publishers and Dates). Analysis of the Global Antimigraine Market.