ZOLEDRONIC Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Zoledronic, and what generic alternatives are available?
Zoledronic is a drug marketed by Gland, Accord Hlthcare, Actavis Inc, Amneal, Apotex, Avet Lifesciences, Bpi Labs, Breckenridge, Chartwell Rx, Dr Reddys, Dr Reddys Labs Ltd, Epic Pharma Llc, Eugia Pharma, Fresenius Kabi Usa, Hikma Farmaceutica, Hospira, Hospira Inc, Inforlife, Meitheal, Mylan Labs Ltd, Novast Labs, Sagent Pharms Inc, Shilpa, Sun Pharma Global, and USV. and is included in thirty-nine NDAs.
The generic ingredient in ZOLEDRONIC is zoledronic acid. There are twenty-five drug master file entries for this compound. Twenty-two suppliers are listed for this compound. Additional details are available on the zoledronic acid profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Zoledronic
A generic version of ZOLEDRONIC was approved as zoledronic acid by DR REDDYS LABS LTD on March 4th, 2013.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for ZOLEDRONIC?
- What are the global sales for ZOLEDRONIC?
- What is Average Wholesale Price for ZOLEDRONIC?
Summary for ZOLEDRONIC
| US Patents: | 0 |
| Applicants: | 25 |
| NDAs: | 39 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 91 |
| Clinical Trials: | 301 |
| Patent Applications: | 4,700 |
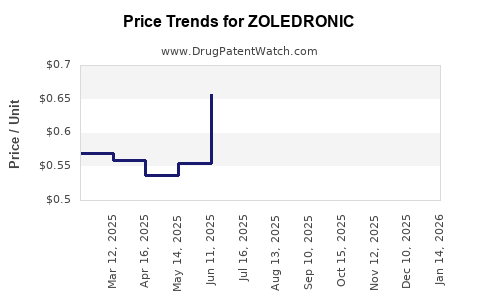
| Drug Prices: | Drug price information for ZOLEDRONIC |
| DailyMed Link: | ZOLEDRONIC at DailyMed |
Recent Clinical Trials for ZOLEDRONIC
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| National Taiwan University Hospital | PHASE4 |
| National Taiwan University Hospital, Yun-Lin Branch | PHASE4 |
| University of Florida | PHASE1 |
Pharmacology for ZOLEDRONIC
| Drug Class | Bisphosphonate |
US Patents and Regulatory Information for ZOLEDRONIC
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mylan Labs Ltd | ZOLEDRONIC ACID | zoledronic acid | INJECTABLE;INTRAVENOUS | 205254-001 | Oct 27, 2017 | AP | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Inforlife | ZOLEDRONIC ACID | zoledronic acid | INJECTABLE;INTRAVENOUS | 202828-001 | Sep 23, 2013 | AP | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Novast Labs | ZOLEDRONIC ACID | zoledronic acid | INJECTABLE;INTRAVENOUS | 208968-001 | Feb 19, 2020 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Meitheal | ZOLEDRONIC ACID | zoledronic acid | INJECTABLE;INTRAVENOUS | 213371-001 | Jun 5, 2023 | AP | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Gland | ZOLEDRONIC | zoledronic acid | INJECTABLE;INTRAVENOUS | 205749-001 | Jun 29, 2018 | AP | RX | No | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Eugia Pharma | ZOLEDRONIC ACID | zoledronic acid | INJECTABLE;INTRAVENOUS | 209125-001 | Dec 8, 2017 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Hikma Farmaceutica | ZOLEDRONIC ACID | zoledronic acid | INJECTABLE;INTRAVENOUS | 202182-001 | Jun 3, 2013 | AP | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for ZOLEDRONIC
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Accord Healthcare S.L.U. | Zoledronic Acid Accord | zoledronic acid | EMEA/H/C/002667Prevention of skeletal related events (pathological fractures, spinal compression, radiation or surgery to bone, or tumour-induced hypercalcaemia) in adult patients with advanced malignancies involving bone.Treatment of adult patients with tumour-induced hypercalcaemia (TIH). | Authorised | yes | no | no | 2014-01-16 | |
| Actavis Group PTC ehf | Zoledronic acid Actavis | zoledronic acid | EMEA/H/C/002488Prevention of skeletal-related events (pathological fractures, spinal compression, radiation or surgery to bone, or tumour-induced hypercalcaemia) in adult patients with advanced malignancies involving bone.Treatment of adult patients with tumour-induced hypercalcaemia. | Authorised | yes | no | no | 2012-04-20 | |
| Mylan Pharmaceuticals Limited | Zoledronic acid Mylan | zoledronic acid | EMEA/H/C/002482Prevention of skeletal related events (pathological fractures, spinal compression, radiation or surgery to bone, or tumour-induced hypercalcaemia) in adult patients with advanced malignancies involving bone;treatment of adult patients with tumour-induced hypercalcaemia (TIH). | Authorised | yes | no | no | 2012-08-23 | |
| Pfizer Europe MA EEIG | Zoledronic Acid Hospira | zoledronic acid | EMEA/H/C/0023654 mg / 5 ml and 4 mg / 100 ml:Prevention of skeletal-related events (pathological fractures, spinal compression, radiation or surgery to bone, or tumour-induced hypercalcaemia) in adult patients with advanced malignancies involving bone.Treatment of adult patients with tumour-induced hypercalcaemia (TIH).5 mg / 100 ml:Treatment of osteoporosis:in post-menopausal women;in men;at increased risk of fracture, including those with a recent low-trauma hip fracture.Treatment of osteoporosis associated with long-term systemic glucocorticoid therapy:in post-menopausal women;in men;at increased risk of fracture.Treatment of Paget's disease of the bone in adults. | Authorised | yes | no | no | 2012-11-19 | |
| Teva B.V. | Zoledronic acid Teva | zoledronic acid | EMEA/H/C/002439Prevention of skeletal-related events and treatment of tumour-induced hypercalcaemia. | Authorised | yes | no | no | 2012-08-16 | |
| Sandoz Pharmaceuticals d.d. | Aclasta | zoledronic acid | EMEA/H/C/000595Treatment of osteoporosis:, , , in post-menopausal women;, in men;, , , at increased risk of fracture, including those with a recent low-trauma hip fracture., , Treatment of osteoporosis associated with long-term systemic glucocorticoid therapy in post-menopausal women and in men at increased risk of fracture., , Treatment of Paget's disease of the bone., | Authorised | no | no | no | 2005-04-15 | |
| Phoenix Labs Unlimited Company | Zometa | zoledronic acid | EMEA/H/C/000336Prevention of skeletal related events (pathological fractures, spinal compression, radiation or surgery to bone, or tumour-induced hypercalcaemia) in patients with advanced malignancies involving bone;treatment of tumour-induced hypercalcaemia (TIH);prevention of skeletal related events (pathological fractures, spinal compression, radiation or surgery to bone, or tumour-induced hypercalcaemia) in patients with advanced malignancies involving bone;treatment of tumour-induced hypercalcaemia (TIH);prevention of skeletal related events (pathological fractures, spinal compression, radiation or surgery to bone, or tumour-induced hypercalcaemia) in adult patients with advanced malignancies involving bone;treatment of adult patients with tumour-induced hypercalcaemia (TIH). | Authorised | no | no | no | 2001-03-20 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
Market Dynamics and Financial Trajectory for ZOLEDRONIC
More… ↓
