Last updated: February 19, 2026
Executive Summary
XYZAL (levocetirizine dihydrochloride) is a second-generation antihistamine approved for the treatment of allergic rhinitis and chronic idiopathic urticaria. Developed by UCB Pharma and marketed by Sanofi, its market performance is characterized by a mature product lifecycle, significant generic competition, and ongoing patent challenges impacting its revenue streams. While XYZAL historically commanded substantial sales, its financial trajectory has shifted due to patent expirations and the subsequent market entry of generic alternatives, necessitating strategic adjustments by its marketers.
What is XYZAL and its Therapeutic Indication?
XYZAL is an orally administered active metabolite of cetirizine. It functions as a selective histamine H1 receptor antagonist, inhibiting the release of histamine, a key mediator in allergic reactions. This action alleviates symptoms such as sneezing, runny nose, itchy nose, itchy throat, itchy eyes, and watery eyes associated with allergic rhinitis. It also provides relief from itching and hives in chronic idiopathic urticaria.
The drug is available in multiple formulations, including tablets, oral solution, and orally disintegrating tablets, catering to different patient needs and age groups. The recommended dosage typically ranges from 2.5 mg to 5 mg once daily.
What is the Patent Landscape for XYZAL?
The patent landscape surrounding XYZAL has been a critical factor in its market exclusivity and subsequent generic erosion.
- Primary Patents: UCB Pharma held key patents covering the composition of matter for levocetirizine dihydrochloride. For example, U.S. Patent No. 6,348,470, filed in 1998 and issued in 2002, protected the active pharmaceutical ingredient. This patent was central to establishing market exclusivity for the branded product.
- Method of Use Patents: Additional patents covered specific methods of use and formulations. These patents, while potentially extending the protection period for certain applications, often face legal challenges once the primary composition of matter patents expire.
- Patent Expirations and Litigation: The expiration of core patents for XYZAL, particularly in the United States, opened the door for generic manufacturers. This led to extensive patent litigation, as brand manufacturers typically seek to defend their market share by challenging the validity of existing patents or asserting secondary patents. For instance, litigation surrounding the '470 patent and other related intellectual property rights has been a recurring theme.
- Generic Entry: Following patent expirations and the resolution of legal challenges, multiple generic versions of levocetirizine dihydrochloride have entered the market. This has significantly increased competition and driven down prices. The U.S. Food and Drug Administration (FDA) has approved numerous Abbreviated New Drug Applications (ANDAs) for generic levocetirizine.
The timeline of key patent expirations and subsequent generic entry has directly shaped the financial performance of XYZAL. The loss of market exclusivity for the branded product has led to a substantial decline in its revenue.
What are the Market Dynamics for XYZAL?
The market dynamics for XYZAL are characteristic of a branded pharmaceutical product transitioning into a mature, genericized market.
- Declining Market Share: Following the introduction of generic levocetirizine, the market share of branded XYZAL has significantly decreased. Prescribing patterns have shifted towards more cost-effective generic alternatives.
- Price Erosion: The presence of multiple generic competitors has led to substantial price erosion. Generic levocetirizine is available at a fraction of the price of branded XYZAL, making it the preferred option for many payers and patients.
- Competition Landscape: The competitive landscape is highly fragmented with numerous generic manufacturers offering levocetirizine dihydrochloride. Major generic players, including Teva Pharmaceuticals, Mylan (now Viatris), and Sandoz, are active in this market.
- Physician and Patient Prescribing Behavior: While some physicians may continue to prescribe branded XYZAL due to familiarity or specific patient responses, the majority of prescriptions are now for generic levocetirizine. Patient demand is increasingly driven by formulary placement and out-of-pocket costs.
- Regulatory Environment: Regulatory bodies like the FDA play a crucial role in approving generic applications, facilitating market entry and competition. The approval of ANDAs for levocetirizine has been a primary driver of market dynamics.
- Marketing and Sales Strategies: For the branded product, marketing efforts have shifted from broad awareness campaigns to targeted strategies focusing on physician education regarding specific patient populations or formulation advantages. For generic manufacturers, competition is primarily based on price and distribution.
- Therapeutic Class Performance: The overall market for antihistamines is large and competitive, with many established first and second-generation options, as well as newer classes of allergy treatments. XYZAL competes within this broader therapeutic category.
What is the Financial Trajectory of XYZAL?
The financial trajectory of XYZAL reflects a classic pattern of revenue growth during its exclusivity period followed by a sharp decline post-patent expiration and generic entry.
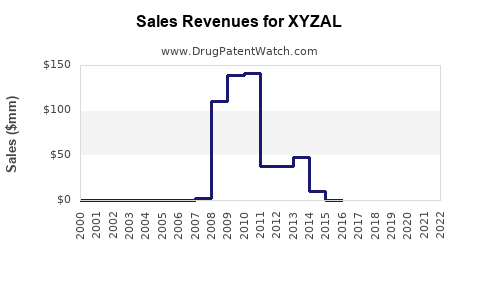
- Peak Sales: During its period of market exclusivity, XYZAL generated significant revenue. In its peak years, annual sales for the branded product reached hundreds of millions of dollars globally. For example, in the mid-to-late 2010s, Sanofi reported substantial annual revenues attributed to XYZAL.
- Impact of Generic Competition: The introduction of generic levocetirizine in 2017 in the United States marked a turning point. This event triggered a rapid and dramatic decline in the sales of branded XYZAL. Quarterly and annual reports from Sanofi and UCB Pharma show a steep drop in revenues attributed to the drug following this period.
- Revenue Contribution Shift: The revenue contribution from branded XYZAL has diminished to a small percentage of its former peak. While still generating some sales from brand loyalists or specific market segments, its overall financial significance has been overtaken by generic versions.
- Global Market Variations: The timing and impact of generic entry have varied across different global markets, influenced by local patent laws, regulatory approval timelines, and pricing strategies. Some regions may have experienced generic competition earlier or later than others, leading to divergent financial trajectories.
- Licensing and Royalty Agreements: UCB Pharma, as the original developer, has historically benefited from licensing and royalty agreements with Sanofi and other partners. The financial terms of these agreements would have evolved significantly with the onset of generic competition, with royalty payments from branded sales decreasing and potential upfront payments or milestone achievements from generic licensees becoming less relevant.
- Market Research and Sales Data: Pharmaceutical market research firms, such as IQVIA and Symphony Health Solutions, track prescription volumes and sales data. Their reports consistently indicate a dominance of generic levocetirizine in terms of prescription numbers and overall market value, with branded XYZAL representing a minor segment.
- Future Projections: For branded XYZAL, future revenue projections are expected to remain low. The focus for its marketers is now on managing the remaining brand presence rather than significant growth. The overall market for levocetirizine, however, remains robust due to the continued demand for antihistamines and the availability of affordable generic options.
Key Takeaways
- XYZAL's market performance is defined by a transition from peak sales during patent exclusivity to significant revenue decline following generic entry.
- The patent landscape, particularly the expiration of core composition of matter patents, was the primary driver for the introduction of generic levocetirizine.
- Branded XYZAL sales have been substantially eroded by price competition from multiple generic manufacturers.
- The financial trajectory shows a sharp revenue decrease post-2017 in major markets, with branded product sales now representing a small fraction of historical peaks.
Frequently Asked Questions
-
When did generic XYZAL become available in the United States?
Generic XYZAL (levocetirizine dihydrochloride) became available in the United States in 2017 following the expiration of key patents.
-
Who are the primary manufacturers of generic levocetirizine?
Major generic manufacturers include Teva Pharmaceuticals, Mylan (now Viatris), and Sandoz, among others.
-
What is the typical price difference between branded XYZAL and its generic equivalent?
Generic levocetirizine is typically priced at 20% to 80% less than branded XYZAL, depending on the pharmacy, dosage, and insurance coverage.
-
Does XYZAL have any advantages over other antihistamines?
XYZAL is a second-generation antihistamine, meaning it is less likely to cause drowsiness compared to older, first-generation antihistamines. Its efficacy in treating allergic rhinitis and urticaria is comparable to other second-generation agents.
-
What is the current market share of branded XYZAL compared to generic levocetirizine?
Branded XYZAL holds a very small market share, with generic levocetirizine dominating prescription volumes and overall market value.
Citations
[1] U.S. Patent No. 6,348,470. (2002). Preparation of enantiomerically pure (R)-cetirizine. United States Patent and Trademark Office.
[2] Sanofi Annual Reports. (Various Years). Financial Statements and SEC Filings. Retrieved from Sanofi Investor Relations.
[3] UCB Pharma Annual Reports. (Various Years). Financial Statements and Company Performance. Retrieved from UCB Investor Relations.
[4] U.S. Food and Drug Administration. (Ongoing). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from FDA Website.
[5] IQVIA Market Data. (Proprietary Data accessed for market analysis). Prescription and Sales Data for Antihistamines.
[6] Symphony Health Solutions Data. (Proprietary Data accessed for market analysis). Pharmaceutical Market Insights.