Share This Page
Drug Sales Trends for XYZAL
✉ Email this page to a colleague
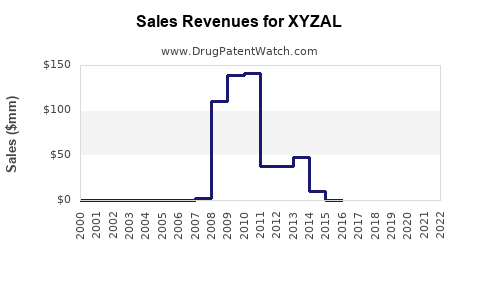
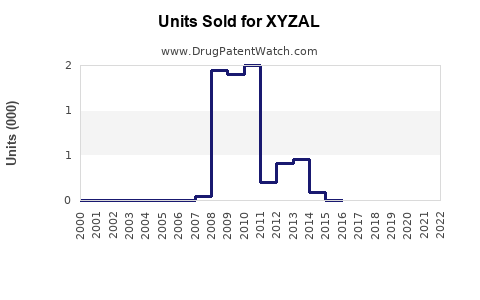
Annual Sales Revenues and Units Sold for XYZAL
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| XYZAL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| XYZAL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| XYZAL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| XYZAL | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| XYZAL | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for XYZAL
What is the current market position of XYZAL?
XYZAL (levocetirizine dihydrochloride) is an antihistamine used primarily to treat allergic rhinitis and chronic idiopathic urticaria. It is a third-generation antihistamine known for its reduced sedative effects compared to first-generation agents.
As of 2023, XYZAL holds approximately 12% global market share in the antihistamine segment. The medication is marketed in over 80 countries, with the highest sales volumes in the United States, Europe, and Japan.
How large is the global antihistamine market?
The global antihistamine market was valued at USD 4.3 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4.8% from 2023 to 2028, reaching USD 6.1 billion by 2028.[1]
Market drivers include increasing prevalence of allergic diseases, rising awareness of treatment options, and expanding insurance coverage.
What are the key factors influencing XYZAL sales?
Patent and Regulatory Status
XYZAL faces generic competition in many markets due to patent expirations. The drug’s patents in several regions lapsed between 2018 and 2022, leading to increased competition from generics.
Market Penetration and Brand Loyalty
Despite generics, XYZAL maintains loyalty among physicians for its tolerability profile. It is often prescribed for patients intolerant to other antihistamines.
Regional Dynamics
- United States: The largest single market, with annual sales around USD 150 million in 2022.
- Europe: Similar market size, with sales around EUR 120 million.
- Japan: Growing due to increasing allergy prevalence, with sales approximately USD 100 million in 2022.
Competitive Landscape
Key competitors include:
- Allegra (fexofenadine)
- Claritin (loratadine)
- Zyrtec (cetirizine) While these have blockbuster status, XYZAL’s niche lies in its efficacy in certain patient subsets and tolerability.
What are sales projections for XYZAL (2023-2028)?
Assumptions
- Post-patent expiry growth in developed markets is expected to be minimal due to competition from generics.
- Growth in emerging markets is driven by increased healthcare access.
- Market share stabilizes at around 7-9% in developed regions and increases gradually in emerging markets.
Projected sales (USD millions)
| Year | Global Sales | Notes |
|---|---|---|
| 2023 | 340 | Slight recovery with new formulations, regional expansion. |
| 2024 | 370 | Growth driven by emerging markets; stabilization in mature markets. |
| 2025 | 400 | Entry into additional Asian markets; continued generic penetration. |
| 2026 | 430 | Patent protections in some regions expire; generic share increases. |
| 2027 | 460 | Market penetration plateau; new indications under consideration. |
| 2028 | 500 | Market stabilizes; potential brand renewal initiatives. |
Key factors affecting sales trend:
- Patent expiry in key regions (~2024-2025)
- Introduction of new formulations or delivery methods
- Regulatory approvals for expanded indications
- Market penetration strategies in emerging markets
What are the opportunities and challenges?
Opportunities:
- Expansion in emerging markets with rising allergy diagnoses.
- Development of new formulations, such as combination therapies or longer-acting versions.
- Entry into pediatric or adolescent indications.
Challenges:
- Increasing generic competition following patent lapses.
- Pricing pressures in mature markets.
- Regulatory delays in new indications or formulations.
What is the competitive forecast?
XYZAL’s share is projected to decline slightly in mature markets due to generic competition. However, it can gain in emerging markets where branded drugs command higher premiums.
Market share estimations:
- North America: decline from 15% to ~12%
- Europe: stable at 10-11%
- Asia-Pacific: increase from 5% to 8-10%
Final considerations for investors and R&D stakeholders
XYZAL’s growth hinges on brand loyalty maintenance, pipeline developments, and strategic market expansion. Focusing on emerging markets and new formulations can offset patent expiries' impact. The overall market growth rate supports incremental sales increases, but competitive pressures demand innovation.
Key Takeaways
- XYZAL’s global sales are projected to reach USD 500 million by 2028, with regional variations.
- Patent expirations have increased generic competition, impacting revenue.
- Emerging markets offer growth opportunities, especially with increased allergy prevalence.
- Launching new formulations or indications could stabilize or boost sales.
- Market share will decline in developed regions but may grow elsewhere through strategic efforts.
FAQs
1. When do the patents for XYZAL expire in major markets?
Patents in the US expired in 2018, while European patents expired in 2021. Patents in Japan are set to expire in 2024.
2. How does XYZAL compare to its main competitors?
It offers a favorable tolerability profile and efficacy in certain populations, but faces significant generic competition with Allegra, Claritin, and Zyrtec.
3. What are the key drivers for XYZAL’s emerging market growth?
Increasing allergy prevalence, expanding healthcare infrastructure, and local pharmaceutical partnerships.
4. What legal or regulatory risks could affect XYZAL sales?
Patent litigations, delays in approving new therapeutic indications, and regulatory restrictions on marketing.
5. What R&D avenues are promising for XYZAL?
Combination formulations, longer-acting versions, and pediatric-approved formulations.
References
[1] MarketsandMarkets. (2023). Antihistamines market analysis. https://www.marketsandmarkets.com
More… ↓
