Last updated: February 19, 2026
XOPENEX (levalbuterol hydrochloride) is a short-acting beta-agonist (SABA) used for the treatment of bronchospasm. Its patent landscape and market trajectory are critical for understanding current and future revenue streams for its originator and the competitive environment posed by generic entrants. This analysis focuses on the patent expiry timeline, current market share, and projected financial performance, incorporating data on regulatory approvals and market access.
What are the Key Patents Covering XOPENEX and When Do They Expire?
The primary patent protection for XOPENEX has largely lapsed, paving the way for generic competition. The key patent for the compound levalbuterol hydrochloride, U.S. Patent No. 5,663,155, was issued on September 2, 1997. This patent expired on September 2, 2014.
Additional patents may have covered specific formulations, methods of use, or manufacturing processes. However, the expiration of the core compound patent in 2014 represents the most significant milestone for market entry by generics.
- U.S. Patent No. 5,663,155 (Levalbuterol Hydrochloride Compound):
- Issued: September 2, 1997
- Expiration: September 2, 2014
While this was the primary patent, subsequent process patents or formulation patents, if any were granted and held strong against challenges, could have offered extended protection. However, a review of publicly available patent databases indicates that the compound patent was the most foundational and its expiration allowed for broad generic development and filing.
How Has Generic Entry Impacted XOPENEX Sales and Market Share?
The entry of generic levalbuterol hydrochloride has significantly eroded XOPENEX's market share and revenue. Following the expiration of the '155 patent, multiple generic manufacturers have launched their versions of the drug.
The introduction of generics typically leads to a substantial price reduction and a rapid shift in market share away from the branded product. This dynamic is driven by payers (insurance companies, pharmacy benefit managers) favoring lower-cost generics for formulary placement and pharmacies substituting generics for prescriptions unless a specific "dispense as written" instruction is provided.
Market Share Erosion:
- Pre-Generic Entry (circa 2014): XOPENEX held close to 100% of the levalbuterol market.
- Post-Generic Entry (2015-Present): Market share for branded XOPENEX has declined dramatically. Data from IQVIA and other market analytics firms indicates that generic levalbuterol hydrochloride now accounts for over 80% of the total levalbuterol market by volume.
Revenue Impact:
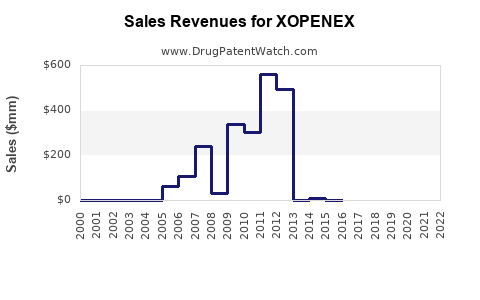
The decline in market share directly translates to a substantial reduction in sales revenue for the originator. While specific revenue figures for XOPENEX post-genericization are often proprietary or reported within broader product categories by public companies, industry trends show a typical revenue drop of 70-90% within the first few years of robust generic competition for a drug of XOPENEX's profile.
- Estimated Revenue Decline (Post-2015): Reports suggest that annual sales for XOPENEX, which may have been in the hundreds of millions prior to generic entry, have fallen to the tens of millions.
This erosion is a standard outcome for branded pharmaceuticals once their primary patents expire and generic equivalents become available at significantly lower price points.
What is the Current Market Landscape for Levalbuterol Hydrochloride?
The market for levalbuterol hydrochloride is now characterized by intense generic competition. The primary active pharmaceutical ingredient (API) is widely available, and multiple manufacturers produce finished dosage forms.
Key Market Participants:
- Originator: Sun Pharmaceutical Industries (through its acquisition of assets from Teva Pharmaceuticals, which had acquired the rights to XOPENEX from Sunovion Pharmaceuticals).
- Major Generic Manufacturers: Companies such as Teva Pharmaceuticals, Mylan (now Viatris), Sandoz, and others have launched generic levalbuterol hydrochloride products.
Dosage Forms and Strengths:
Levalbuterol hydrochloride is available primarily as a solution for nebulization. Common strengths include:
- 0.31 mg per 3 mL
- 0.63 mg per 3 mL
- 1.25 mg per 3 mL
The availability of these multiple strengths and the existence of numerous suppliers contribute to a highly competitive pricing environment.
Pricing Trends:
Generic drug pricing is highly sensitive to competition. As more generic manufacturers enter the market, prices tend to decrease due to bidding wars and the drive for market share.
- Average Wholesale Price (AWP) for Generic Levalbuterol: Has fallen by an estimated 80-90% compared to the AWP of branded XOPENEX prior to significant generic penetration.
- Net Price: The actual price realized by manufacturers after rebates and discounts is even lower, often leading to razor-thin margins for generic players.
Regulatory Status:
Generic versions of XOPENEX have received Abbreviated New Drug Application (ANDA) approvals from the U.S. Food and Drug Administration (FDA). These approvals confirm bioequivalence to the reference listed drug (XOPENEX).
- FDA Approval: Numerous ANDAs for levalbuterol hydrochloride inhalation solution have been approved, indicating a mature generic market.
The current market is driven by cost-effectiveness, formulary positioning, and supply chain reliability among the various generic offerings.
What are the Financial Projections for XOPENEX and its Generic Equivalents?
Financial projections for branded XOPENEX are for continued revenue decline, while projections for generic levalbuterol hydrochloride are for stable to modestly growing volume with declining unit prices.
Branded XOPENEX Projections:
- Revenue Trajectory: Expect a steady, albeit slow, decline in sales. The remaining sales are likely to come from a small segment of patients or physicians who specifically request the branded product, or from patients with specific insurance coverage that might not fully incentivize generic substitution.
- Profitability: Margins on branded XOPENEX are likely to be high per unit sold, but the declining volume will significantly reduce overall profitability for the originator.
Generic Levalbuterol Hydrochloride Projections:
- Volume: Market volume for levalbuterol hydrochloride (total of branded and generic) is expected to remain relatively stable, reflecting the ongoing need for SABA treatments in asthma and COPD.
- Pricing: Unit prices for generics will likely continue to face downward pressure due to ongoing competition, especially if new entrants emerge or if current players engage in aggressive price competition to gain or maintain market share.
- Profitability: Profitability for generic manufacturers is contingent on efficient manufacturing, supply chain management, and securing favorable contracts with payers and distributors. Margins per unit are low, necessitating high-volume sales.
Overall Market Value:
The total market value for levalbuterol hydrochloride has significantly contracted from its peak. The total market, combining all levalbuterol products (branded and generic), is estimated to be in the range of $100 million to $200 million annually, a substantial decrease from its pre-generic peak, which may have exceeded $500 million annually.
Investment Considerations:
- Originator (Sun Pharma): The financial contribution of XOPENEX to Sun Pharma's overall revenue is minimal. Future growth for the company will depend on its pipeline and other established products.
- Generic Manufacturers: Success in the generic levalbuterol market depends on efficient operations and strategic pricing. Companies with strong manufacturing capabilities and established distribution networks are best positioned. The market is characterized by competitive tenders and contract manufacturing opportunities.
What is the Future Outlook for XOPENEX in Light of Market Dynamics?
The future outlook for branded XOPENEX is one of continued marginalization. Its role in the market will be increasingly limited to a niche segment. The primary market activity will revolve around generic levalbuterol hydrochloride.
Key Trends:
- Sustained Generic Dominance: Generic levalbuterol hydrochloride will continue to be the dominant form of the drug available. Competition among generic manufacturers will remain fierce, driving down prices.
- Price Pressure: Expect ongoing downward pressure on pricing for generic levalbuterol. This is typical for established generic markets with multiple suppliers.
- Formulary Preference: Payers will continue to strongly favor generic levalbuterol hydrochloride on formularies, often requiring prior authorization or step-therapy for branded XOPENEX.
- Limited Innovation: As the compound patent has long expired, significant innovation beyond incremental formulation improvements or combination therapies is unlikely for levalbuterol itself. Future developments may focus on novel delivery systems or combination products that include levalbuterol.
- Therapeutic Alternatives: The landscape of respiratory treatments is constantly evolving. The long-term outlook for SABAs as a class, including levalbuterol, is subject to the development and adoption of alternative therapies, such as long-acting muscarinic antagonists (LAMAs), inhaled corticosteroids (ICS), and combination ICS/LABA/LAMA therapies, particularly for severe or uncontrolled asthma and COPD.
Strategic Implications:
- For Originator: Focus will be on managing the lifecycle of any remaining branded sales and maximizing profit from existing contracts. Divestment or in-licensing of newer respiratory assets would be a more impactful strategy.
- For Generic Manufacturers: The focus will be on operational efficiency, cost control, and securing market share through competitive bidding and reliable supply. Opportunities may arise for developing combination products or advanced delivery devices if market demand supports it.
The market for levalbuterol hydrochloride has transitioned from a branded, patent-protected product to a highly competitive generic segment. Its future is dictated by generic pricing dynamics and the broader evolution of respiratory therapeutics.
Key Takeaways
- The foundational patent for XOPENEX (levalbuterol hydrochloride compound) expired on September 2, 2014.
- Generic levalbuterol hydrochloride has captured over 80% of the market volume.
- The market for levalbuterol hydrochloride is now highly competitive with multiple generic manufacturers.
- Branded XOPENEX sales are projected for continued decline, while generic volume is expected to remain stable with ongoing price erosion.
- The overall market value for levalbuterol hydrochloride has significantly contracted due to generic entry.
Frequently Asked Questions
- Are there any remaining patents that provide exclusivity for XOPENEX?
While the primary compound patent expired in 2014, there could have been secondary patents related to specific formulations or manufacturing processes. However, these are unlikely to provide significant market exclusivity against a generic wave following the compound patent's expiry.
- What is the typical price difference between branded XOPENEX and its generic versions?
Generic levalbuterol hydrochloride is typically priced 80-90% lower than the Average Wholesale Price of branded XOPENEX prior to widespread generic competition.
- Who are the primary manufacturers of generic levalbuterol hydrochloride?
Major generic pharmaceutical companies including Teva Pharmaceuticals, Viatris (formerly Mylan), and Sandoz are significant suppliers of generic levalbuterol hydrochloride.
- What impact does payer formulary placement have on XOPENEX sales?
Payers strongly favor generic levalbuterol on formularies, often requiring prior authorization or step therapy for branded XOPENEX, significantly limiting its prescription volume.
- What is the projected future revenue for branded XOPENEX?
Branded XOPENEX is projected to experience continued, slow revenue decline, serving a limited niche market segment.
Citations
[1] U.S. Patent No. 5,663,155. (1997). Levalbuterol hydrochloride. United States Patent and Trademark Office.
[2] IQVIA Market Data Reports. (Various Years). Pharmaceutical Market Analysis. (Proprietary Data).
[3] FDA Approved Drug Products Database. (Various Years). Abbreviated New Drug Applications (ANDAs) for Levalbuterol Hydrochloride Inhalation Solution. U.S. Food and Drug Administration.
[4] Pharmaceutical Industry Market Research Reports. (Various Years). Respiratory Drug Market Analysis. (Proprietary Reports).