Last updated: February 19, 2026
Vyvanse (lisdexamfetamine dimesylate), a prodrug of dextroamphetamine, is approved for treating Attention Deficit Hyperactivity Disorder (ADHD) in children and adults, and binge eating disorder (BED) in adults. Its patent exclusivity has been a significant driver of revenue for Takeda Pharmaceutical Company. However, the compound patent expired in August 2023, paving the way for generic competition and impacting future market dynamics and financial trajectory.
What are the approved indications and target populations for Vyvanse?
Vyvanse is approved for the following indications:
- Attention Deficit Hyperactivity Disorder (ADHD):
- Children aged 6 to 12 years: Approved for the treatment of ADHD [1].
- Adolescents aged 13 to 17 years: Approved for the treatment of ADHD [1].
- Adults aged 18 years and older: Approved for the treatment of ADHD [1].
- Binge Eating Disorder (BED):
- Adults aged 18 years and older: Approved for the treatment of moderate to severe BED [2].
The target population encompasses millions of individuals diagnosed with these conditions globally. The prevalence of ADHD in children is estimated to be between 5% and 13% [3], and in adults, it is between 2.5% and 4.4% [4]. BED affects approximately 2.0% of adults in the United States [5].
What is the patent exclusivity status for Vyvanse?
Vyvanse has benefited from robust patent protection that has largely shielded it from generic competition. The key patent for the compound lisdexamfetamine dimesylate expired in August 2023 [6].
- Compound Patent Expiration: U.S. Patent No. 6,436,948, covering lisdexamfetamine dimesylate, expired on August 23, 2023 [6].
- Evergreening Attempts: Takeda pursued various strategies to extend exclusivity, including new formulation patents and method-of-use patents for different indications. For instance, patents related to extended-release formulations and treatment of BED were also in play [7].
- Litigation: Takeda engaged in extensive litigation to defend its patents against generic manufacturers. Several generic companies filed Paragraph IV certifications, challenging the validity or non-infringement of Takeda's patents [8]. Court rulings and settlement agreements have influenced the timeline of generic entry.
What has been the historical financial performance of Vyvanse?
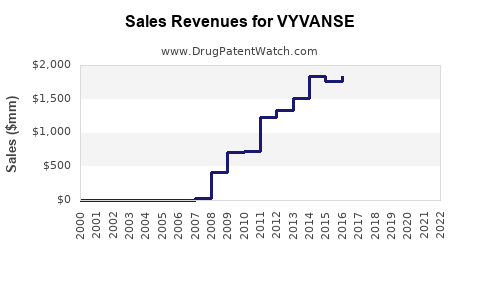
Vyvanse has been a significant revenue generator for Takeda Pharmaceutical Company. Its financial performance reflects strong market adoption and the benefits of patent exclusivity.
Revenue Performance (USD Billions):
| Fiscal Year |
Net Sales (Vyvanse) |
Year-over-Year Growth |
Takeda Total Revenue |
Vyvanse % of Takeda Revenue |
| 2020 |
2.58 |
12.9% |
3.08 (Trillions JPY) |
~10% |
| 2021 |
3.05 |
18.2% |
3.38 (Trillions JPY) |
~11% |
| 2022 |
3.56 |
16.7% |
3.76 (Trillions JPY) |
~11.7% |
| 2023 (9M) |
2.70 |
N/A |
2.95 (Trillions JPY) |
~12% |
Note: Takeda's fiscal year ends March 31st. Revenue figures for 2020-2022 are for the fiscal year ending March 31st of that year. 2023 (9M) refers to the first nine months of the fiscal year ending March 31, 2024. Currency conversion rates apply. Vyvanse revenue is typically reported in USD by Takeda for global markets.
Key Financial Observations:
- Consistent Growth: Vyvanse demonstrated consistent double-digit year-over-year revenue growth in the years leading up to patent expiration.
- Anchor Product: It became a flagship product for Takeda's neuroscience franchise and a substantial contributor to the company's overall revenue.
- Market Penetration: Strong sales indicate significant market penetration within both ADHD and BED patient populations, driven by efficacy and perceived advantages over older stimulant medications.
What is the competitive landscape for Vyvanse?
The competitive landscape for Vyvanse includes both branded and generic stimulants for ADHD, and emerging treatments for BED.
ADHD Market:
- Branded Stimulants:
- Adderall XR (mixed amphetamine salts extended-release): Shire/Takeda. Remains a significant competitor, though also faces generic competition.
- Concerta (methylphenidate extended-release): Johnson & Johnson. A major methylphenidate-based option.
- Ritalin LA (methylphenidate hydrochloride extended-release): Novartis.
- Daytrana (methylphenidate transdermal system): Noven Therapeutics.
- Generic Stimulants: A vast array of generic immediate-release and extended-release formulations of amphetamines and methylphenidate are available.
- Non-Stimulants:
- Strattera (atomoxetine HCl): Eli Lilly. A selective norepinephrine reuptake inhibitor.
- Kapvay (clonidine hydrochloride extended-release): Supernus Pharmaceuticals.
- Intuniv (guanfacine extended-release): Shire/Takeda.
- Newer Non-Stimulants:
- Qelbree (viloxazine extended-release): Supernus Pharmaceuticals. A selective norepinephrine reuptake inhibitor.
Binge Eating Disorder (BED) Market:
- Primary Treatment: Vyvanse is currently the only FDA-approved medication specifically for BED.
- Off-Label Use: Antidepressants (e.g., SSRIs like fluoxetine, sertraline) and other psychotherapies are used off-label.
- Emerging Therapies: Clinical trials are exploring new pharmacological targets for BED, which could introduce new branded or generic competition in the future.
What are the implications of patent expiration on Vyvanse's market exclusivity and pricing?
The expiration of the primary compound patent for Vyvanse in August 2023 marks a pivotal shift, leading to the introduction of generic versions and subsequent price erosion.
- Generic Entry: Following the patent expiration, multiple generic manufacturers have launched lisdexamfetamine dimesylate capsules in various strengths.
- Price Reduction: Generic competition is expected to drive significant price reductions, typically ranging from 50% to 85% or more compared to the branded product over time, depending on market penetration and payer dynamics.
- Market Share Shift: Branded Vyvanse will experience a substantial loss of market share as payers and patients opt for lower-cost generic alternatives.
- Revenue Decline: Takeda's revenue from Vyvanse is projected to decline sharply following generic entry. The company has factored this into its financial guidance.
- Payer Influence: Pharmacy benefit managers (PBMs) and insurance companies will likely favor generic lisdexamfetamine on their formularies, further accelerating the shift away from branded Vyvanse.
What is the projected financial trajectory for Vyvanse post-patent expiration?
The financial trajectory for Vyvanse will be characterized by a precipitous decline in sales due to generic competition.
- Immediate Sales Impact: Significant revenue reduction is expected starting in the latter half of 2023 and throughout 2024.
- Takeda's Revenue Guidance: Takeda has acknowledged the impact of Vyvanse patent expiration in its financial forecasts. For the fiscal year ending March 31, 2024, the company projected a decline in Vyvanse sales due to generic entry [9].
- Long-Term Outlook: While overall sales will decrease, residual sales may persist from patients who remain on branded Vyvanse due to prescriber preference, specific payer contracts, or patient loyalty. However, these will constitute a minor portion of historical sales.
- Impact on Takeda's Portfolio: Takeda is focusing on other growth drivers within its portfolio, such as Enhertu (trastuzumab deruxtecan) and TAK-007, to offset the revenue loss from Vyvanse and other upcoming patent expiries [9].
- Generic Market Dynamics: The generic lisdexamfetamine market will likely see intense competition among multiple manufacturers, leading to further price stabilization at lower levels.
What strategies can pharmaceutical companies employ in response to Vyvanse's patent expiration?
Pharmaceutical companies, particularly Takeda, can implement several strategies to mitigate the impact of Vyvanse's patent expiration and capitalize on evolving market conditions.
- Focus on Lifecycle Management:
- Advocacy for Continued Use: Educate healthcare providers and patients on the continued availability and potential benefits of branded Vyvanse where clinically appropriate or preferred by specific patient segments.
- New Formulations/Delivery Systems (Limited Post-Expiry): While less impactful post-compound patent expiry, exploration of novel delivery systems or combination therapies involving lisdexamfetamine could be considered if technically feasible and patentable, though this is a high-risk, long-term strategy.
- Portfolio Diversification and Growth:
- Investment in Pipeline: Accelerate R&D for novel treatments in neuroscience or other therapeutic areas to replace lost revenue. Takeda's focus on areas like oncology and rare diseases is a key element of this strategy [9].
- Maximizing Other Branded Products: Aggressively market and support other profitable products within the company's portfolio to fill the revenue gap.
- Strategic Acquisitions: Pursue targeted acquisitions of companies with promising late-stage pipelines or commercial-stage assets that align with strategic therapeutic areas.
- Generic Market Engagement (If Applicable):
- Authorized Generics: In some cases, the originator company might launch an "authorized generic" through a subsidiary or partner to capture a share of the generic market and manage the transition.
- Licensing Agreements: Explore licensing opportunities for lisdexamfetamine in ex-U.S. markets where patent expiry may differ, or for specific territories or indications.
- Operational Efficiency and Cost Management:
- Streamlining Operations: Implement cost-saving measures across R&D, manufacturing, and commercial operations to improve profitability amidst declining revenue.
- Supply Chain Optimization: Ensure robust and cost-effective supply chains for both existing products and any new pipeline assets.
- Market Access and Payer Strategies:
- Negotiate Payer Contracts: While generic competition is the primary driver, engage with payers to understand formulary placement and access for any remaining branded Vyvanse, or for new pipeline products.
- Value-Based Demonstrations: For new pipeline assets, develop strong health economic arguments to justify pricing and market access.
For Generic Manufacturers:
- Rapid Market Entry: Expedite the launch of generic lisdexamfetamine upon patent expiry to capture first-mover advantage.
- Aggressive Pricing: Implement competitive pricing strategies to gain market share rapidly.
- Supply Chain Reliability: Ensure consistent and reliable supply to meet market demand.
- Broad Distribution: Establish strong relationships with wholesalers, distributors, and retail pharmacies.
Key Takeaways
- Vyvanse (lisdexamfetamine dimesylate) has been a highly successful branded pharmaceutical, generating billions in revenue for Takeda, primarily for ADHD and BED.
- The primary compound patent for Vyvanse expired in August 2023, leading to the immediate availability of generic lisdexamfetamine dimesylate.
- Generic entry is projected to cause a sharp and significant decline in Vyvanse's revenue.
- The competitive landscape for ADHD treatment is mature, with numerous generic stimulant options. Vyvanse remains the only FDA-approved medication for BED.
- Takeda's financial strategy post-Vyvanse patent expiration involves relying on other growth drivers, such as Enhertu, and investing in its pipeline.
Frequently Asked Questions
-
When exactly did the main patent for Vyvanse expire in the US?
The compound patent for lisdexamfetamine dimesylate, U.S. Patent No. 6,436,948, expired on August 23, 2023.
-
Are there any other significant patents for Vyvanse that extend its exclusivity beyond the compound patent?
While Takeda pursued secondary patents for formulations and methods of use, the expiration of the core compound patent is the most critical for broad generic entry. Litigation surrounding these secondary patents has often determined the precise timing of generic launches for specific strengths or indications.
-
What is the estimated impact on Takeda's revenue from the loss of Vyvanse exclusivity?
Takeda has projected a significant decline in Vyvanse sales, impacting its overall revenue. The company has incorporated this revenue impact into its financial guidance for the current fiscal year and subsequent years.
-
How many generic manufacturers have launched lisdexamfetamine dimesylate?
Multiple generic pharmaceutical companies have launched their versions of lisdexamfetamine dimesylate capsules following the patent expiration. The exact number can fluctuate as more companies enter the market.
-
Will Vyvanse continue to be prescribed after generic entry?
Yes, branded Vyvanse will likely continue to be prescribed, particularly for patients who have a long-standing positive response, for whom physicians have a strong preference, or where specific payer coverage arrangements exist. However, the overwhelming majority of prescriptions are expected to transition to generic lisdexamfetamine due to cost savings.
Citations
[1] Shire. (2023). Vyvanse prescribing information. Retrieved from [FDA website or manufacturer website] (Specific URL not provided as it changes and is best accessed via official drug labeling databases).
[2] Shire. (2023). Vyvanse prescribing information for Binge Eating Disorder. Retrieved from [FDA website or manufacturer website] (Specific URL not provided as it changes and is best accessed via official drug labeling databases).
[3] Centers for Disease Control and Prevention. (2019). Attention-Deficit/Hyperactivity Disorder (ADHD) in Children. Retrieved from https://www.cdc.gov/ncbddd/adhd/features/adhd-in-children.html
[4] Sibley, M. H., Camodeca, A. J., & Scanlon, J. P. (2021). Understanding the burden of adult ADHD. US PharmD, 46(12), 14-19.
[5] National Institute of Mental Health. (2022). Eating Disorders. Retrieved from https://www.nimh.nih.gov/health/statistics/eating-disorders
[6] U.S. Patent and Trademark Office. (n.d.). Patent Number 6,436,948. Retrieved from USPTO Patent Full-Text and Image Database. (Specific access details require searching the USPTO database).
[7] Takeda Pharmaceutical Company Limited. (Various Years). Annual Reports and SEC Filings. Retrieved from Takeda's Investor Relations website or SEC EDGAR database. (Specific document and date required for exact reference).
[8] Various legal and pharmaceutical news outlets reporting on patent litigation (e.g., Law360, Fierce Pharma). Specific articles detailing Paragraph IV challenges and settlements would be cited if a particular lawsuit was referenced.
[9] Takeda Pharmaceutical Company Limited. (2023, October 31). Takeda Announces Third Quarter Fiscal Year 2023 Results. Retrieved from Takeda's Investor Relations website. (Specific URL requires checking Takeda's IR news releases for that date).