VOSEVI Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Vosevi, and what generic alternatives are available?
Vosevi is a drug marketed by Gilead Sciences Inc and is included in one NDA. There are eighteen patents protecting this drug.
This drug has six hundred and twenty-two patent family members in forty-nine countries.
The generic ingredient in VOSEVI is sofosbuvir; velpatasvir; voxilaprevir. There are nine drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the sofosbuvir; velpatasvir; voxilaprevir profile page.
DrugPatentWatch® Generic Entry Outlook for Vosevi
Vosevi was eligible for patent challenges on June 28, 2020.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be December 1, 2037. This may change due to patent challenges or generic licensing.
There have been three patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for VOSEVI?
- What are the global sales for VOSEVI?
- What is Average Wholesale Price for VOSEVI?
Summary for VOSEVI
| International Patents: | 622 |
| US Patents: | 18 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Clinical Trials: | 3 |
| Formulation / Manufacturing: | see details |
| Drug Prices: | Drug price information for VOSEVI |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for VOSEVI |
| What excipients (inactive ingredients) are in VOSEVI? | VOSEVI excipients list |
| DailyMed Link: | VOSEVI at DailyMed |
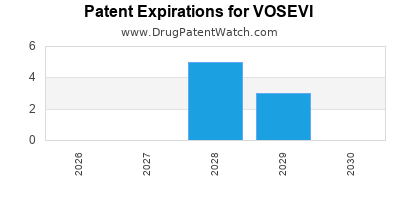

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for VOSEVI
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for VOSEVI
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Peking University People's Hospital | Phase 4 |
| Partners in Health | Phase 4 |
| Gilead Sciences | Phase 3 |
Pharmacology for VOSEVI
US Patents and Regulatory Information for VOSEVI
VOSEVI is protected by nineteen US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of VOSEVI is ⤷ Sign Up.
This potential generic entry date is based on patent ⤷ Sign Up.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting VOSEVI
Combination formulation of three antiviral compounds
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Combination formulation of two antiviral compounds
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Combination formulation of three antiviral compounds
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Nucleoside phosphoramidate prodrugs
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Nucleoside phosphoramidate prodrugs
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Antiviral compounds
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Nucleoside phosphoramidate prodrugs
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Nucleoside phosphoramidates
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Nucleoside phosphoramidates
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Nucleoside phosphoramidate prodrugs
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Compositions and methods for treating hepatitis C virus
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Antiviral compounds
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Antiviral compounds
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Nucleoside phosphoramidate prodrugs
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH CHRONIC HCV INFECTION WHO HAVE GENOTYPE 1A OR 3 INFECTION AND HAVE PREVIOUSLY BEEN TREATED WITH AN HCV REGIMEN CONTAINING SOFOSBUVIR WITHOUT AN NS5A INHIBITOR
Nucleoside phosphoramidate prodrugs
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH CHRONIC HCV INFECTION WHO HAVE GENOTYPE 1, 2, 3, 4, 5, OR 6 INFECTION AND HAVE PREVIOUSLY BEEN TREATED WITH AN HCV REGIMEN CONTAINING AN NS5A INHIBITOR
Nucleoside phosphoramidate prodrugs
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Nucleoside phosphoramidates
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Inhibitors of hepatitis C virus
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Antiviral compounds
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gilead Sciences Inc | VOSEVI | sofosbuvir; velpatasvir; voxilaprevir | TABLET;ORAL | 209195-001 | Jul 18, 2017 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| Gilead Sciences Inc | VOSEVI | sofosbuvir; velpatasvir; voxilaprevir | TABLET;ORAL | 209195-001 | Jul 18, 2017 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Gilead Sciences Inc | VOSEVI | sofosbuvir; velpatasvir; voxilaprevir | TABLET;ORAL | 209195-001 | Jul 18, 2017 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Gilead Sciences Inc | VOSEVI | sofosbuvir; velpatasvir; voxilaprevir | TABLET;ORAL | 209195-001 | Jul 18, 2017 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Gilead Sciences Inc | VOSEVI | sofosbuvir; velpatasvir; voxilaprevir | TABLET;ORAL | 209195-001 | Jul 18, 2017 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for VOSEVI
When does loss-of-exclusivity occur for VOSEVI?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 8616
Patent: FORMULACIÓN COMBINADA DE TRES COMPUESTOS ANTIVIRALES
Estimated Expiration: ⤷ Sign Up
Australia
Patent: 17273851
Patent: Combination formulation of three antiviral compounds
Estimated Expiration: ⤷ Sign Up
Brazil
Patent: 2017011025
Patent: formulação de combinação de três compostos antivirais
Estimated Expiration: ⤷ Sign Up
Canada
Patent: 25380
Patent: FORMULATION COMBINEE DE TROIS COMPOSES ANTIVIRAUX (COMBINATION FORMULATION OF THREE ANTIVIRAL COMPOUNDS)
Estimated Expiration: ⤷ Sign Up
China
Patent: 9310678
Patent: 三种抗病毒化合物的组合制剂 (Combination formulation of three antiviral compounds)
Estimated Expiration: ⤷ Sign Up
Eurasian Patent Organization
Patent: 1892376
Patent: КОМБИНИРОВАННЫЙ СОСТАВ ТРЕХ ПРОТИВОВИРУСНЫХ СОЕДИНЕНИЙ
Estimated Expiration: ⤷ Sign Up
European Patent Office
Patent: 63346
Patent: FORMULATION COMBINÉE DE TROIS COMPOSÉS ANTIVIRAUX (COMBINATION FORMULATION OF THREE ANTIVIRAL COMPOUNDS)
Estimated Expiration: ⤷ Sign Up
Israel
Patent: 3155
Patent: פורמולציה משולבת של שלוש תרכובות אנטי-ויראליות (Combination formulation of three antiviral compounds)
Estimated Expiration: ⤷ Sign Up
Japan
Patent: 19517492
Patent: 3種の抗ウイルス化合物の組み合わせ製剤
Estimated Expiration: ⤷ Sign Up
Mexico
Patent: 18014790
Patent: FORMULACION COMBINADA DE TRES COMPUESTOS ANTIVIRALES. (COMBINATION FORMULATION OF THREE ANTIVIRAL COMPOUNDS.)
Estimated Expiration: ⤷ Sign Up
Morocco
Patent: 142
Patent: FORMULATION COMBINÉE DE TROIS COMPOSÉS ANTIVIRAUX
Estimated Expiration: ⤷ Sign Up
Singapore
Patent: 201810189S
Patent: COMBINATION FORMULATION OF THREE ANTIVIRAL COMPOUNDS
Estimated Expiration: ⤷ Sign Up
South Korea
Patent: 190014536
Patent: 3종의 항바이러스 화합물의 조합 제제
Estimated Expiration: ⤷ Sign Up
Taiwan
Patent: 1818932
Patent: Combination formulation of three antiviral compounds
Estimated Expiration: ⤷ Sign Up
Uruguay
Patent: 261
Patent: FORMULACIÓN COMBINADA DE TRES COMPUESTOS ANTIVIRALES
Estimated Expiration: ⤷ Sign Up
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering VOSEVI around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Japan | 2016053096 | C型肝炎ウイルスを処置するための組成物および方法 (COMPOSITIONS AND METHODS FOR TREATING HEPATITIS C VIRUS) | ⤷ Sign Up |
| Argentina | 080819 | SINTESIS ESTEREOSELECTIVA DE ACTIVOS QUE CONTIENEN FOSFORO | ⤷ Sign Up |
| South Korea | 101603817 | ⤷ Sign Up | |
| Malaysia | 187735 | COMPOSITIONS AND METHODS FOR TREATING HEPATITIS C VIRUS | ⤷ Sign Up |
| San Marino | T201500285 | (S)-ISOPROPIL 2-(((S)-(((2R,3R,4R,5R)-5-(2,4-DIOSSO-3,4-DIIDROPIRIMIDIN-1-(2H)-IL)-4-FLUORO-3-IDROSSI-4-METILTETRAIDROFURAN-2-IL)METOSSI)(FENOSSI)FOSFORIL)AMMINO)PROPANOATO CRISTALLINO | ⤷ Sign Up |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for VOSEVI
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2203462 | 67/2014 | Austria | ⤷ Sign Up | PRODUCT NAME: SOFOSBUVIR; REGISTRATION NO/DATE: EU/1/13/894 20140117 |
| 2203462 | 300704 | Netherlands | ⤷ Sign Up | PRODUCT NAME: SOFOSBUVIR; REGISTRATION NO/DATE: EU/1/13/894/001-002 20140117 |
| 2635588 | C02635588/01 | Switzerland | ⤷ Sign Up | PRODUCT NAME: VELPATASVIR + SOFOSBUVIR + VOXILAPREVIR; REGISTRATION NO/DATE: SWISSMEDIC-ZULASSUNG 66510 08.12.2017 |
| 2203462 | C201430078 | Spain | ⤷ Sign Up | PRODUCT NAME: SOFOSBUVIR; NATIONAL AUTHORISATION NUMBER: EU/1/13/894; DATE OF AUTHORISATION: 20140116; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/13/894; DATE OF FIRST AUTHORISATION IN EEA: 20140116 |
| 2203462 | 1491066-5 | Sweden | ⤷ Sign Up | PRODUCT NAME: SOFOSBUVIR; FIRST MARKETING AUTHORIZATION NUMBER SE: EG EU/1/13/894, 2014-01-17 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


