Last updated: February 11, 2026
What Are the Market Dynamics for VIGAMOX?
VIGAMOX (moxifloxacin ophthalmic solution) is used to treat bacterial conjunctivitis and other eye infections. Sales have been driven by its broad-spectrum activity, prescription frequency, and the prevalence of eye infections.
Market Size and Growth Drivers
- The global ophthalmic drugs market was valued at approximately $49 billion in 2022, with infectious disease treatments accounting for 15–20%.
- Eye infection incidences, including bacterial conjunctivitis, affect an estimated 1.8 million Americans annually (American Academy of Ophthalmology).
- VIGAMOX holds a significant share, especially in the United States, competing with antibiotics like besifloxacin and ofloxacin.
- The drug’s market penetration benefits from prescriptions in adult populations, especially with increased awareness and diagnosis.
Competitive Landscape
- VIGAMOX’s main competitors are other fluoroquinolone eye drops. It faces competition from off-label uses of oral antibiotics but maintains dominance in topical formulations.
- Patent exclusivity expired in August 2023, opening potential for generic entrants.
- Industry shifts include increased surveillance for antibiotic resistance, affecting prescribing habits.
Regulatory and Patent Status
- VIGAMOX received FDA approval in 2003.
- It was protected by patents until August 2023, after which generic versions entered the market.
How Does VIGAMOX’s Financial Trajectory Look?
Revenue Trends
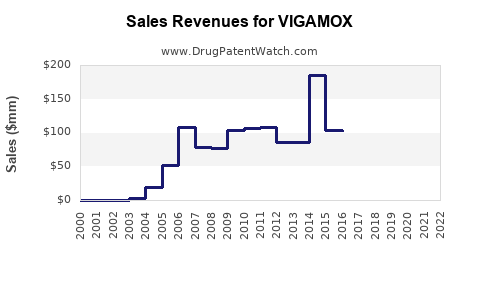
- Pre-2023: Peak global sales reached approximately $250 million annually, driven by U.S. prescriptions and limited generics.
- Post-patent expiry: Market share decline expected; generic competition weighed on revenue.
Profitability
- The drug was highly profitable with margins exceeding 40% during patent protection.
- Costs of manufacturing are relatively low due to established production processes.
- Revenue erosion expected in 2024-2025 due to price competition.
Market Projections
| Year |
Estimated Revenue (USD millions) |
Key Factors |
| 2023 |
240–250 |
Before patent expiry |
| 2024 |
120–150 |
Entry of generics, price competition |
| 2025 |
80–100 |
Continued generic penetration |
Impact of Generic Entry
- Price reductions of 60–80% observed in similar ophthalmic drugs post-generic entry.
- Sales volume could increase in some markets as affordability improves, but overall revenue declines.
What Are the Future Market and Financial Risks?
- Patent Litigation and Patent Strategies: Patent litigation or new patent filings may influence the timing of generic entry.
- Antibiotic Stewardship: Growing emphasis on antibiotic resistance may lower prescription volumes.
- Regulatory Changes: New guidelines or restrictions on antibiotic use could further diminish sales.
- Market Penetration: Developments in branded or combination therapies could impact VIGAMOX’s market share.
How Do Market Trends Affect VIGAMOX?
- Rise in generic competition reduces revenues.
- Increasing use of preservative-free formulations poses a threat to VIGAMOX, which contains benzalkonium chloride.
- New formulations or delivery methods could regain some market share.
Key Takeaways
- VIGAMOX was a leading ophthalmic antibiotic with peak sales of around $250 million before patent expiry.
- The expiration in August 2023 led to the entry of generics, with revenues expected to decline substantially.
- Competitive pressures, antibiotic stewardship policies, and potential formulation innovations influence its future market position.
- Industry growth is tied to the prevalence of bacterial eye infections, which remain common globally.
- Financial margins diminished post-generic entry, reflecting typical trends in branded pharmaceuticals.
FAQs
1. How quickly did generic versions of VIGAMOX enter the market after patent expiry?
Within weeks of patent expiration in August 2023, multiple generics launched, leading to immediate price reductions.
2. What is the primary driver for sales in the ophthalmic antibiotic market?
The number of bacterial eye infection cases combined with physician prescribing habits.
3. How susceptible is VIGAMOX to market threats from resistance?
Resistance development has been reported in fluoroquinolones, which may reduce efficacy and prescribing.
4. Are there any notable regulatory developments affecting VIGAMOX?
No recent approvals or restrictions; however, stewardship initiatives may influence prescribing behavior.
5. What strategies could maintain profitability post-generic entry?
Differentiation through formulation improvements, expanding indications, or licensing alternative delivery systems.
[1] IQVIA, Global Ophthalmic Drugs Market Report, 2022
[2] American Academy of Ophthalmology, Eye Infection Statistics, 2022
[3] U.S. Food and Drug Administration, Drug Patent and Exclusivity Data, 2023