VIEKIRA XR Drug Patent Profile
✉ Email this page to a colleague
When do Viekira Xr patents expire, and what generic alternatives are available?
Viekira Xr is a drug marketed by Abbvie and is included in one NDA. There are eighteen patents protecting this drug.
This drug has five hundred and fifty-two patent family members in fifty-two countries.
The generic ingredient in VIEKIRA XR is dasabuvir sodium; ombitasvir; paritaprevir; ritonavir. Additional details are available on the dasabuvir sodium; ombitasvir; paritaprevir; ritonavir profile page.
DrugPatentWatch® Generic Entry Outlook for Viekira Xr
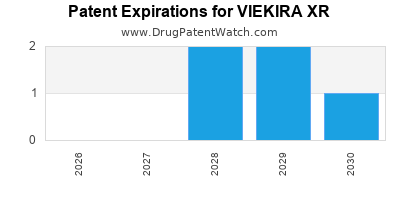
Viekira Xr was eligible for patent challenges on December 19, 2018.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be April 13, 2032. This may change due to patent challenges or generic licensing.
There have been seventeen patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for VIEKIRA XR?
- What are the global sales for VIEKIRA XR?
- What is Average Wholesale Price for VIEKIRA XR?
Summary for VIEKIRA XR
| International Patents: | 552 |
| US Patents: | 18 |
| Applicants: | 1 |
| NDAs: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Clinical Trials: | 3 |
| Patent Applications: | 72 |
| Drug Prices: | Drug price information for VIEKIRA XR |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for VIEKIRA XR |
| DailyMed Link: | VIEKIRA XR at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for VIEKIRA XR
Generic Entry Date for VIEKIRA XR*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET, EXTENDED RELEASE;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for VIEKIRA XR
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Massachusetts General Hospital | Phase 4 |
| Patient-Centered Outcomes Research Institute | Phase 4 |
| Merck Sharp & Dohme Corp. | Phase 4 |
US Patents and Regulatory Information for VIEKIRA XR
VIEKIRA XR is protected by eighteen US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of VIEKIRA XR is ⤷ Start Trial.
This potential generic entry date is based on patent ⤷ Start Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Abbvie | VIEKIRA XR | dasabuvir sodium; ombitasvir; paritaprevir; ritonavir | TABLET, EXTENDED RELEASE;ORAL | 208624-001 | Jul 22, 2016 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Abbvie | VIEKIRA XR | dasabuvir sodium; ombitasvir; paritaprevir; ritonavir | TABLET, EXTENDED RELEASE;ORAL | 208624-001 | Jul 22, 2016 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Abbvie | VIEKIRA XR | dasabuvir sodium; ombitasvir; paritaprevir; ritonavir | TABLET, EXTENDED RELEASE;ORAL | 208624-001 | Jul 22, 2016 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Abbvie | VIEKIRA XR | dasabuvir sodium; ombitasvir; paritaprevir; ritonavir | TABLET, EXTENDED RELEASE;ORAL | 208624-001 | Jul 22, 2016 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Abbvie | VIEKIRA XR | dasabuvir sodium; ombitasvir; paritaprevir; ritonavir | TABLET, EXTENDED RELEASE;ORAL | 208624-001 | Jul 22, 2016 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for VIEKIRA XR
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Abbvie | VIEKIRA XR | dasabuvir sodium; ombitasvir; paritaprevir; ritonavir | TABLET, EXTENDED RELEASE;ORAL | 208624-001 | Jul 22, 2016 | ⤷ Start Trial | ⤷ Start Trial |
| Abbvie | VIEKIRA XR | dasabuvir sodium; ombitasvir; paritaprevir; ritonavir | TABLET, EXTENDED RELEASE;ORAL | 208624-001 | Jul 22, 2016 | ⤷ Start Trial | ⤷ Start Trial |
| Abbvie | VIEKIRA XR | dasabuvir sodium; ombitasvir; paritaprevir; ritonavir | TABLET, EXTENDED RELEASE;ORAL | 208624-001 | Jul 22, 2016 | ⤷ Start Trial | ⤷ Start Trial |
| Abbvie | VIEKIRA XR | dasabuvir sodium; ombitasvir; paritaprevir; ritonavir | TABLET, EXTENDED RELEASE;ORAL | 208624-001 | Jul 22, 2016 | ⤷ Start Trial | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for VIEKIRA XR
When does loss-of-exclusivity occur for VIEKIRA XR?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 7060
Patent: COMPUESTOS ANTIVIRALES
Estimated Expiration: ⤷ Start Trial
Patent: 3240
Patent: COMPOSICIONES SOLIDAS PARA TRATAMIENTO DE INFECCIONES DEL HCV (HEPATITIS C)
Estimated Expiration: ⤷ Start Trial
Patent: 3398
Patent: COMPUESTOS ANTIVIRALES
Estimated Expiration: ⤷ Start Trial
Patent: 2505
Patent: COMPUESTOS ANTIVIRALES
Estimated Expiration: ⤷ Start Trial
Patent: 4816
Patent: COMPUESTOS ANTIVIRALES
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 10258769
Patent: Anti-viral compounds to treat HCV infection
Estimated Expiration: ⤷ Start Trial
Patent: 11264823
Patent: Solid compositions
Estimated Expiration: ⤷ Start Trial
Patent: 11316506
Patent: Anti-viral compounds
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2012031500
Estimated Expiration: ⤷ Start Trial
Patent: 2013005701
Patent: compostos antivirais
Estimated Expiration: ⤷ Start Trial
Patent: 2012031500
Estimated Expiration: ⤷ Start Trial
Patent: 1004894
Patent: compostos antivirais
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 37601
Patent: COMPOSES ANTIVIRAUX (ANTI-VIRAL COMPOUNDS)
Estimated Expiration: ⤷ Start Trial
Patent: 02180
Patent: COMPOSITIONS SOLIDES (SOLID COMPOSITIONS)
Estimated Expiration: ⤷ Start Trial
Patent: 07847
Patent: COMPOSES ANTIVIRAUX (ANTI-VIRAL COMPOUNDS)
Estimated Expiration: ⤷ Start Trial
Patent: 19894
Patent: COMPOSES ANTIVIRAUX (ANTI-VIRAL COMPOUNDS)
Estimated Expiration: ⤷ Start Trial
Patent: 28495
Patent: COMPOSES ANTIVIRAUX (ANTI-VIRAL COMPOUNDS)
Estimated Expiration: ⤷ Start Trial
Patent: 47910
Patent: COMPOSES ANTIVIRAUX (ANTI-VIRAL COMPOUNDS)
Estimated Expiration: ⤷ Start Trial
Patent: 38547
Patent: COMPOSES ANTIVIRAUX (ANTI-VIRAL COMPOUNDS)
Estimated Expiration: ⤷ Start Trial
Patent: 97170
Patent: DERIVES ANTI-VIRAUX DE TETRAHYDROFURANE (ANTI-VIRAL TETRAHYDROFURANE DERIVATIVES)
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 11000689
Patent: Compuesto derivado de ((4-terbutilfenil)pirrolidin-2,5-diil)bis(4,1-fenilen)sustituido, inhibidor de la replicacion del vhc; composicion farmaceutica; util en el tratamiento de una infeccion por vhc.
Estimated Expiration: ⤷ Start Trial
Patent: 12003470
Patent: Una composicion solida que comprende 1) un compuesto inhibidor del hcv de formula definida, o una sal farmaceuticamente aceptable de este, en una forma amorfa, 2) un polimero hidrofilico, y opcionalmente 3) un agente tensoactivo; su proceso de preparacion, util para tratar una infeccion del hcv.
Estimated Expiration: ⤷ Start Trial
Patent: 13000970
Patent: Compuestos derivados de heterociclos nitrogenados sustituidos; sus composiciones farmaceuticas, y uso como inhiibidores de la replicacion del hcv para el tratamiento de la hepatitis c.
Estimated Expiration: ⤷ Start Trial
Patent: 13002299
Patent: Compuestos derivados de heterociclos, inhibidores de la replicacion del virus de la hepatitis c; composicion farmaceutica que los comprende; util en el tratamiento de una infeccion por vhc. (divisional solicitud 689-2011).
Estimated Expiration: ⤷ Start Trial
Patent: 14000059
Patent: Compuestos derivados de heterociclos nitrogenados sustituidos; proceso de preparacion; compuesto intermediario; composiciones farmaceuticas; utiles como inhiibidores de la replicacion del hcv para tratar la hepatitis c. (div. sol. 970-13)
Estimated Expiration: ⤷ Start Trial
China
Patent: 2333772
Patent: Anti-viral compounds
Estimated Expiration: ⤷ Start Trial
Patent: 3153988
Patent: Anti-viral compounds
Estimated Expiration: ⤷ Start Trial
Patent: 3172620
Patent: Anti-viral compounds
Estimated Expiration: ⤷ Start Trial
Patent: 3209686
Patent: Solid compositions
Estimated Expiration: ⤷ Start Trial
Patent: 3354808
Patent: Anti-viral compounds
Estimated Expiration: ⤷ Start Trial
Patent: 3596941
Patent: Anti-viral compounds
Estimated Expiration: ⤷ Start Trial
Patent: 3819459
Patent: Anti-Viral Compounds
Estimated Expiration: ⤷ Start Trial
Patent: 3819537
Patent: Anti-viral compounds
Estimated Expiration: ⤷ Start Trial
Patent: 4193729
Patent: ANTI-VIRAL COMPOUNDS
Estimated Expiration: ⤷ Start Trial
Patent: 6986861
Patent: 抗病毒化合物 (Anti-viral compounds)
Estimated Expiration: ⤷ Start Trial
Patent: 8350016
Patent: 抗病毒四氢呋喃衍生物 (Anti-viral tetrahydrofurane derivatives)
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 40538
Patent: COMPUESTOS ANTIVIRALES
Estimated Expiration: ⤷ Start Trial
Patent: 60490
Patent: Composiciones solidas
Estimated Expiration: ⤷ Start Trial
Patent: 61348
Patent: Compuestos antivirales
Estimated Expiration: ⤷ Start Trial
Costa Rica
Patent: 120650
Patent: COMPOSICIONES SÓLIDAS
Estimated Expiration: ⤷ Start Trial
Patent: 130170
Patent: COMPUESTOS ANTIVIRALES
Estimated Expiration: ⤷ Start Trial
Patent: 140021
Patent: COMPUESTOS ANTIVIRALES
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0130671
Estimated Expiration: ⤷ Start Trial
Patent: 0150017
Estimated Expiration: ⤷ Start Trial
Patent: 0150926
Estimated Expiration: ⤷ Start Trial
Patent: 0151389
Estimated Expiration: ⤷ Start Trial
Patent: 0160408
Estimated Expiration: ⤷ Start Trial
Patent: 0181658
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 14356
Estimated Expiration: ⤷ Start Trial
Patent: 16060
Estimated Expiration: ⤷ Start Trial
Patent: 16748
Estimated Expiration: ⤷ Start Trial
Patent: 17188
Estimated Expiration: ⤷ Start Trial
Patent: 17456
Estimated Expiration: ⤷ Start Trial
Patent: 22267
Estimated Expiration: ⤷ Start Trial
Patent: 15012
Estimated Expiration: ⤷ Start Trial
Patent: 17031
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 68890
Estimated Expiration: ⤷ Start Trial
Patent: 55376
Estimated Expiration: ⤷ Start Trial
Patent: 79854
Estimated Expiration: ⤷ Start Trial
Patent: 28481
Estimated Expiration: ⤷ Start Trial
Patent: 92346
Estimated Expiration: ⤷ Start Trial
Patent: 54892
Estimated Expiration: ⤷ Start Trial
Dominican Republic
Patent: 011000087
Patent: Compuestos eficaces para inhibir la replicación del virus de la hepatitis c("HCV")
Estimated Expiration: ⤷ Start Trial
Patent: 012000307
Patent: COMPOSICIONES SOLIDAS
Estimated Expiration: ⤷ Start Trial
Patent: 013000078
Patent: COMPUESTO DERIVADO DE HETEROCICLOS NITROGENADOS SUSTITUIDOS, EFICAZ COMO INHIBIDOR DE LA REPLICACIÓN DEL VIRUS DE LA HEPATITIS C, COMPOSICIÓN QUE LO COMPRENDE Y SU USO
Estimated Expiration: ⤷ Start Trial
Patent: 013000185
Patent: COMPUESTOS ANTIVIRALES
Estimated Expiration: ⤷ Start Trial
Ecuador
Patent: 11010937
Patent: COMPUESTOS ANTIVIRALES
Estimated Expiration: ⤷ Start Trial
Patent: 13010937
Patent: COMPUESTOS ANTIVIRALES
Estimated Expiration: ⤷ Start Trial
Patent: 13012382
Patent: COMPOSICIONES SÓLIDAS
Estimated Expiration: ⤷ Start Trial
Patent: 13012622
Patent: COMPUESTOS ANTIVIRALES
Estimated Expiration: ⤷ Start Trial
Patent: 14012622
Patent: COMPUESTOS ANTIVIRALES
Estimated Expiration: ⤷ Start Trial
Patent: 17064986
Patent: COMPUESTOS ANTIVIRALES
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 0031
Patent: ПРОТИВОВИРУСНЫЕ СОЕДИНЕНИЯ (ANTI-VIRAL COMPOUNDS)
Estimated Expiration: ⤷ Start Trial
Patent: 3570
Patent: ПРОТИВОВИРУСНЫЕ СОЕДИНЕНИЯ (ANTI-VIRAL COMPOUNDS)
Estimated Expiration: ⤷ Start Trial
Patent: 4100
Patent: МЕТИЛ {(2S,3R)-1-[(2S)-2-{5-[(2R,5R)-1-{3,5-ДИФТОР-4-[4-(4-ФТОРФЕНИЛ)ПИПЕРИДИН-1-ИЛ]ФЕНИЛ}-5-(6-ФТОР-2-{(2S)-1-[N-(МЕТОКСИКАРБОНИЛ)-О-МЕТИЛ-L-ТРЕОНИЛ]ПИРРОЛИДИН-2-ИЛ}-1H-БЕНЗИМИДАЗОЛ-5-ИЛ)ПИРРОЛИДИН-2-ИЛ]-6-ФТОР-1H-БЕНЗИМИДАЗОЛ-2-ИЛ}ПИРРОЛИДИН-2-ИЛ]-3-МЕТОКСИ-1-ОКСОБУТАН-2-ИЛ}КАРБАМАТ, ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ, ВКЛЮЧАЮЩАЯ ЕГО, И СПОСОБ ЛЕЧЕНИЯ ИНФЕКЦИИ ГЕПАТИТА C (METHYL {(2S,3R)-1-[(2S)-2-{5-[(2R,5R)-1-{3,5-DIFLUORO-4-[4-(4-FLUOROPHENYL)PIPERIDIN-1-YL]PHENYL}-5-(6-FLUORO-2-{(2S)-1-[N-(METHOXYCARBONYL)-O-METHYL-L-THREONYL]PYRROLIDIN-2-yl}-1H-BENZIMIDAZOL-5-YL)PYRROLIDIN-2-YL]-6-FLUORO-1H-BENZIMIDAZOL-2-YL}PYRROLIDIN-1-YL]-3-METHOXY-1-OXOBUTAN-2-YL}CARBAMATE, PHARMACEUTICAL COMPOSITION COMPRISING SAME, AND METHOD OF TREATING HCV INFECTION)
Estimated Expiration: ⤷ Start Trial
Patent: 4538
Patent: ТВЕРДАЯ ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ, СОДЕРЖАЩАЯ СОЕДИНЕНИЕ С АНТИ-ВГС (HCV) АКТИВНОСТЬЮ (SOLID PHARMACEUTICAL COMPOSITION COMPRISING A COMPOUND WITH ANTI-HCV ACTIVITY)
Estimated Expiration: ⤷ Start Trial
Patent: 6848
Patent: ПРОТИВОВИРУСНЫЕ СОЕДИНЕНИЯ (ANTIVIRAL COMPOUNDS)
Estimated Expiration: ⤷ Start Trial
Patent: 3332
Patent: СПОСОБ ПОЛУЧЕНИЯ МЕТИЛ {(2S,3R)-1-[(2S)-2-{5-[(2R,5R)-1-{3,5-ДИФТОР-4-[4-(4-ФТОРФЕНИЛ)ПИПЕРИДИН-1-ИЛ]ФЕНИЛ}-5-(6-ФТОР-2-{(2S)-1-[N-(МЕТОКСИКАРБОНИЛ)-О-МЕТИЛ-L-ТРЕОНИЛ]ПИРРОЛИДИН-2-ИЛ}-1Н-БЕНЗИМИДАЗОЛ-5-ИЛ)ПИРРОЛИДИН-2-ИЛ]-6-ФТОР-1Н-БЕНЗИМИДАЗОЛ-2-ИЛ}ПИРРОЛИДИН-1-ИЛ]-3-МЕТОКСИ-1-ОКСОБУТАН-2-ИЛ} КАРБАМАТА (PROCESS FOR PREPARING METHYL{(2S,3R)-1-[(2S)-2-{5-[(2R,5R)-1-{3,5-DIFLUORO-4-[4-(4-FLUOROPHENYL)PIPERIDINE-1-YL]PHENYL}-5-(6-FLUORO-2-{(2S)-1-[N-(METHOXYCARBONYL)-O-METHYL-L-THREONYL]PYRROLIDINE-2-YL}-1H-BENZIMIDAZOLE-5-YL)pyrrolidinePYRROLIDINE-2-YL]-6-FLUORO-1H-BENZIMIDAZOLE-2-YL}PYRROLIDINE-1-YL]-3-METHOXY-1-OXOBUTANE-2-YL}CARBAMATE)
Estimated Expiration: ⤷ Start Trial
Patent: 1170401
Patent: ПРОТИВОВИРУСНЫЕ СОЕДИНЕНИЯ
Estimated Expiration: ⤷ Start Trial
Patent: 1291394
Patent: ТВЕРДЫЕ КОМПОЗИЦИИ
Estimated Expiration: ⤷ Start Trial
Patent: 1300495
Patent: ПРОТИВОВИРУСНЫЕ СОЕДИНЕНИЯ
Estimated Expiration: ⤷ Start Trial
Patent: 1301158
Patent: ПРОТИВОВИРУСНЫЕ СОЕДИНЕНИЯ
Estimated Expiration: ⤷ Start Trial
Patent: 1390538
Patent: ПРОТИВОВИРУСНЫЕ СОЕДИНЕНИЯ
Estimated Expiration: ⤷ Start Trial
Patent: 1400115
Patent: ПРОТИВОВИРУСНЫЕ СОЕДИНЕНИЯ
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 37781
Patent: COMPOSÉS ANTIVIRAUX POUR LE TRAITEMENT D'INFECTIONS HCV (ANTI-VIRAL COMPOUNDS TO TREAT HCV INFECTION)
Estimated Expiration: ⤷ Start Trial
Patent: 68890
Patent: Inhibiteurs de la hépatite C (Hepatitis C virus inhibitors)
Estimated Expiration: ⤷ Start Trial
Patent: 55376
Patent: Composés hétérocycliques comme inhibiteurs du virus de'l hepatite C (HCV) (Heterocyclic compounds as inhibitors of hepatitis C virus (HCV))
Estimated Expiration: ⤷ Start Trial
Patent: 79854
Patent: COMPOSITIONS SOLIDES (SOLID COMPOSITIONS)
Estimated Expiration: ⤷ Start Trial
Patent: 27651
Patent: COMPOSÉS ANTIVIRAUX (ANTI-VIRAL COMPOUNDS)
Estimated Expiration: ⤷ Start Trial
Patent: 28481
Patent: Dérivés hétérocycliques trisubstitués comme inhibiteurs de la réplication du virus de l'hépatite C HCV (Trisubstituted heterocycles as replication inhibitors of hepatitis C virus HCV)
Estimated Expiration: ⤷ Start Trial
Patent: 51885
Patent: COMPOSÉS ANTIVIRAUX (ANTI-VIRAL COMPOUNDS)
Estimated Expiration: ⤷ Start Trial
Patent: 78334
Patent: COMPOSÉS ANTIVIRAUX (ANTI-VIRAL COMPOUNDS)
Estimated Expiration: ⤷ Start Trial
Patent: 92346
Patent: Dérivé antiviraux de 2,5-dibenzimidazol-5-yl-1-phényl-pyrrolidine (An antiviral 1-phenyl-2,5-dibenzimidazol-5-yl-pyrrolidine derivative)
Estimated Expiration: ⤷ Start Trial
Patent: 92726
Patent: Composés antiviraux (Anti-viral compounds)
Estimated Expiration: ⤷ Start Trial
Patent: 53531
Patent: Composés antiviraux (Antiviral compounds)
Estimated Expiration: ⤷ Start Trial
Patent: 54892
Patent: COMPOSITIONS SOLIDES (SOLID COMPOSITIONS)
Estimated Expiration: ⤷ Start Trial
Patent: 44642
Patent: DÉRIVÉS ANTI-VIRAUX DE TÉTRAHYDROFURANE (ANTI-VIRAL TETRAHYDROFURANE DERIVATIVES)
Estimated Expiration: ⤷ Start Trial
Patent: 38106
Patent: COMPOSÉS ANTIVIRAUX (ANTI-VIRAL COMPOUNDS)
Estimated Expiration: ⤷ Start Trial
Patent: 56318
Patent: COMPOSITIONS SOLIDES (SOLID COMPOSITIONS)
Estimated Expiration: ⤷ Start Trial
France
Patent: C1040
Estimated Expiration: ⤷ Start Trial
Guatemala
Patent: 1100074
Patent: COMPUESTOS ANTIVIRALES
Estimated Expiration: ⤷ Start Trial
Patent: 1200332
Patent: COMPOSICIONES SÒLIDAS
Estimated Expiration: ⤷ Start Trial
Patent: 1300093
Patent: COMPUESTOS ANTIVIRALES
Estimated Expiration: ⤷ Start Trial
Patent: 1300093A
Patent: COMPUESTOS ANTIVIRALES (SOLICITUD DIVISIONAL FRACCIONARIA DE LA SOLICITUD A-2013-00093)
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 52620
Patent: 治療 感染的抗病毒化合物 (ANTI-VIRAL COMPOUNDS TO TREAT HCV INFECTION HCV)
Estimated Expiration: ⤷ Start Trial
Patent: 61245
Patent: HEPATITIS C VIRUS INHIBITORS
Estimated Expiration: ⤷ Start Trial
Patent: 70739
Patent: 作為丙型肝炎病毒 抑制劑的雜環化合物 (HETEROCYCLIC COMPOUNDS AS INHIBITORS OF HEPATITIS C VIRUS (HCV) (HCV))
Estimated Expiration: ⤷ Start Trial
Patent: 84068
Patent: 固體組合物 (SOLID COMPOSITIONS)
Estimated Expiration: ⤷ Start Trial
Patent: 86415
Patent: 作為丙型肝炎病毒 的複製抑制劑的三取代雜環 (TRISUBSTITUTED HETEROCYCLES AS REPLICATION INHIBITORS OF HEPATITIS C VIRUS HCV HCV)
Estimated Expiration: ⤷ Start Trial
Patent: 88717
Patent: 種抗病毒 -苯基- -二苯並咪唑- -基-吡咯烷衍生物 (AN ANTIVIRAL 1-PHENYL-2,5-DIBENZIMIDAZOL-5-YL-PYRROLIDINE DERIVATIVE 1--25--5--)
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 25758
Estimated Expiration: ⤷ Start Trial
Patent: 26832
Estimated Expiration: ⤷ Start Trial
Patent: 28825
Estimated Expiration: ⤷ Start Trial
Patent: 39719
Estimated Expiration: ⤷ Start Trial
Patent: 500019
Estimated Expiration: ⤷ Start Trial
Patent: 700040
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 1792
Patent: דימתיל (2s,2's)-1,'1-((2s,2's)-2,'2-(4,'4-((2s,5s)-1-(4-טרט-בוטילפניל)פירולידין-5,2-דיאיל)ביס(1,4 פנילן))ביס (אזאנדיאיל)ביס(אוקסומתילן)ביס(פירולידין-1,2-דיאיל))ביס(3- מתיל-1-אוקסובוטן-1,2-דיאיל)דיקארבמאט והרכב רוקחי המכיל אותו (Dimethyl (2s, 2's)-1, 1'-((2s,2's)-2,2'-(4,4' -((2s,5s)-1 -(4-tert-butylphenyl) pyrrolidine-2,5 -diyl) bis (4,1 phenylene)) bis (azanediyl) bis (oxomethylene) bis (pyrrolidine-2,1-diyl)) bis (3-methyl-1-oxobutane-2,1 -diyl) dicarbamate and a pharmaceutical composition comprising same)
Estimated Expiration: ⤷ Start Trial
Patent: 3535
Patent: הרכבים תרופתיים מוצקים (Solid pharmaceutical compositions)
Estimated Expiration: ⤷ Start Trial
Patent: 5010
Patent: מתיל (2s,3r)-1-((1s)-2-(-2-(-5-((2r,3r)-1-(3,5,דיפלואורו-4-(4-(4-פלואורופניל)פיפרידינ-1-איל(פניל)-5-(6-פלואורו-2-((2s)-1-(n-(מתוקסיקרבוניל)-o-מתיל-l-תראוניל)פירולידינ-1-איל)-1י-בנזואימידזול-5-איל)פירולידין-2-איל)-6-פלואורו-1h-בנזאימידזול-2-איל)פירולידינ-1-איל)-3-מתוקסי-1-אוקסובוטאנ-2-איל)קרבאמט והרכב רוקחי המכיל אותו (Methyl {(2s,3r)-1-[(2s)-2-{5-[(2r,5r)-1-{3,5-difluoro-4-[4-(4-fluorophenyl)piperidin-1-yl]phenyl}-5-(6-fluoro-2-{(2s)-1-[n-(methoxycarbonyl)-o-methyl-l-threonyl]pyrrolidin-2-yl}-1h-benzimidazol-5-yl)pyrrolidin-2-yl]-6-fluoro-1h-benzimidazol-2-yl}pyrrolidin-1-yl]-3-methoxy-1-oxobutan-5 2-yl}carbamate and a pharmaceutical composition comprising it)
Estimated Expiration: ⤷ Start Trial
Patent: 9248
Patent: תרכובות נגד נגיפים והרכבים רוקחיים המכילים אותן (Anti-viral compounds and pharmaceutical compositions comprising same)
Estimated Expiration: ⤷ Start Trial
Patent: 3857
Patent: תרכובות נגד נגיפים (Anti-viral compounds)
Estimated Expiration: ⤷ Start Trial
Patent: 4781
Patent: תרכובות נגד נגיפים (Anti-viral compounds)
Estimated Expiration: ⤷ Start Trial
Patent: 1206
Patent: תרכובות אנטי-וירליות (Anti-viral compounds)
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 30514
Estimated Expiration: ⤷ Start Trial
Patent: 14356
Estimated Expiration: ⤷ Start Trial
Patent: 34085
Estimated Expiration: ⤷ Start Trial
Patent: 06253
Estimated Expiration: ⤷ Start Trial
Patent: 11838
Estimated Expiration: ⤷ Start Trial
Patent: 22699
Estimated Expiration: ⤷ Start Trial
Patent: 59830
Estimated Expiration: ⤷ Start Trial
Patent: 59736
Estimated Expiration: ⤷ Start Trial
Patent: 86147
Estimated Expiration: ⤷ Start Trial
Patent: 43135
Estimated Expiration: ⤷ Start Trial
Patent: 90202
Estimated Expiration: ⤷ Start Trial
Patent: 12529534
Estimated Expiration: ⤷ Start Trial
Patent: 13528225
Estimated Expiration: ⤷ Start Trial
Patent: 13539791
Estimated Expiration: ⤷ Start Trial
Patent: 14065710
Patent: ANTI-VIRAL COMPOUNDS FOR TREATING HCV INFECTION
Estimated Expiration: ⤷ Start Trial
Patent: 14144973
Patent: ANTI-VIRAL COMPOUND FOR TREATING HCV INFECTION
Estimated Expiration: ⤷ Start Trial
Patent: 14504296
Estimated Expiration: ⤷ Start Trial
Patent: 14510063
Estimated Expiration: ⤷ Start Trial
Patent: 16106075
Patent: 抗ウィルス化合物 (ANTI-VIRUS COMPOUND)
Estimated Expiration: ⤷ Start Trial
Patent: 16128456
Patent: HCV感染を治療するための抗ウィルス化合物 (ANTI-VIRAL COMPOUNDS FOR TREATING HCV INFECTION)
Estimated Expiration: ⤷ Start Trial
Patent: 17171680
Patent: HCV感染を治療するための抗ウィルス化合物 (ANTI-VIRAL COMPOUNDS TO TREAT HCV INFECTION)
Estimated Expiration: ⤷ Start Trial
Patent: 18065828
Patent: 抗ウィルス化合物 (ANTI-VIRUS COMPOUND)
Estimated Expiration: ⤷ Start Trial
Patent: 18529671
Patent: 抗ウィルス性テトラヒドロフラン誘導体
Estimated Expiration: ⤷ Start Trial
Patent: 19194254
Patent: HCV感染を治療するための抗ウィルス化合物 (ANTI-VIRAL COMPOUNDS FOR TREATING HCV INFECTION)
Estimated Expiration: ⤷ Start Trial
Patent: 20059696
Patent: 抗ウィルス化合物 (ANTI-VIRAL COMPOUNDS)
Estimated Expiration: ⤷ Start Trial
Patent: 21035964
Patent: 抗ウィルス化合物 (ANTI-VIRAL COMPOUNDS)
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 54892
Estimated Expiration: ⤷ Start Trial
Patent: 368890
Estimated Expiration: ⤷ Start Trial
Patent: 692346
Estimated Expiration: ⤷ Start Trial
Patent: 2017033
Estimated Expiration: ⤷ Start Trial
Luxembourg
Patent: 668
Estimated Expiration: ⤷ Start Trial
Patent: 0038
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 4064
Patent: ANTI-VIRAL COMPOUNDS
Estimated Expiration: ⤷ Start Trial
Patent: 4607
Patent: SOLID COMPOSITIONS
Estimated Expiration: ⤷ Start Trial
Patent: 6633
Patent: HETEROCYCLIC COMPOUNDS AS INHIBITORS OF HEPATITIS C VIRUS (HCV)
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 9989
Patent: COMPUESTOS ANTI-VIRALES. (ANTI-VIRAL COMPOUNDS TO TREAT HCV INFECTION.)
Estimated Expiration: ⤷ Start Trial
Patent: 4092
Patent: COMPUESTOS ANTIVIRALES. (ANTI-VIRAL COMPOUNDS.)
Estimated Expiration: ⤷ Start Trial
Patent: 6264
Patent: COMPUESTOS ANTIVIRALES. (ANTI-VIRAL COMPOUNDS.)
Estimated Expiration: ⤷ Start Trial
Patent: 11005673
Patent: COMPUESTOS ANTI-VIRALES PARA TRATAR INFECCION POR VHC. (ANTI-VIRAL COMPOUNDS TO TREAT HCV INFECTION.)
Estimated Expiration: ⤷ Start Trial
Patent: 12014384
Patent: COMPOSICIONES SOLIDAS. (SOLID COMPOSITIONS.)
Estimated Expiration: ⤷ Start Trial
Patent: 13004150
Patent: CONPUESTOS ANTIVIRALES. (ANTI-VIRAL COMPOUNDS.)
Estimated Expiration: ⤷ Start Trial
Patent: 13006951
Patent: COMPUESTOS ANTIVIRALES. (ANTI-VIRAL COMPOUNDS.)
Estimated Expiration: ⤷ Start Trial
Patent: 13009763
Patent: COMPUESTOS ANTIVIRALES. (ANTI-VIRAL COMPOUNDS.)
Estimated Expiration: ⤷ Start Trial
Patent: 18002707
Patent: DERIVADOS DE TETRAHIDROFURANO ANTIVIRALES. (ANTI-VIRAL TETRAHYDROFURANE DERIVATIVES.)
Estimated Expiration: ⤷ Start Trial
Patent: 20002151
Patent: COMPUESTOS ANTIVIRALES. (ANTI-VIRAL COMPOUNDS.)
Estimated Expiration: ⤷ Start Trial
Montenegro
Patent: 201
Patent: PRIPRAVCI U KRUTOM STANJU (SOLID COMPOSITIONS)
Estimated Expiration: ⤷ Start Trial
Patent: 328
Patent: ANTIVIRUSNI 2, 5-DIBENZIMIDAZOL- 5- IL-1-FENIL- PIROLIDINDERIVAT (An antiviral 1- phenyl- 2,5-dibenzimidazol-5-yl-pyrrolidine derivative)
Estimated Expiration: ⤷ Start Trial
Netherlands
Patent: 0731
Estimated Expiration: ⤷ Start Trial
Patent: 0901
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 1973
Patent: ANTI-VIRAL COMPOUNDS TO TREAT HCV INFECTION
Estimated Expiration: ⤷ Start Trial
Patent: 5440
Patent: SOLID COMPOSITIONS COMPRISING AN HCV INHIBITOR
Estimated Expiration: ⤷ Start Trial
Patent: 6645
Patent: Anti-viral compounds
Estimated Expiration: ⤷ Start Trial
Patent: 5562
Patent: Anti-viral compounds
Estimated Expiration: ⤷ Start Trial
Norway
Patent: 15012
Estimated Expiration: ⤷ Start Trial
Patent: 17057
Estimated Expiration: ⤷ Start Trial
Patent: 21039
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 110679
Patent: DERIVADOS DE (4-TERT-BUTILFENIL)PIRROLIDIN-2,5-DIFENIL COMO INHIBIDORES DEL HCV
Estimated Expiration: ⤷ Start Trial
Patent: 131036
Patent: COMPOSICIONES SOLIDAS QUE COMPRENDEN COMPUESTOS ANTI-HCV
Estimated Expiration: ⤷ Start Trial
Patent: 140038
Patent: COMPUESTOS ANTIVIRALES
Estimated Expiration: ⤷ Start Trial
Patent: 140835
Patent: COMPUESTOS ANTIVIRALES
Estimated Expiration: ⤷ Start Trial
Patent: 141083
Patent: COMPUESTOS ANTIVIRALES
Estimated Expiration: ⤷ Start Trial
Philippines
Patent: 012502442
Estimated Expiration: ⤷ Start Trial
Patent: 013500708
Estimated Expiration: ⤷ Start Trial
Patent: 015500289
Patent: ANTI-VIRAL COMPOUNDS
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 68890
Estimated Expiration: ⤷ Start Trial
Patent: 55376
Estimated Expiration: ⤷ Start Trial
Patent: 79854
Estimated Expiration: ⤷ Start Trial
Patent: 28481
Estimated Expiration: ⤷ Start Trial
Patent: 92346
Estimated Expiration: ⤷ Start Trial
Patent: 54892
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 68890
Estimated Expiration: ⤷ Start Trial
Patent: 55376
Estimated Expiration: ⤷ Start Trial
Patent: 79854
Estimated Expiration: ⤷ Start Trial
Patent: 92346
Estimated Expiration: ⤷ Start Trial
Patent: 54892
Estimated Expiration: ⤷ Start Trial
San Marino
Patent: 01500263
Patent: COMPOSIZIONI SOLIDE
Estimated Expiration: ⤷ Start Trial
Patent: 01800532
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 854
Patent: INHIBITORI HEPATITIS C VIRUSA (HEPATITIS C VIRUS INHIBITORS)
Estimated Expiration: ⤷ Start Trial
Patent: 856
Patent: HETEROCIKLIČNA JEDINJENJA KAO INHIBITORI HEPATITIS C VIRUSA (HETEROCYCLIC COMPOUNDS AS INHIBITORS OF HEPATITIS C VIRUS (HCV))
Estimated Expiration: ⤷ Start Trial
Patent: 282
Patent: ČVRSTE SMEŠE (SOLID COMPOSITIONS)
Estimated Expiration: ⤷ Start Trial
Patent: 619
Patent: ANTIVIRUSNI DERIVAT 1-FENIL-2,5-DIBENZIMIDAZOL-5-IL-PIROLIDINA (AN ANTIVIRAL 1-PHENYL-2,5-DIBENZIMIDAZOL-5-YL-PYRROLIDINE DERIVATIVE)
Estimated Expiration: ⤷ Start Trial
Patent: 790
Patent: TRISUPSTITUISANI HETEROCIKLI KAO INHIBITORI REPLIKACIJE VIRUSA HEPATITISA C HCV (TRISUBSTITUTED HETEROCYCLES AS REPLICATION INHIBITORS OF HEPATITIS C VIRUS HCV)
Estimated Expiration: ⤷ Start Trial
Patent: 982
Patent: ČVRSTI SASTAVI (SOLID COMPOSITIONS)
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 201702522U
Patent: ANTI-VIRAL COMPOUNDS TO TREAT HCV INFECTION
Estimated Expiration: ⤷ Start Trial
Patent: 1708
Patent: ANTI-VIRAL COMPOUNDS TO TREAT HCV INFECTION
Estimated Expiration: ⤷ Start Trial
Patent: 6251
Patent: SOLID COMPOSITIONS
Estimated Expiration: ⤷ Start Trial
Patent: 8951
Patent: ANTI-VIRAL COMPOUNDS
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 68890
Estimated Expiration: ⤷ Start Trial
Patent: 55376
Estimated Expiration: ⤷ Start Trial
Patent: 79854
Estimated Expiration: ⤷ Start Trial
Patent: 28481
Estimated Expiration: ⤷ Start Trial
Patent: 92346
Estimated Expiration: ⤷ Start Trial
Patent: 54892
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1102425
Patent: ANTI-VIRAL COMPOUNDS.
Estimated Expiration: ⤷ Start Trial
Patent: 1203502
Patent: ANTI-VIRAL COMPOUNDS
Estimated Expiration: ⤷ Start Trial
Patent: 1203503
Patent: ANTI-VIRAL COMPOUNDS
Estimated Expiration: ⤷ Start Trial
Patent: 1300112
Patent: SOLID COMPOSITIONS
Estimated Expiration: ⤷ Start Trial
Patent: 1302269
Patent: ANTI-VIRAL COMPOUNDS
Estimated Expiration: ⤷ Start Trial
Patent: 1705519
Patent: ANTI-VIRAL COMPOUNDS
Estimated Expiration: ⤷ Start Trial
Patent: 1903284
Patent: ANTI-VIRAL COMPOUNDS
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1452916
Estimated Expiration: ⤷ Start Trial
Patent: 1481395
Estimated Expiration: ⤷ Start Trial
Patent: 1586215
Estimated Expiration: ⤷ Start Trial
Patent: 1677481
Estimated Expiration: ⤷ Start Trial
Patent: 1831154
Estimated Expiration: ⤷ Start Trial
Patent: 1990936
Estimated Expiration: ⤷ Start Trial
Patent: 2059386
Estimated Expiration: ⤷ Start Trial
Patent: 120117620
Patent: ANTI-VIRAL COMPOUNDS TO TREAT HCV INFECTION
Estimated Expiration: ⤷ Start Trial
Patent: 130053440
Patent: 고체 조성물 (SOLID COMPOSITIONS)
Estimated Expiration: ⤷ Start Trial
Patent: 140032338
Patent: ANTI-VIRAL COMPOUNDS
Estimated Expiration: ⤷ Start Trial
Patent: 140037974
Patent: ANTI-VIRAL COMPOUNDS TO TREAT HCV INFECTION
Estimated Expiration: ⤷ Start Trial
Patent: 140143152
Patent: ANTI-VIRAL COMPOUNDS
Estimated Expiration: ⤷ Start Trial
Patent: 150008151
Patent: SOLID COMPOSITIONS
Estimated Expiration: ⤷ Start Trial
Patent: 180023014
Patent: 고체 조성물 (SOLID COMPOSITIONS)
Estimated Expiration: ⤷ Start Trial
Patent: 180026775
Patent: 항바이러스 화합물 (ANTI-VIRAL COMPOUNDS)
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 14934
Estimated Expiration: ⤷ Start Trial
Patent: 11440
Estimated Expiration: ⤷ Start Trial
Patent: 26908
Estimated Expiration: ⤷ Start Trial
Patent: 46767
Estimated Expiration: ⤷ Start Trial
Patent: 60842
Estimated Expiration: ⤷ Start Trial
Patent: 65536
Estimated Expiration: ⤷ Start Trial
Patent: 24246
Estimated Expiration: ⤷ Start Trial
Patent: 91625
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1102063
Patent: Anti-viral compounds
Estimated Expiration: ⤷ Start Trial
Patent: 1204713
Patent: Solid compositions
Estimated Expiration: ⤷ Start Trial
Patent: 1238948
Patent: Anti-viral compounds
Estimated Expiration: ⤷ Start Trial
Patent: 1247648
Patent: Anti-viral compounds
Estimated Expiration: ⤷ Start Trial
Patent: 1334778
Patent: Anti-viral compounds
Estimated Expiration: ⤷ Start Trial
Patent: 1347759
Patent: Anti-viral compounds
Estimated Expiration: ⤷ Start Trial
Patent: 1412707
Patent: Anti-viral compounds
Estimated Expiration: ⤷ Start Trial
Patent: 1519891
Patent: Anti-viral compounds
Estimated Expiration: ⤷ Start Trial
Patent: 02070
Estimated Expiration: ⤷ Start Trial
Patent: 19874
Estimated Expiration: ⤷ Start Trial
Patent: 69780
Estimated Expiration: ⤷ Start Trial
Patent: 86159
Estimated Expiration: ⤷ Start Trial
Patent: 87700
Estimated Expiration: ⤷ Start Trial
Patent: 86660
Estimated Expiration: ⤷ Start Trial
Patent: 21611
Estimated Expiration: ⤷ Start Trial
Turkey
Patent: 1815161
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 3052
Patent: ПРОТИВІРУСНІ СПОЛУКИ[ПРОТИВОВИРУСНЫЕ СОЕДИНЕНИЯ (ANTI-VIRAL COMPOUNDS)
Estimated Expiration: ⤷ Start Trial
Patent: 5434
Patent: ТВЕРДАЯ КОМПОЗИЦИЯ И СПОСОБ ЛЕЧЕНИЯ ГЕПАТИТА С;ТВЕРДА КОМПОЗИЦІЯ ТА СПОСІБ ЛІКУВАННЯ ВІРУСУ ГЕПАТИТУ С (SOLID COMPOSITION AND METHOD FOR TREATMENT OF HEPATITIS C)
Estimated Expiration: ⤷ Start Trial
Patent: 8904
Patent: ПРОТИВІРУСНІ СПОЛУКИ
Estimated Expiration: ⤷ Start Trial
Patent: 3048
Patent: ПРОТИВІРУСНІ СПОЛУКИ
Estimated Expiration: ⤷ Start Trial
Patent: 8080
Patent: ПРОТИВІРУСНІ СПОЛУКИ
Estimated Expiration: ⤷ Start Trial
Uruguay
Patent: 699
Patent: COMPUESTOS ANTIVIRALES EFICACES PARA INHIBIR LA REPLICACIÓN DEL VIRUS DE LA HEPATITIS C ("HCV"), PROCESOS, COMPOSICIONES, Y MÉTODOS RELACIONADOS
Estimated Expiration: ⤷ Start Trial
Patent: 446
Patent: COMPOSICIONES SOLIDAS
Estimated Expiration: ⤷ Start Trial
Patent: 667
Patent: COMPUESTOS ANTIVIRALES
Estimated Expiration: ⤷ Start Trial
Patent: 983
Patent: COMPUESTOS ANTIVIRALES EFICACES PARA INHIBIR LA REPLICACIÓN DEL VIRUS DE LA HEPATITIS C ("HCV") Y C OMPOSICIONES RELACIONADAS
Estimated Expiration: ⤷ Start Trial
Patent: 266
Patent: COMPUESTOS ANTIVIRALES
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering VIEKIRA XR around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Peru | 20061016 | UNA FORMULACION SOLIDA DE DOSIFICACION FARMACEUTICA | ⤷ Start Trial |
| China | 105363035 | ⤷ Start Trial | |
| Norway | 2015011 | ⤷ Start Trial | |
| Eurasian Patent Organization | 033332 | ⤷ Start Trial | |
| Hong Kong | 1148273 | 抗感染嘧啶及其用途 (ANTI-INFECTIVE PYRIMIDINES AND USES THEREOF) | ⤷ Start Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for VIEKIRA XR
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2692346 | 1790050-7 | Sweden | ⤷ Start Trial | PRODUCT NAME: PIBRENTASVIR OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF; REG. NO/DATE: EU/1/17/1213 20170728 |
| 2368890 | 300731 | Netherlands | ⤷ Start Trial | DETAILS ASSIGNMENT: CHANGE OF OWNER(S), ASSIGNMENT |
| 2692346 | 122017000074 | Germany | ⤷ Start Trial | PRODUCT NAME: PIBRENTASVIR ODER EIN PHARMAZEUTISCH VERTRAEGLICHES SALZ DAVON; REGISTRATION NO/DATE: EU/1/17/1213 20170726 |
| 2203431 | CA 2015 00014 | Denmark | ⤷ Start Trial | PRODUCT NAME: DASABUVIR OR A SALT THEREOF, INCLUDING DASABUVIR SODIUM MONOHYDRATE; REG. NO/DATE: EU/1/14/983 20150115 |
| 2203431 | SPC/GB15/008 | United Kingdom | ⤷ Start Trial | PRODUCT NAME: DASABUVIR, OR A SALT THEREOF; REGISTERED: UK EU/1/14/983 20150119 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
VIEKIRA XR: Market Dynamics and Financial Trajectory
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.