Share This Page
Drug Sales Trends for VICODIN HP
✉ Email this page to a colleague
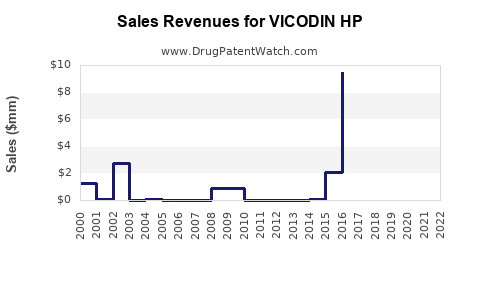
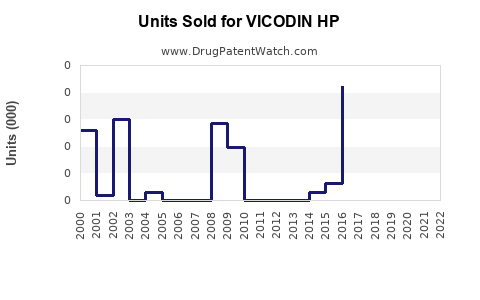
Annual Sales Revenues and Units Sold for VICODIN HP
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| VICODIN HP | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| VICODIN HP | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| VICODIN HP | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| VICODIN HP | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| VICODIN HP | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for VICODIN HP
What is VICODIN HP?
VICODIN HP is a combination analgesic that contains hydrocodone and acetaminophen. It is used to manage moderate to severe pain. The drug's formulation is designed for extended-release with a higher dose of hydrocodone compared to standard formulations. It is available in the United States through Schedule II controlled substances regulations.
Market Overview
Regulatory Status and Patent Outlook
VICODIN HP is subject to strict regulatory oversight. The FDA approved its original formulation in the early 2000s, with patent protections expiring around 2010. Generic versions have since entered the market, intensifying competition.
The manufacturer, Mallinckrodt, faced legal actions related to marketing practices, influencing market dynamics. Patent litigation involving formulation patents has limited new entry for extended-release hydrocodone products until recent regulatory shifts.
Current Market Demand
The demand for opioid analgesics like VICODIN HP remains significant in pain management, particularly for chronic pain conditions. Despite increased regulatory scrutiny and the opioid epidemic, prescriptions continue driven by:
- Clinical guidelines favoring opioids for severe pain.
- A lack of equivalent non-opioid, extended-release options.
- Patient and provider preference for established formulations.
Annual prescriptions for hydrocodone products in the U.S. exceed 60 million, with a sizable share attributed to extended-release formulations.
Competitive Landscape
Major competitors include:
- Purdue Pharma's OxyContin (oxycodone)
- Teva's generic hydrocodone combinations
- Endo's extended-release opioids
- Lilly’s formulations (if any)
The generics account for approximately 70% of the hydrocone market, with branded products capturing the remaining share.
Market Risks
- Regulatory tightening on opioid sales and prescribing.
- Growing consumer and legislative pressure to reduce opioid use.
- Potential for market shrinkage due to alternative therapies or abuse-deterrent formulations.
Sales Projections (2023-2028)
Assumptions
- Prescriptions sustain at current levels with slight declines due to regulatory pressures.
- Generic market penetration remains high.
- No major breakthroughs in non-opioid pain management.
Revenue Estimations
In 2022, the U.S. sales of hydrocodone extended-release products, including VICODIN HP, were approximately $2.1 billion.
| Year | Estimated Sales (USD Million) | Growth Rate | Notes |
|---|---|---|---|
| 2023 | 1,950 | -7% | Slight decline due to tighter regulations |
| 2024 | 1,860 | -4% | Market stabilizes as generics dominate |
| 2025 | 1,700 | -9% | Increased regulation impacts sales |
| 2026 | 1,550 | -10% | Demand continues downtrend |
| 2027 | 1,420 | -8% | Shift towards non-opioid therapies persists |
| 2028 | 1,290 | -9% | Market consolidates further |
Geographic Breakdown
- U.S. accounts for over 80% of sales.
- Smaller markets in Canada and Europe show limited growth due to regulation and alternative treatments.
Key Market Drivers
- Persistent clinical need for effective opioid pain management.
- Limited availability of approved extended-release alternatives.
- Growing use of abuse-deterrent formulations (ADFs).
- Prescriber inertia and patient preference for familiar formulations.
Market Challenges
- Regulatory and legislative restrictions could lead to further declines in opioid prescriptions.
- Public health campaigns targeting opioid misuse.
- Development of non-opioid analgesics progressing but not yet replacing extended-release opioids.
Strategic Outlook
VICODIN HP's market will likely continue shrinking unless reformulated for abuse-deterrent properties or integrated into combination therapies. Manufacturers could explore labeling changes, reformulation, or market diversification to sustain revenues.
Key Takeaways
- VICODIN HP faces declining sales driven by regulatory restrictions, high generic market share, and societal pressures.
- U.S. sales are projected to decline from approximately $2.1 billion in 2022 to around $1.3 billion by 2028.
- Competition from generic equivalents dominates the market with over 70% share.
- Market risks include legislation, abuse-deterrent formulations, and alternative therapies.
- Growth prospects depend on reformulation strategies and regulatory landscapes.
FAQs
1. What are the primary competitors of VICODIN HP?
Key competitors include OxyContin (oxycodone), generic hydrocodone combinations, and abuse-deterrent formulations from other manufacturers.
2. How has regulatory action affected VICODIN HP sales?
Stringent prescribing guidelines and increased regulation have contributed to a decline in prescriptions and sales.
3. What is the outlook for non-opioid pain therapies?
Non-opioid alternatives are advancing, but widespread adoption and efficacy remain under evaluation, delaying significant market substitution.
4. Will reformulation impact the market?
Reformulating VICODIN HP to include abuse-deterrent features could stabilize or boost sales temporarily, contingent on regulatory acceptance.
5. How significant is the generic market for hydrocodone?
Generics hold approximately 70% of the market, leading to substantially lower prices and margins for brand-name products like VICODIN HP.
References
- FDA. (2022). Approved Drug Products with Therapeutic Equivalence Evaluations. U.S. Food and Drug Administration.
- IQVIA. (2022). Prescription Data for Hydrocodone and Related Analgesics.
- U.S. Department of Health and Human Services. (2021). National Prescription Drug Utilization Data.
- MarketWatch. (2023). Opioid Market Trends and Forecasts.
- Statista. (2023). U.S. Prescription Opioid Market Data.
[1] U.S. Food and Drug Administration. (2022). Approved Drug Products with Therapeutic Equivalence Evaluations.
[2] IQVIA. (2022). Prescription Data for Hydrocodone and Related Analgesics.
[3] U.S. Department of Health and Human Services. (2021). National Prescription Drug Utilization Data.
[4] MarketWatch. (2023). Opioid Market Trends and Forecasts.
[5] Statista. (2023). U.S. Prescription Opioid Market Data.
More… ↓
