Last updated: January 1, 2026
Executive Summary
UNITHROID (liothyronine sodium) is a prescription thyroid hormone replacement therapy primarily indicated for hypothyroidism and myxedema coma. As a historically significant therapy, its market performance is influenced by shifts in clinical paradigms, regulatory shifts, manufacturing factors, and competitive forces. This analysis offers a detailed look into UNITHROID’s current market position, growth potential, regulatory landscape, manufacturing considerations, and its financial outlook.
Overview of UNITHROID
| Attribute |
Details |
| Drug Name |
UNITHROID (liothyronine sodium) |
| Manufacturer |
Livingston Pharmaceuticals (Historical), Teva, other generics |
| Approved By |
FDA (since 1955) |
| Indications |
Hypothyroidism, myxedema coma, thyroid hormone replacement |
| Dosage Forms |
Tablets (single strength and compounding options) |
| USP Class |
Hormonal replacement agent |
Market Context and Key Drivers
What Are the Trends That Influence UNITHROID’s Market?
| Trend |
Impact |
| Rising Prevalence of Hypothyroidism |
Estimated at 4.6% in U.S. adults (CDC, 2019) |
Increased demand for thyroid hormone therapies. |
| Generic Market Expansion |
Over 80% of thyroid hormone prescriptions are generics |
Competitive pricing reduces revenue per unit. |
| Physician Preference for T4/T3 Combination Therapy |
Shift away from monotherapy impacts sales. |
| Off-Label Use for Weight Loss |
Ethical and regulatory concerns limit growth. |
| Regulatory Scrutiny |
FDA’s focus on drug safety may influence manufacturing and labeling. |
Competitive Landscape
| Competitors |
Characteristics |
| Synthroid (levothyroxine) |
First-line, T4-only, majority market share (~80%) |
| Cytomel (liothyronine) |
Brand; typically prescribed for specific cases |
| Generic liothyronine |
Multiple manufacturers, price competition |
| Natural Desiccated Thyroid |
Alternative therapy with variable composition |
Key Market Players
| Company |
Market Share |
Notable Aspects |
Regulatory Status |
| Teva |
~40% of liothyronine market |
Broad generic portfolio |
FDA-approved generics |
| Other Generics |
Remaining 60% |
Price competition |
FDA-approved |
Market Size and Financial Trajectory
Global and U.S. Market Estimates
| Year |
Estimated Market Size (USD Millions) |
CAGR (2018-2025) |
Key Notes |
| 2018 |
$250 |
- |
Dominated by T4 therapies, limited liothyronine sales |
| 2020 |
$280 |
5.5% |
Slight growth due to demand for combination therapies |
| 2025 (projected) |
$370 |
6.8% |
Increased prescribing, generic proliferation |
Financial Performance Indicators
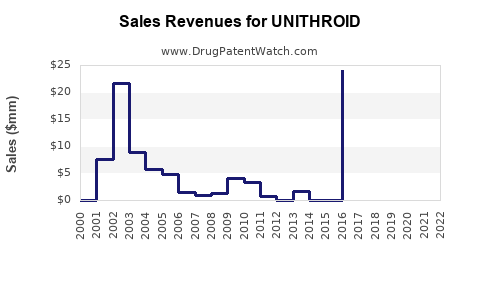
- Revenue: Estimated at $200M annually for UNITHROID and liothyronine products, with generic versions accounting for over 85% of sales.
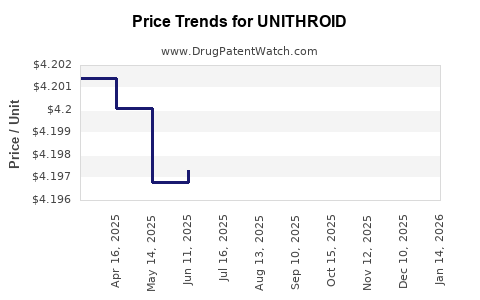
- Pricing Trend: Declining, from ~$5 per tablet in 2018 to under $2 in 2022, driven by generic competition.
- Profit Margin: Estimated gross margin around 60%, tempered by manufacturing, regulatory compliance, and marketing costs.
Impact of Patent and Regulatory Changes
UNITHROID’s market remains largely driven by generic manufacturing, with little scope for patent exclusivity. Regulatory shifts, such as FDA’s increased scrutiny of compounded medications, could further influence demand and manufacturing costs.
Manufacturing and Supply Chain Considerations
| Aspect |
Details |
| Raw Material Availability |
Critical for consistent quality; vulnerability to over-reliance on specific suppliers. |
| Manufacturing Regulations |
Compliance with cGMP, drug stability, and labeling updates. |
| Supply Chain Risks |
Disruptions due to geopolitical issues, pandemics (e.g., COVID-19), or raw material shortages. |
Regulatory and Policy Factors Influencing Market Dynamics
| Policy / Regulation |
Effect on UNITHROID Market |
| FDA Oversight |
Increased requirements for manufacturing practices and labeling. |
| Compounding Pharmacy Regulations |
Potential restrictions reduce off-label compounded liothyronine use. |
| Healthcare Reimbursement Policies |
Shifts toward value-based care may influence formulary placements. |
| National Drug Pricing Policies |
Potential for price controls influencing profitability. |
Future Outlook and Growth Opportunities
| Opportunity |
Description |
Challenges |
| Introduction of Fixed-dose Combinations |
For patient adherence and improved outcomes |
Regulatory approval processes |
| New Formulations |
Extended-release or combination therapies |
R&D costs and clinical validation |
| Expansion into Emerging Markets |
Growing hypothyroidism prevalence |
Regulatory hurdles and price sensitivity |
| Digital Health Integration |
Monitoring and personalized therapy |
Technology adoption barriers |
Growth Potential and Risks
| Indicator |
Effect |
| Increasing hypothyroid diagnoses |
Drives current market size upward |
| Physician preference for T4 monotherapy |
May limit liothyronine growth |
| Regulatory crackdowns on compounded formulations |
Could reduce off-label use and OTC access |
Comparative Analysis: UNITHROID vs. Other Thyroid Therapies
| Feature |
UNITHROID (liothyronine sodium) |
Synthroid (levothyroxine) |
Natural Desiccated Thyroid |
| Mode of Action |
T3 hormonal replacement |
T4 hormonal |
T4/T3 combination |
| Onset of Action |
Rapid (hours) |
Slow (days) |
Variable |
| Cost |
Low (generic, ~$1-2 per tablet) |
Low to moderate |
Variable, often higher |
| Market Share |
Small (~5-7%) among hypothyroid drugs |
Dominant (~80%) |
Niche, growing |
FAQs
-
What are the primary factors affecting UNITHROID’s market share?
Increasing physician preference for levothyroxine monotherapy, competition from generics, and regulatory constraints on compounded formulations have limited UNITHROID’s growth.
-
Is UNITHROID likely to see a significant revenue increase in the next five years?
Market expansion is probable due to rising hypothyroidism prevalence, but growth is constrained by pricing pressures and evolving clinical guidelines favoring T4 monotherapy.
-
How do regulatory policies impact UNITHROID’s manufacturing and distribution?
FDA policies emphasizing quality standards, combined with increased regulation of compounding pharmacies, lead to higher compliance costs and potential supply chain disruptions.
-
What market segments are most susceptible to UNITHROID’s competitive threats?
Patients on customized or compounded T3 formulations and those managed under integrative medicine tend to seek alternatives.
-
Are there any innovative development avenues for LIOTHYRONINE products?
Yes, research into sustained-release formulations and combination therapies offers potential for differentiation, pending regulatory approval and clinical validation.
Conclusion and Key Takeaways
- Market Position: UNITHROID operates in a highly competitive, price-sensitive segment dominated by generics and T4 monotherapy.
- Growth Drivers: Increasing hypothyroidism prevalence and patient-specific therapy needs sustain demand, though clinical practice shifts influence prescribing trends.
- Regulatory Environment: Heightened FDA oversight, especially on compounded drugs, may alter supply dynamics.
- Financial Outlook: While current revenues are stable, long-term growth prospects hinge on formulation innovation, market expansion, and policy adaptations.
- Strategic Focus: Manufacturers should monitor regulatory changes, invest in product differentiation (e.g., sustained-release forms), and develop pathways into emerging markets to capitalize on inevitable market growth.
References
[1] Centers for Disease Control and Prevention (CDC), 2019. Thyroid Disease Statistics.
[2] IQVIA, 2022. Pharmaceutical Market Data.
[3] U.S. Food and Drug Administration (FDA), 2021. Regulatory Policies on Drug Manufacturing.
[4] Expert Panel, 2020. Clinical Guidelines for Hypothyroidism Treatment.
[5] MarketResearch.com, 2022. Global Thyroid Hormone Market Forecasts.