TURALIO Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Turalio, and what generic alternatives are available?
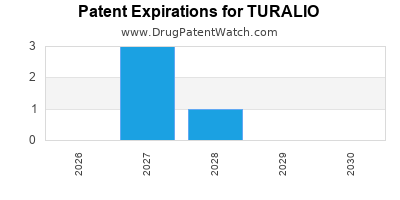
Turalio is a drug marketed by Daiichi Sankyo Inc and is included in one NDA. There are twelve patents protecting this drug.
This drug has one hundred and thirty-eight patent family members in forty-two countries.
The generic ingredient in TURALIO is pexidartinib hydrochloride. One supplier is listed for this compound. Additional details are available on the pexidartinib hydrochloride profile page.
DrugPatentWatch® Generic Entry Outlook for Turalio
Turalio was eligible for patent challenges on August 2, 2023.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be May 5, 2036. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for TURALIO?
- What are the global sales for TURALIO?
- What is Average Wholesale Price for TURALIO?
Summary for TURALIO
| International Patents: | 138 |
| US Patents: | 12 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 66 |
| Clinical Trials: | 1 |
| Patent Applications: | 1,603 |
| Drug Prices: | Drug price information for TURALIO |
| What excipients (inactive ingredients) are in TURALIO? | TURALIO excipients list |
| DailyMed Link: | TURALIO at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for TURALIO
Generic Entry Date for TURALIO*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
CAPSULE;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for TURALIO
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| National Cancer Institute (NCI) | Phase 1/Phase 2 |
Pharmacology for TURALIO
US Patents and Regulatory Information for TURALIO
TURALIO is protected by twelve US patents and two FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of TURALIO is ⤷ Start Trial.
This potential generic entry date is based on patent ⤷ Start Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Daiichi Sankyo Inc | TURALIO | pexidartinib hydrochloride | CAPSULE;ORAL | 211810-002 | Oct 14, 2022 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Daiichi Sankyo Inc | TURALIO | pexidartinib hydrochloride | CAPSULE;ORAL | 211810-001 | Aug 2, 2019 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Daiichi Sankyo Inc | TURALIO | pexidartinib hydrochloride | CAPSULE;ORAL | 211810-001 | Aug 2, 2019 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Daiichi Sankyo Inc | TURALIO | pexidartinib hydrochloride | CAPSULE;ORAL | 211810-002 | Oct 14, 2022 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for TURALIO
When does loss-of-exclusivity occur for TURALIO?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 16258027
Patent: Solid forms of a compound modulating kinases
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2017023490
Patent: formas sólidas de um composto modulando quinases
Estimated Expiration: ⤷ Start Trial
Patent: 2017023540
Patent: síntese de derivados de 1 h-pirrolo[2,3-b]piridin que modulam quinases
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 84899
Patent: SYNTHESE DE DERIVES 1H-PYRROLO[2,3-B]PYRIDINE QUI MODULENT LES KINASES (SYNTHESIS OF 1 H-PYRROLO[2,3-B]PYRIDIN DERIVATIVES THAT MODULATE KINASES)
Estimated Expiration: ⤷ Start Trial
Patent: 84910
Patent: FORMES SOLIDES D'UN COMPOSE MODULANT LES KINASES (SOLID FORMS OF A COMPOUND MODULATING KINASES)
Estimated Expiration: ⤷ Start Trial
China
Patent: 7531706
Patent: 调节激酶的1H‑吡咯并[2,3‑b]吡啶衍生物的合成 (Synthesis of 1H-pyrrolo[2,3-b]pyridin derivatives modulating kinases)
Estimated Expiration: ⤷ Start Trial
Patent: 7548394
Patent: SOLID FORMS OF A COMPOUND MODULATING KINASES
Estimated Expiration: ⤷ Start Trial
Patent: 2574196
Patent: 调节激酶的1H-吡咯并[2,3-b]吡啶衍生物的合成 (Synthesis of 1H-pyrrolo[2,3-b]pyridin derivatives that modulate kinases)
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 17011534
Patent: Formas sólidas de quinasas que modulan un compuesto
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0201383
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 23796
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 92123
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 92122
Patent: SYNTHÈSE DE DÉRIVÉS 1H-PYRROLO[2,3-B]PYRIDINE QUI MODULENT LES KINASES (SYNTHESIS OF 1H-PYRROLO[2,3-B]PYRIDIN DERIVATIVES THAT MODULATE KINASES)
Estimated Expiration: ⤷ Start Trial
Patent: 92123
Patent: FORMES SOLIDES D'UN COMPOSÉ MODULANT LES KINASES (SOLID FORMS OF A COMPOUND MODULATING KINASES)
Estimated Expiration: ⤷ Start Trial
Patent: 81573
Patent: SYNTHÈSE D'UN COMPOSÉ MODULANT DES KINASES (SYNTHESIS OF A COMPOUND THAT MODULATES KINASES)
Estimated Expiration: ⤷ Start Trial
Patent: 57104
Patent: FORMES SOLIDES D'UN COMPOSÉ MODULANT LES KINASES (SOLID FORMS OF A COMPOUND MODULATING KINASES)
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 48675
Patent: 調節激酶的1H 吡咯並[2,3-B]吡啶衍生物的合成 (SYNTHESIS OF 1 H-PYRROLO[2,3-B]PYRIDIN DERIVATIVES THAT MODULATE KINASES)
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 47657
Estimated Expiration: ⤷ Start Trial
Patent: 50506
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 5305
Patent: סינתיזה של תולדות h1-פירולו[3,2-b]פירידין המאפננות קינאזים (Synthesis of 1 h-pyrrolo[2,3-b]pyridin derivatives that modulate kinases)
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 30826
Estimated Expiration: ⤷ Start Trial
Patent: 46194
Estimated Expiration: ⤷ Start Trial
Patent: 86045
Estimated Expiration: ⤷ Start Trial
Patent: 36721
Estimated Expiration: ⤷ Start Trial
Patent: 18515490
Patent: キナーゼを調節する化合物の固体形態
Estimated Expiration: ⤷ Start Trial
Patent: 18516880
Patent: キナーゼを修飾する1H−ピロロ[2,3−B]ピリジン誘導体の合成
Estimated Expiration: ⤷ Start Trial
Patent: 19147828
Patent: キナーゼを修飾する1H−ピロロ[2,3−B]ピリジン誘導体の合成 (SYNTHESIS OF 1H-PYRROLO[2,3-B]PYRIDINE DERIVATIVE MODIFYING KINASE)
Estimated Expiration: ⤷ Start Trial
Patent: 21185163
Patent: キナーゼを修飾する1H−ピロロ[2,3−B]ピリジン誘導体の合成 (SYNTHESIS OF 1 H-PYRROLO[2,3-B]PYRIDIN DERIVATIVES THAT MODULATE KINASES)
Estimated Expiration: ⤷ Start Trial
Patent: 24056918
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 92123
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 2352
Patent: SOLID FORMS OF A COMPOUND MODULATING KINASES
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 6963
Estimated Expiration: ⤷ Start Trial
Patent: 17013975
Patent: FORMAS SOLIDAS DE QUINASAS QUE MODULAN UN COMPUESTO. (SOLID FORMS OF A COMPOUND MODULATING KINASES.)
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 7616
Patent: Solid forms of a compound modulating kinases
Estimated Expiration: ⤷ Start Trial
Philippines
Patent: 017501995
Patent: SOLID FORMS OF A COMPOUND MODULATING KINASES
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 92123
Estimated Expiration: ⤷ Start Trial
Russian Federation
Patent: 30506
Patent: ТВЕРДЫЕ ФОРМЫ СОЕДИНЕНИЯ, МОДУЛИРУЮЩЕГО КИНАЗЫ (SOLID FORMS OF THE COMPOUND MODULATING THE KINASES)
Estimated Expiration: ⤷ Start Trial
Patent: 17141035
Patent: ТВЕРДЫЕ ФОРМЫ СОЕДИНЕНИЯ, МОДУЛИРУЮЩЕГО КИНАЗЫ
Estimated Expiration: ⤷ Start Trial
San Marino
Patent: 02000535
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 731
Patent: ČVRSTI OBLICI JEDINJENJA KOJE MODULIRA KINAZE (SOLID FORMS OF A COMPOUND MODULATING KINASES)
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 201912355T
Patent: SOLID FORMS OF A COMPOUND MODULATING KINASES
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 92123
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1708007
Patent: SOLID FORMS OF A COMPOUND MODULATING KINASES
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 2630889
Estimated Expiration: ⤷ Start Trial
Patent: 2637844
Estimated Expiration: ⤷ Start Trial
Patent: 180002741
Patent: 키나제를 조절하는 화합물의 고형 형태
Estimated Expiration: ⤷ Start Trial
Patent: 180015142
Patent: 키나아제를 조절하는 1H-피롤로[2,3B]피리딘 유도체의 합성
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 74178
Estimated Expiration: ⤷ Start Trial
Patent: 20827
Estimated Expiration: ⤷ Start Trial
Patent: 23943
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1702244
Patent: Solid forms of a compound modulating kinases
Estimated Expiration: ⤷ Start Trial
Patent: 1706271
Patent: Synthesis of a compound that modulates kinases
Estimated Expiration: ⤷ Start Trial
Patent: 60956
Estimated Expiration: ⤷ Start Trial
Patent: 27949
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering TURALIO around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| World Intellectual Property Organization (WIPO) | 2016179415 | ⤷ Start Trial | |
| Norway | 342175 | ⤷ Start Trial | |
| South Korea | 102615829 | ⤷ Start Trial | |
| Japan | 6530826 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Market Dynamics and Financial Trajectory for TURALIO (pexidartinib)
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.