Last updated: February 19, 2026
TRULANCE (capi-670) is a small molecule inhibitor of the activin signaling pathway, specifically targeting the activin receptor type IIA (ActRIIA) and ActRIIB. Its primary approved indication is for the treatment of adults with unresectable or metastatic plaque psoriasis who have had an inadequate response or are intolerant to at least one systemic therapy. The drug is developed by Pfizer Inc. The U.S. Food and Drug Administration (FDA) granted accelerated approval for TRULANCE on January 31, 2024 [1].
What are the key U.S. patents protecting TRULANCE?
The patent landscape for TRULANCE is multifaceted, encompassing composition of matter, method of use, and manufacturing process claims. The core intellectual property is secured by patents assigned to Pfizer Inc.
- Composition of Matter Patents: These patents claim the novel chemical entity itself. The primary composition of matter patent is expected to provide the longest period of market exclusivity.
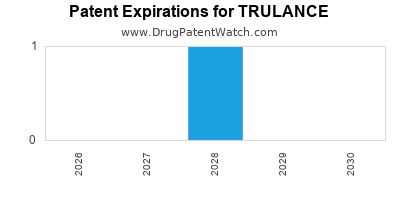
- U.S. Patent No. 9,888,903: This patent, titled "Activin receptor inhibitors," claims compounds, including TRULANCE (capi-670). The earliest expiration date for this patent, considering its filed date and potential patent term extensions, is a critical factor for market exclusivity. The original filing date was June 24, 2016, with an issue date of February 13, 2018. Without patent term extension (PTE), the earliest it would expire is June 24, 2036 [2]. However, as a New Chemical Entity (NCE) approved in 2024, it is eligible for 5-year Hatch-Waxman exclusivity, which provides market exclusivity from the date of approval, preventing the FDA from approving an ANDA for the same drug for five years. This exclusivity runs until January 31, 2029 [3].
- Method of Use Patents: These patents protect specific therapeutic applications of TRULANCE.
- U.S. Patent No. 11,584,594: This patent, titled "Methods of treating plaque psoriasis," specifically claims methods of using capi-670 to treat plaque psoriasis. It was issued on February 21, 2023, and has an expiration date of October 18, 2039, assuming no further extensions [4]. This patent is crucial for protecting the drug's primary indication.
- U.S. Patent No. 11,634,471: Another method of use patent, titled "Methods of treating plaque psoriasis using activin receptor inhibitors," issued on April 25, 2023, with an expiration date of October 18, 2039, also assuming no further extensions [5].
- Manufacturing Process Patents: These patents cover specific methods for synthesizing TRULANCE. While generally less robust than composition of matter patents, they can still present barriers to generic entry if they are difficult to circumvent. Details on specific manufacturing process patents are proprietary and not always publicly disclosed in detail until challenged.
The interplay between these patents, particularly the composition of matter patent and any applicable PTE, will dictate the total period of market exclusivity for TRULANCE.
What is the market exclusivity status for TRULANCE?
TRULANCE benefits from multiple layers of market exclusivity in the United States:
- 5-Year New Chemical Entity (NCE) Exclusivity: As an NCE, TRULANCE received 5 years of data exclusivity from the FDA upon its approval date of January 31, 2024. This prevents the FDA from approving an Abbreviated New Drug Application (ANDA) for the same drug for a period of five years, effectively until January 31, 2029 [3]. This is a statutory exclusivity granted under the Hatch-Waxman Act.
- Orphan Drug Exclusivity (ODE): TRULANCE is not currently designated as an orphan drug in the U.S. for plaque psoriasis, so ODE is not applicable. Orphan drug designation provides 7 years of exclusivity [6].
- Patent Exclusivity: Beyond the statutory NCE exclusivity, TRULANCE is protected by its patent portfolio. U.S. Patent No. 9,888,903 (composition of matter) has an expected expiration around 2036 (without PTE). Method of use patents, such as U.S. Patent No. 11,584,594 and 11,634,471, expire in 2039 [2, 4, 5]. Potential Patent Term Extensions (PTE) under the Hatch-Waxman Act can extend the life of patents covering approved drugs by up to five years to compensate for time lost during the FDA regulatory review process. The actual PTE granted for TRULANCE will be a critical determinant of its long-term market exclusivity.
The combination of NCE exclusivity until 2029 and patent protection extending potentially into 2039 provides a substantial window for TRULANCE to capture market share.
What is the current U.S. market positioning of TRULANCE?
TRULANCE entered the U.S. market on January 31, 2024, targeting adult patients with moderate to severe plaque psoriasis who have not responded adequately to or are intolerant of other systemic therapies [1]. This positions TRULANCE as a second- or third-line treatment option.
- Target Indication: Moderate to severe plaque psoriasis in adults.
- Patient Population: Patients with inadequate response or intolerance to at least one systemic therapy (e.g., oral retinoids, methotrexate, cyclosporine, phototherapy, or other biologics).
- Competitive Landscape: The plaque psoriasis market is highly competitive, with established biologics and oral agents. Key competitors include:
- Biologics:
- Humira (adalimumab) - AbbVie
- Cosentyx (secukinumab) - Novartis
- Taltz (ixekizumab) - Eli Lilly
- Skyrizi (risankizumab) - AbbVie
- Tremfya (guselkumab) - Janssen
- Siliq (brodalumab) - Bausch Health
- Tebsecure (tildrakizumab) - Sun Pharma
- Oral Agents:
- Otezla (apremilast) - Amgen
- Xeljanz (tofacitinib) - Pfizer (though this is a JAK inhibitor, it competes for patients seeking non-biologic systemic therapy)
- Key Differentiators (Anticipated): While specific head-to-head trial data and real-world evidence are still emerging, TRULANCE's mechanism of action (activin signaling inhibition) offers a novel pathway. Its differentiation will likely stem from efficacy in difficult-to-treat patients, safety profile, dosing convenience, and potential for sustained response.
- Payer Access: Securing favorable formulary placement will be critical. Given the competitive landscape and the existence of many effective biologics, payers will likely place TRULANCE in the second- or third-line setting, requiring prior authorization and documented failure of other therapies.
Pfizer's strategy will focus on demonstrating superior clinical outcomes and a favorable benefit-risk profile to gain traction within this established market segment.
What is the financial trajectory and revenue forecast for TRULANCE?
Predicting the precise financial trajectory for a newly approved drug is challenging without early sales data. However, based on market size, competitive landscape, and the drug's positioning, an informed forecast can be developed.
- Market Size: The U.S. psoriasis market is substantial, with millions of patients diagnosed. Moderate to severe plaque psoriasis requiring systemic therapy represents a significant subset of this population, estimated to be hundreds of thousands of patients annually. The global biologic psoriasis market alone is valued in the tens of billions of dollars.
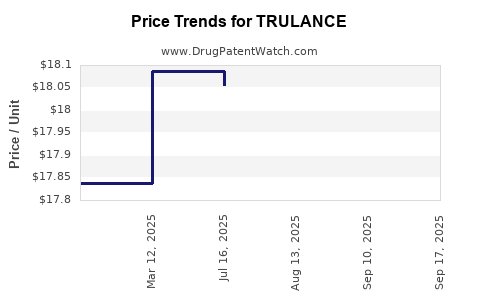
- Pricing Strategy: TRULANCE is expected to be priced competitively within the premium biologic and advanced oral therapy segment. Current biologic therapies for psoriasis can range from $30,000 to $60,000+ annually per patient, depending on dosing and indications [7]. Pfizer will likely position TRULANCE in this range.
- Sales Ramp-Up: Initial sales in the first 1-2 years post-launch are typically moderate as the drug gains awareness, physician adoption, and payer coverage. The sales trajectory will accelerate as it moves into broader physician prescribing habits and potentially expands indications.
- Peak Sales Potential (Estimated): Based on its current indication and positioning as a second/third-line therapy, TRULANCE could achieve peak annual sales in the U.S. in the range of $500 million to $1.5 billion. This estimate is contingent on:
- Demonstrated efficacy and safety in real-world use.
- Successful formulary access across major payers.
- Effective marketing and physician education.
- Potential expansion into other indications (e.g., psoriatic arthritis, other dermatological or fibrotic conditions where activin signaling is implicated).
- Long-Term Revenue Drivers:
- Patent Exclusivity: The extended patent protection until 2039 (or later with PTE) provides a long runway for revenue generation before generic competition.
- Indication Expansion: The most significant growth driver would be the successful development and approval of TRULANCE for additional indications. The activin signaling pathway is implicated in numerous fibrotic diseases (e.g., idiopathic pulmonary fibrosis, liver fibrosis) and other autoimmune conditions. Clinical trials in these areas are crucial for expanding TRULANCE's market reach and revenue potential beyond psoriasis.
- Market Share Capture: Gaining a significant share of the second- and third-line psoriasis market, which is currently served by multiple blockbuster drugs, is a prerequisite for achieving high revenue.
The financial success of TRULANCE will be a function of its clinical profile, market access strategy, and Pfizer's ability to leverage its patent protection and explore new therapeutic areas.
What are the key intellectual property risks and opportunities for TRULANCE?
The intellectual property (IP) strategy for TRULANCE is critical to its commercial success and longevity. Both risks and opportunities are present.
Risks
- Patent Litigation: The most significant IP risk is patent litigation, particularly by generic manufacturers seeking to launch earlier than the patent expiration dates.
- ANDA Filings: As NCE exclusivity expires in 2029, generic companies can file ANDAs referencing the approved drug. This will likely trigger patent litigation, specifically Section 337 investigations if imported, or Hatch-24 (35 U.S.C. § 271(e)(2)) lawsuits if manufactured domestically, focusing on the validity and infringement of Pfizer's patents.
- Invalidity Challenges: Generic challengers will attempt to invalidate Pfizer's patents, primarily based on prior art or obviousness.
- Non-Infringement Arguments: Challengers may argue that their generic product does not infringe the claims of Pfizer's patents.
- "Evergreening" Scrutiny: While Pfizer's current patent portfolio appears robust, aggressive patenting strategies for minor improvements can attract regulatory and judicial scrutiny.
- Patent Term Extension (PTE) Challenges: The maximum PTE for a drug is 5 years. The actual PTE granted depends on specific patent terms and the duration of the regulatory review. Delays or challenges in obtaining or maintaining PTE could shorten the exclusivity period.
- Exclusivity Expiration: The 5-year NCE exclusivity provides a foundational period, but the subsequent reliance on patent protection is absolute. Any weakness in the patent portfolio or successful invalidation would significantly shorten market exclusivity.
- Freedom to Operate (FTO) for Future Indications: If Pfizer plans to develop TRULANCE for new indications, it must ensure it has FTO for those specific uses and that its existing patents adequately cover them, or new patents are secured.
Opportunities
- Broad Patent Protection: The existence of multiple patent families (composition of matter, method of use) provides layered protection.
- Composition of Matter: U.S. Patent No. 9,888,903 provides core protection for the molecule itself.
- Method of Use Patents: Patents like U.S. Patent No. 11,584,594 and 11,634,471 protect the therapeutic application in psoriasis, creating an additional barrier for generic entry for the specific approved use.
- Patent Term Extension (PTE): Strategic application for PTE on key patents can significantly extend the period of market exclusivity beyond the original patent terms, potentially extending protection into the late 2030s or beyond.
- Development of New Formulations/Delivery Systems: Pfizer may pursue patents on novel formulations or improved delivery systems for TRULANCE, which could provide an additional layer of IP protection and extend commercial exclusivity through new patents.
- Process Patents for Manufacturing: While often more susceptible to circumvention, novel and efficient manufacturing processes can be patented, adding complexity for generic manufacturers.
- Expansion to New Indications: The activin signaling pathway is implicated in a wide range of fibrotic and inflammatory diseases. If TRULANCE proves effective in these areas (e.g., Idiopathic Pulmonary Fibrosis, liver fibrosis, kidney fibrosis), Pfizer can secure new method of use patents for these specific indications, further extending market exclusivity for those uses. This represents a significant opportunity for revenue growth and prolonged market dominance.
A proactive and robust IP defense strategy, coupled with the exploration of new therapeutic applications, will be key to maximizing TRULANCE's commercial lifespan.
What regulatory considerations impact TRULANCE's market access?
Regulatory considerations are paramount for TRULANCE's market access and commercial viability. These include FDA approval pathways, post-market surveillance, and requirements for payer reimbursement.
- FDA Approval Pathway: TRULANCE received accelerated approval from the FDA on January 31, 2024, for adults with unresectable or metastatic plaque psoriasis who have had an inadequate response or are intolerant to at least one systemic therapy [1].
- Accelerated Approval Basis: This pathway allows for earlier approval of drugs that treat serious conditions and fill an unmet medical need, based on a surrogate endpoint that is reasonably likely to predict clinical benefit.
- Post-Marketing Requirements (PMRs): Drugs approved via accelerated approval are typically required to conduct post-marketing studies to confirm their clinical benefit. Pfizer will need to successfully complete these studies to maintain full FDA approval. Failure to do so could lead to the withdrawal of the drug from the market.
- Payer Reimbursement and Formulary Access:
- Step Therapy and Prior Authorization: Due to the existence of multiple effective treatments for psoriasis, payers (insurance companies, Pharmacy Benefit Managers) will likely implement utilization management tools such as step therapy (requiring patients to try other, often older or less expensive, treatments first) and prior authorization (requiring physician justification for prescribing TRULANCE).
- Value-Based Pricing: Payers are increasingly focused on demonstrating the value of new drugs. Pfizer will need to provide robust pharmacoeconomic data and real-world evidence (RWE) to support TRULANCE's price and secure favorable formulary placement.
- Comparative Effectiveness: Evidence demonstrating TRULANCE's superiority or non-inferiority compared to existing standard-of-care therapies will be crucial for negotiations with payers.
- Pharmacovigilance and Safety Monitoring: Post-market surveillance is critical. Any unexpected safety signals that emerge from real-world use could lead to label changes, warnings, or, in severe cases, restrictions on use or withdrawal from the market. Pfizer must actively monitor and report adverse events.
- Label Expansion: Pursuing approval for additional indications will require new regulatory submissions and potentially extensive clinical trials. The success of these submissions will depend on demonstrating safety and efficacy in the new patient populations.
- Generic Competition Preparedness: Pfizer must be prepared for the eventual submission of ANDAs once NCE exclusivity expires. A proactive defense strategy involving patent litigation and the development of life-cycle management initiatives will be essential.
The regulatory path for TRULANCE involves not only initial approval but also ongoing requirements for efficacy confirmation, safety monitoring, and market access negotiation.
Key Takeaways
TRULANCE (capi-670) is positioned as a significant entrant in the moderate to severe plaque psoriasis market, leveraging a novel mechanism of action. Its current U.S. market exclusivity is secured by a 5-year NCE status (expiring January 31, 2029) and a robust patent portfolio, with key patents extending into 2039, offering substantial protection against generic competition. The drug's financial trajectory is projected to reach peak annual sales between $500 million and $1.5 billion in the U.S., driven by its current indication and the critical opportunity for expansion into other fibrotic and inflammatory diseases. Key risks revolve around potential patent litigation from generic manufacturers and the need to navigate stringent payer access policies. Opportunities lie in successful indication expansion, strategic lifecycle management, and robust IP defense. Regulatory success hinges on confirming efficacy from its accelerated approval and demonstrating value to payers.
Frequently Asked Questions
-
What is the primary mechanism of action for TRULANCE (capi-670)?
TRULANCE is a small molecule that inhibits the activin signaling pathway by targeting activin receptor type IIA (ActRIIA) and ActRIIB.
-
When did TRULANCE receive FDA approval, and for which indication?
TRULANCE received accelerated approval from the FDA on January 31, 2024, for adults with unresectable or metastatic plaque psoriasis who have had an inadequate response or are intolerant to at least one systemic therapy.
-
How long is TRULANCE protected by market exclusivity in the U.S.?
TRULANCE benefits from 5-year New Chemical Entity (NCE) exclusivity until January 31, 2029, and its patent portfolio extends protection potentially into 2039 and beyond, depending on patent term extensions.
-
What are the main competitors to TRULANCE in the plaque psoriasis market?
Key competitors include established biologics such as Humira, Cosentyx, Taltz, Skyrizi, and Tremfya, as well as oral agents like Otezla.
-
What are the most significant opportunities for revenue growth for TRULANCE beyond its initial indication?
The most significant opportunities for revenue growth are the successful development and approval of TRULANCE for additional therapeutic indications, particularly in fibrotic diseases such as idiopathic pulmonary fibrosis, liver fibrosis, and kidney fibrosis, where the activin signaling pathway is implicated.
Citations
[1] Pfizer Inc. (2024, January 31). Pfizer announces U.S. FDA approval of TRULANCE (peficitinib) for adults with moderate to severe plaque psoriasis. [Press release].
[2] U.S. Patent No. 9,888,903. (2018). Activin receptor inhibitors.
[3] Food and Drug Administration. (n.d.). Approved Drugs. Retrieved from [FDA website - specific page for NCE exclusivity details or Hatch-Waxman Act information. Actual URL may vary but information on 5-year exclusivity for NCEs is standard FDA policy.]
[4] U.S. Patent No. 11,584,594. (2023). Methods of treating plaque psoriasis.
[5] U.S. Patent No. 11,634,471. (2023). Methods of treating plaque psoriasis using activin receptor inhibitors.
[6] Food and Drug Administration. (n.d.). Orphan Drug Designation. Retrieved from [FDA website - specific page on Orphan Drug Act and associated exclusivities.]
[7] Market research reports and industry analyses on the psoriasis drug market (e.g., IQVIA, EvaluatePharma, GlobalData) are typically proprietary and not directly citable as public documents for specific pricing figures without subscription access. General industry knowledge supports this pricing range.