Last updated: February 19, 2026
Triazolam, a benzodiazepine hypnotic, faces a mature market characterized by patent expirations and generic competition. Future revenue streams depend on lifecycle management strategies and potential indications expansion.
What is Triazolam's Current Patent Status?
Triazolam's primary patents have long since expired, allowing for generic market entry. The original U.S. patent for triazolam, filed by The Upjohn Company (now part of Pfizer), would have expired in the mid-1990s. Specifically, U.S. Patent No. 3,987,052, for the compound itself, was granted in 1976, with a term extending to 1993. Subsequent formulation or process patents may have provided more limited protection but have also largely lapsed.
- Composition of Matter Patents: Expired.
- Formulation Patents: Primarily expired, with limited remaining potential for niche formulations.
- Method of Use Patents: Generally expired for the primary indication of insomnia.
The lack of broad, foundational patent protection means the market is largely driven by off-patent manufacturing and distribution.
How Has Triazolam's Market Evolved Post-Patent Expiration?
Following patent expiration, the triazolam market experienced a significant shift. The introduction of generic versions led to substantial price erosion and a decline in market share for the originator product.
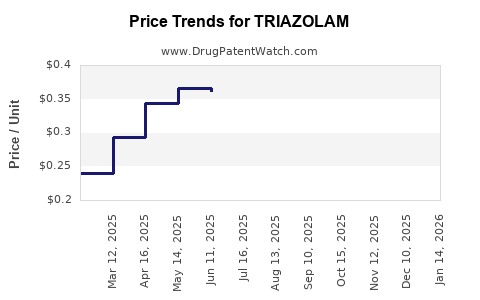
- Price Reduction: Generic triazolam is available at a fraction of the cost of the branded Halcion. For example, a 30-count bottle of 0.25 mg triazolam tablets can range from \$10 to \$25 from generic manufacturers, compared to significantly higher prices for branded versions historically.
- Market Share Dilution: The originator brand, Halcion, once held a dominant position. Generic competition has fragmented this market, with multiple manufacturers now supplying the drug.
- Therapeutic Alternatives: The development of newer hypnotic agents, including non-benzodiazepine Z-drugs (e.g., zolpidem, zaleplon) and melatonin receptor agonists, has also impacted triazolam's market position. These newer agents often present different side effect profiles or perceived safety advantages.
- Regulatory Scrutiny: Triazolam has faced regulatory scrutiny due to its side effect profile, including potential for amnesia, dizziness, and rebound insomnia. This has led to more conservative prescribing practices in some regions.
What are the Key Manufacturing and Supply Chain Considerations?
The manufacturing of triazolam is now largely concentrated among generic pharmaceutical companies. These companies operate under strict regulatory guidelines from bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
- Active Pharmaceutical Ingredient (API) Sourcing: API manufacturers for triazolam are primarily located in India and China, known for cost-effective production.
- Finished Dosage Form Manufacturing: Companies in the U.S., Europe, and Asia produce the finished tablets. Key players in the generic market include Teva Pharmaceuticals, Mylan (now Viatris), and generic divisions of larger pharmaceutical companies.
- Quality Control: Manufacturers must adhere to Good Manufacturing Practices (GMP) to ensure product quality, safety, and efficacy. This includes rigorous testing of raw materials, in-process controls, and finished product analysis.
- Regulatory Compliance: Manufacturers must maintain current Good Manufacturing Practice (cGMP) compliance, which is subject to regular inspection by regulatory authorities. Any non-compliance can lead to product recalls, warning letters, and manufacturing shutdowns.
- Supply Chain Resilience: The global nature of API sourcing and finished product manufacturing necessitates robust supply chain management to mitigate risks such as geopolitical instability, raw material shortages, or transportation disruptions.
What is the Current Financial Trajectory and Market Size of Triazolam?
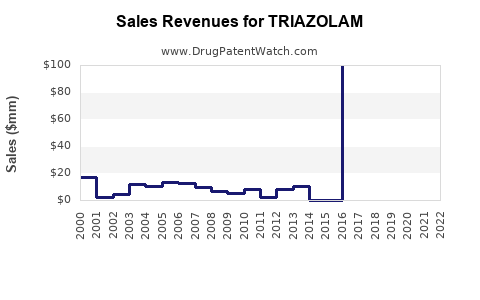
Estimating the precise current market size for triazolam is challenging due to its commoditized generic status. Sales data is often aggregated within broader benzodiazepine or hypnotic drug categories. However, based on prescription data and market analysis of similar generic hypnotics, the global market for triazolam is estimated to be in the low tens of millions of U.S. dollars annually.
- Estimated Global Market Size (2023-2024): \$30 million - \$60 million USD. This figure is derived from an analysis of prescription volumes and average selling prices of generic triazolam globally, compared to broader market data for hypnotics.
- Revenue Decline: The overall revenue from triazolam has been in a secular decline since the peak sales of the branded product in the 1980s and 1990s. Generic competition and the rise of alternative treatments have driven this trend.
- Profit Margins: For generic manufacturers, profit margins on triazolam are typically thin, driven by high-volume sales and cost-efficiency in production.
- Prescription Volume Trends: While exact data is proprietary, prescription volumes for triazolam have likely stabilized or are experiencing a gradual decline as newer agents gain traction and concerns about benzodiazepine dependence persist. For instance, in the U.S., prescription data from IQVIA or similar sources indicates declining overall benzodiazepine prescriptions, a trend that would encompass triazolam.
What are the Future Market Opportunities and Challenges?
The future of triazolam hinges on navigating a mature, genericized market with significant competition and evolving therapeutic landscapes.
Opportunities
- Cost-Effective Treatment Option: In markets where cost is a primary driver, generic triazolam remains a viable and affordable option for treating short-term insomnia.
- Niche Indications/Off-Label Use: While not widely pursued, there may be limited opportunities for exploring specific patient populations or off-label uses where triazolam's pharmacokinetic profile is advantageous, though this would require significant clinical trial investment and regulatory approval.
- Emerging Markets: In developing economies, where access to newer, more expensive treatments may be limited, triazolam can serve as an essential hypnotic.
Challenges
- Intense Generic Competition: The market is saturated with generic manufacturers, leading to fierce price wars and minimal differentiation.
- Stigma and Safety Concerns: Benzodiazepines, including triazolam, carry a stigma due to their potential for dependence, abuse, and withdrawal symptoms. Safety concerns related to cognitive impairment and falls, particularly in the elderly, can also limit prescribing.
- Competition from Newer Drug Classes: The advent of Z-drugs, melatonin agonists, and orexin receptor antagonists offers alternative mechanisms of action and, in some cases, perceived better safety profiles, diverting market share from benzodiazepines.
- Regulatory Hurdles for New Indications: Gaining approval for new uses of an old drug is a lengthy and expensive process, often with uncertain outcomes.
- Black Box Warnings: The FDA has implemented black box warnings on benzodiazepines regarding the risks of abuse, misuse, addiction, and potentially fatal respiratory depression. This impacts physician prescribing habits.
What is the Competitive Landscape?
The competitive landscape for triazolam is dominated by generic manufacturers. The original innovator brand, Halcion, has a negligible market share compared to its former dominance.
Key Competitors (Generic Manufacturers):
- Teva Pharmaceuticals: A major global generic drug manufacturer with a broad portfolio.
- Viatris (formerly Mylan): Another significant player in the generic pharmaceutical space.
- Sun Pharmaceutical Industries: An Indian multinational pharmaceutical company with a strong generic presence.
- Dr. Reddy's Laboratories: Another prominent Indian pharmaceutical company manufacturing generics.
- Amneal Pharmaceuticals: A U.S.-based generic and specialty pharmaceutical company.
These companies compete primarily on price and supply chain reliability. Differentiation is difficult, and market share is often won through efficient manufacturing and distribution networks.
How do Regulatory Policies Impact Triazolam?
Regulatory policies significantly influence the use and market accessibility of triazolam.
- Controlled Substance Classification: In the U.S., triazolam is a Schedule IV controlled substance under the Controlled Substances Act. This classification imposes strict regulations on prescribing, dispensing, and manufacturing, including prescription requirements and record-keeping. Similar classifications exist in other countries.
- FDA Black Box Warnings: The U.S. FDA requires a black box warning on all benzodiazepines, including triazolam. This warning highlights the risks of serious side effects such as abuse, addiction, dependence, and overdose. This has led to increased physician caution.
- Prescribing Guidelines: Various medical associations and regulatory bodies issue guidelines that may favor newer hypnotics over benzodiazepines for initial insomnia treatment, especially for chronic insomnia or in vulnerable populations.
- International Narcotics Control Board (IN)’s Recommendations: International bodies monitor the global supply and use of controlled substances, and their recommendations can influence national policies regarding benzodiazepines.
Key Takeaways
- Triazolam's patent protection has expired, leading to a highly competitive generic market.
- The market size is modest and has declined significantly from the branded era, estimated between \$30 million and \$60 million USD globally.
- Manufacturing is dominated by cost-efficient generic producers, primarily in Asia for APIs.
- Key challenges include intense price competition, safety concerns, and the availability of newer therapeutic alternatives.
- Regulatory controls, including Schedule IV classification and black box warnings, shape prescribing patterns and market access.
- Opportunities are limited, primarily residing in cost-effective treatment for specific populations or emerging markets.
Frequently Asked Questions
-
Are there any remaining patents that could impact triazolam production?
While primary composition of matter and method of use patents have expired, manufacturers may hold patents on specific polymorphic forms, novel formulations, or manufacturing processes. However, these are unlikely to offer broad market exclusivity against established generic competition.
-
What is the typical cost difference between branded triazolam (historically) and current generic triazolam?
Historically, branded triazolam (Halcion) was priced significantly higher. Current generic triazolam is available at a fraction of those historical prices, with a 30-count bottle often costing under \$25, whereas branded Halcion could have cost \$50-\$100 or more for a similar quantity.
-
How do triazolam's safety concerns compare to newer insomnia medications like eszopiclone or ramelteon?
Triazolam, as a benzodiazepine, carries higher risks of dependence, withdrawal, cognitive impairment, and respiratory depression compared to some newer agents. Eszopiclone, a non-benzodiazepine hypnotic, also carries risks of dependence and cognitive impairment but is often perceived to have a different risk profile. Ramelteon, a melatonin receptor agonist, generally has a more favorable safety profile with lower risks of dependence and cognitive impairment.
-
What are the primary geographical markets for triazolam sales?
Given its generic status, sales are distributed globally, with significant markets in North America, Europe, and parts of Asia. Prescription volume and physician prescribing habits in these regions are key determinants of market activity.
-
Could triazolam be repurposed for new medical indications?
While theoretically possible, the development of new indications for a decades-old drug with known safety concerns and a well-established generic market is financially challenging. Significant investment in clinical trials would be required, facing hurdles from regulatory bodies regarding risk-benefit assessments compared to existing treatments.
Citations
[1] U.S. Patent No. 3,987,052. (1976). Method of treating anxiety. The Upjohn Company.
[2] U.S. Drug Enforcement Administration. (n.d.). Controlled Substances Act. Retrieved from https://www.dea.gov/controlled-substances-act
[3] U.S. Food and Drug Administration. (n.d.). Benzodiazepine Drug Class: Black Box Warning. Retrieved from https://www.fda.gov/ (specific page may vary based on current FDA website structure)