Share This Page
Drug Price Trends for TRIAZOLAM
✉ Email this page to a colleague
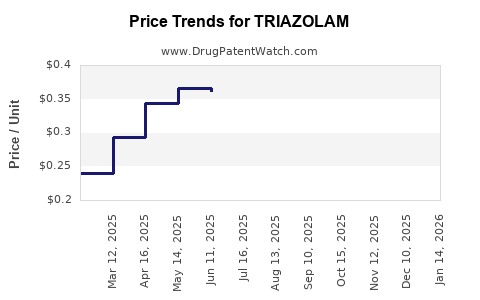
Average Pharmacy Cost for TRIAZOLAM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TRIAZOLAM 0.25 MG TABLET | 50742-0646-01 | 0.36913 | EACH | 2026-04-22 |
| TRIAZOLAM 0.125 MG TABLET | 70710-1521-01 | 0.33429 | EACH | 2026-04-22 |
| TRIAZOLAM 0.25 MG TABLET | 00054-4859-29 | 0.36913 | EACH | 2026-04-22 |
| TRIAZOLAM 0.125 MG TABLET | 00054-4858-25 | 0.33429 | EACH | 2026-04-22 |
| TRIAZOLAM 0.25 MG TABLET | 00054-4859-25 | 0.36913 | EACH | 2026-04-22 |
| TRIAZOLAM 0.125 MG TABLET | 50742-0645-01 | 0.33429 | EACH | 2026-04-22 |
| TRIAZOLAM 0.125 MG TABLET | 59762-3717-09 | 0.33429 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
TRIAZOLAM: PATENT LANDSCAPE AND MARKET FORECAST
Triazolam, a potent benzodiazepine used for short-term treatment of insomnia, faces a mature market characterized by expired primary patents, significant generic competition, and stable but modest demand. The drug's efficacy in managing severe sleep disturbances is offset by concerns regarding dependence and withdrawal, limiting its expansion potential. Price projections indicate continued stability driven by generic pricing structures, with minimal significant growth anticipated in the absence of new therapeutic indications or significant shifts in regulatory landscapes.
What is the current patent status of Triazolam?
The primary patents for Triazolam, originally developed by Upjohn (now Pfizer), have long expired. The compound's initial patent protection, U.S. Patent No. 3,597,445, was granted in 1971 and expired in the early 1990s [1]. This early expiration allowed for the rapid entry of generic manufacturers into the market.
Subsequent patent filings have focused on specific formulations, manufacturing processes, or novel delivery methods. For example, patents might cover extended-release formulations or specific salt forms that aim to improve pharmacokinetic profiles or reduce side effects. However, these secondary patents have generally not provided significant market exclusivity beyond that offered by the original compound patent and have also faced or are approaching expiration.
A review of the U.S. Patent and Trademark Office (USPTO) database and commercial patent databases reveals no active, broad composition-of-matter patents for Triazolam that would prevent generic production. The landscape is dominated by generics, with most innovation centered on process improvements or specific niche formulations that have not fundamentally altered the market exclusivity.
Who are the key generic manufacturers of Triazolam?
The generic Triazolam market is fragmented, with numerous pharmaceutical companies producing and distributing the drug. Major generic manufacturers with active FDA Abbreviated New Drug Applications (ANDAs) for Triazolam include:
- Teva Pharmaceuticals: A leading global generic drug manufacturer with a broad portfolio.
- Perrigo Company plc: Specializes in over-the-counter and generic prescription products.
- Apotex Inc.: A Canadian-based pharmaceutical company with a significant global presence in generics.
- Mylan N.V. (now Viatris): Merged with Pfizer’s Upjohn division, Viatris is a major player in generic and biosimilar medicines.
- Sandoz International GmbH (Novartis): A subsidiary of Novartis, Sandoz is one of the world's largest generic drug manufacturers.
- Lannett Company, Inc.: A U.S.-based manufacturer focused on generic oral solid dosage pharmaceuticals.
These companies compete primarily on price and market access, contributing to the drug's established price point. The availability of multiple bioequivalent generic versions ensures robust competition and limits the pricing power of any single manufacturer.
What is the projected market size and growth rate for Triazolam?
The global market for Triazolam is considered mature, with demand largely dictated by the prevalence of severe insomnia and the availability of alternative treatments. Market size is difficult to quantify precisely due to the proprietary nature of sales data for individual generic drugs and the focus on prescription volume rather than dollar value in many analyses. However, industry reports and market research generally place the global market for benzodiazepines used in sleep disorders in the hundreds of millions of U.S. dollars annually.
Projected Market Growth Rate: The market for Triazolam is expected to remain relatively stagnant, with a compound annual growth rate (CAGR) of approximately 0% to 1% over the next five years.
Factors influencing market size and growth:
- Prevalence of Insomnia: While insomnia is a widespread condition, the specific indications for Triazolam (severe, short-term management) limit its patient population.
- Generic Competition: Intense competition among generic manufacturers drives down prices and limits overall revenue growth.
- Therapeutic Alternatives: The market is saturated with other hypnotic agents, including other benzodiazepines, non-benzodiazepine hypnotics (e.g., zolpidem, eszopiclone), and newer agents with different mechanisms of action (e.g., melatonin receptor agonists, orexin receptor antagonists). These alternatives offer varying safety profiles and efficacy, leading to patient and physician choice.
- Safety Concerns: Triazolam's potential for dependence, withdrawal symptoms, amnesia, and next-day sedation restricts its long-term use and can steer prescribers toward alternatives perceived as safer. Regulatory bodies have also implemented stricter prescribing guidelines for benzodiazepines [2].
- Lack of New Indications: There have been no significant new therapeutic indications approved for Triazolam, limiting opportunities for market expansion.
The demand is expected to be primarily driven by existing prescriptions and patient reliance rather than new patient acquisition.
What are the average wholesale prices (AWP) and projected price trends for Triazolam?
The Average Wholesale Price (AWP) for Triazolam varies significantly based on dosage strength (0.125 mg, 0.25 mg), pill count per bottle (e.g., 30, 100, 500), and the specific generic manufacturer. As of late 2023 and early 2024, typical AWP ranges for a 30-count bottle of 0.25 mg Triazolam are approximately $15 to $50 USD.
Projected Price Trends:
Prices for Triazolam are anticipated to remain stable with slight downward pressure.
- Stability: The established generic market and mature demand create a stable pricing environment. Manufacturers have optimized production, and supply chains are robust.
- Downward Pressure: Ongoing competition among generic suppliers, coupled with pressures from pharmacy benefit managers (PBMs) and payers to reduce healthcare costs, will likely exert slight downward pressure on AWP over the next few years. Price reductions for generics typically occur as more manufacturers enter the market or existing ones engage in competitive bidding.
- Reimbursement Policies: Payer formularies often place Triazolam in lower cost-sharing tiers due to its generic status, further reinforcing price stability.
It is important to note that AWP is a benchmark price and actual contract prices with wholesalers, distributors, and pharmacies will differ. Net prices received by manufacturers will be lower than AWP after rebates and discounts.
What are the primary drivers and challenges for the Triazolam market?
Primary Market Drivers:
- Efficacy in Severe Insomnia: Triazolam remains a highly effective agent for the short-term treatment of severe sleep-onset and sleep-maintenance insomnia when other treatments have failed. Its rapid onset and potent sedative effects are its primary clinical advantages.
- Established Generic Availability: The wide availability of multiple generic manufacturers ensures a consistent supply and competitive pricing, making it accessible within healthcare systems.
- Physician Familiarity: Many physicians are familiar with Triazolam's profile and historical use, leading to continued prescribing, particularly for patients who have responded well to it previously.
- Short-Term Treatment Necessity: For acute episodes of severe insomnia, a short course of a potent agent like Triazolam can be clinically indicated and utilized.
Primary Market Challenges:
- Dependence and Withdrawal: The risk of psychological and physical dependence, along with potentially severe withdrawal symptoms upon discontinuation, is a major deterrent to long-term use and a significant safety concern.
- Adverse Drug Reactions: Side effects such as anterograde amnesia, dizziness, impaired coordination, and next-day drowsiness can limit patient adherence and create safety risks, especially in older adults.
- Regulatory Scrutiny and Prescribing Guidelines: Regulatory agencies worldwide, including the U.S. Food and Drug Administration (FDA), have issued warnings and recommendations for the cautious use of benzodiazepines, including Triazolam, due to abuse potential and safety risks. This has led to more conservative prescribing practices [2].
- Competition from Newer Hypnotics: The development of newer classes of sleep aids with potentially improved safety profiles or different mechanisms of action (e.g., orexin antagonists like suvorexant, dual orexin receptor antagonists like lemborexant, melatonin receptor agonists like ramelteon) offers alternatives that may be preferred by physicians and patients seeking to mitigate benzodiazepine-related risks.
- Off-Label Use Limitations: While benzodiazepines are sometimes used off-label for anxiety or sedation, Triazolam’s specific indication for insomnia and its short half-life make it less suitable for broader indications compared to longer-acting benzodiazepines.
- Black Box Warnings: The existence of FDA black box warnings regarding the risk of abuse, misuse, and addiction, serious respiratory depression, and fetal harm during pregnancy places significant constraints on prescribing and patient selection.
What is the outlook for Triazolam in R&D and investment?
The research and development (R&D) landscape for Triazolam itself is effectively dormant. Given the expiration of primary patents and the presence of a mature generic market, there is little incentive for significant new R&D investment focused on developing novel formulations or expanding its indications. The primary focus for R&D in the insomnia market has shifted towards:
- novel therapeutic targets: Identifying new neurotransmitter systems or pathways involved in sleep regulation, such as the orexin system.
- drugs with improved safety profiles: Developing hypnotics with a lower risk of dependence, cognitive impairment, and residual next-day effects.
- personalized medicine approaches: Tailoring sleep disorder treatment based on individual patient characteristics and genetic predispositions.
For investors, Triazolam represents a low-growth, stable generic product within the broader sleep disorder market. Investment opportunities related to Triazolam would likely be within diversified generic pharmaceutical portfolios rather than standalone ventures.
- Investment Focus: Any investment consideration would center on companies with efficient manufacturing capabilities and strong distribution networks for generic pharmaceuticals. The profitability of such investments is tied to volume sales and cost management rather than market expansion or innovation.
- Risk Profile: The risks associated with Triazolam include increased regulatory scrutiny on benzodiazepines, potential class-wide safety concerns, and continued erosion of market share by newer, potentially safer alternatives.
- Strategic Value: The strategic value of Triazolam for a company is primarily as a portfolio filler, contributing to revenue streams through established market channels rather than as a growth engine.
The market is not attractive for significant new entrants or substantial R&D investment aimed at revitalizing the drug's commercial prospects.
Key Takeaways
- Triazolam's core composition-of-matter patents have expired, leading to a highly competitive generic market.
- The market is dominated by established generic manufacturers such as Teva, Perrigo, and Apotex.
- The global market size for Triazolam is modest, with projected CAGR of 0-1% due to its mature status and limited growth drivers.
- Average Wholesale Prices for a 30-count bottle of 0.25 mg Triazolam range from $15 to $50, with stable to slightly downward price trends expected.
- Key drivers include its efficacy for severe insomnia and broad generic availability, while challenges stem from dependence risks, adverse effects, and competition from newer hypnotics.
- R&D and investment in Triazolam are minimal; focus has shifted to novel sleep disorder mechanisms and safer alternatives. Investment opportunities are limited to generic portfolio diversification.
Frequently Asked Questions
- Can new patents be filed for Triazolam to extend market exclusivity? While patents can be filed for novel formulations, manufacturing processes, or specific combinations, these are typically secondary patents with limited exclusivity and are unlikely to significantly alter the market dynamics or extend protection beyond the original compound patent's lifespan.
- What is the typical prescriber profile for Triazolam in the current market? Prescribers are generally specialists in sleep medicine, psychiatry, or neurology, or primary care physicians treating patients with severe, short-term insomnia that has not responded to other therapies. Prescribing is cautious due to safety warnings.
- How does Triazolam compare in terms of safety profile to newer insomnia medications? Triazolam, as a benzodiazepine, carries a higher risk of dependence, withdrawal, and cognitive impairment compared to some newer classes like orexin receptor antagonists or melatonin receptor agonists, which are generally considered to have more favorable safety profiles for long-term use.
- Are there any specific geographic markets where Triazolam demand is increasing? No significant increases in demand are projected for Triazolam in any major geographic market. Demand is stable globally, reflecting the drug's established therapeutic niche and the universal challenges associated with benzodiazepines.
- What is the typical duration of Triazolam treatment recommended by healthcare providers? Triazolam is recommended for short-term use only, typically ranging from a few days to a maximum of two weeks, to mitigate the risk of dependence and tolerance. Extended use requires careful medical supervision.
Citations
[1] U.S. Patent No. 3,597,445, issued August 3, 1971. [2] U.S. Food and Drug Administration. (2017, February 22). FDA Drug Safety Communication: FDA is warning about a possible new risk of “sleep-driving” and other complex sleep-related behaviors with sedative-hypnotic medicines. Retrieved from https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-warning-about-possible-new-risk-sleep-driving-and-other-complex
More… ↓
