TRADJENTA Drug Patent Profile
✉ Email this page to a colleague
When do Tradjenta patents expire, and when can generic versions of Tradjenta launch?
Tradjenta is a drug marketed by Boehringer Ingelheim and is included in one NDA. There are nine patents protecting this drug and one Paragraph IV challenge.
This drug has four hundred and fifty-nine patent family members in forty-five countries.
The generic ingredient in TRADJENTA is linagliptin. There are nineteen drug master file entries for this compound. Three suppliers are listed for this compound. Additional details are available on the linagliptin profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Tradjenta
A generic version of TRADJENTA was approved as linagliptin by SUNSHINE on August 31st, 2021.
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for TRADJENTA?
- What are the global sales for TRADJENTA?
- What is Average Wholesale Price for TRADJENTA?
Summary for TRADJENTA
| International Patents: | 459 |
| US Patents: | 9 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 3 |
| Raw Ingredient (Bulk) Api Vendors: | 93 |
| Clinical Trials: | 10 |
| Patent Applications: | 1,600 |
| Formulation / Manufacturing: | see details |
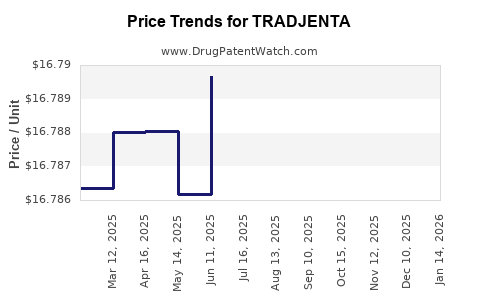
| Drug Prices: | Drug price information for TRADJENTA |
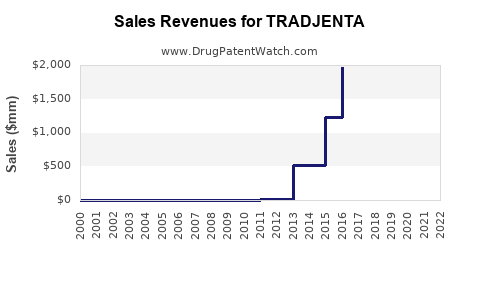
| Drug Sales Revenues: | Drug sales revenues for TRADJENTA |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for TRADJENTA |
| What excipients (inactive ingredients) are in TRADJENTA? | TRADJENTA excipients list |
| DailyMed Link: | TRADJENTA at DailyMed |
Recent Clinical Trials for TRADJENTA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| EMS | Phase 3 |
| University of Miami | Phase 4 |
| Northwell Health | Phase 4 |
Pharmacology for TRADJENTA
| Drug Class | Dipeptidyl Peptidase 4 Inhibitor |
| Mechanism of Action | Dipeptidyl Peptidase 4 Inhibitors |
Paragraph IV (Patent) Challenges for TRADJENTA
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| TRADJENTA | Tablets | linagliptin | 5 mg | 201280 | 11 | 2015-05-04 |
US Patents and Regulatory Information for TRADJENTA
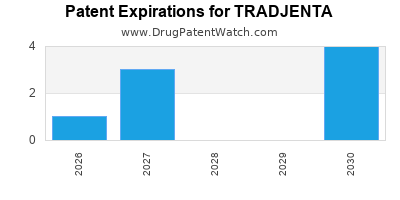
TRADJENTA is protected by nine US patents and two FDA Regulatory Exclusivities.
Patents protecting TRADJENTA
Treatment for diabetes in patients inappropriate for metformin therapy
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
DPP IV inhibitor formulations
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: LINAGLIPTIN (5 MG DAILY DOSE) AND METFORMIN (WITH OR WITHOUT INSULIN) FOR TREATING TYPE 2 DIABETES PATIENTS WITH RENAL IMPAIRMENT AND INSUFFICIENT GLYCEMIC CONTROL DESPITE PREVIOUS TREATMENT WITH METFORMIN ALONE OR IN COMBINATION WITH INSULIN
8-[3-amino-piperidin-1-yl]-xanthines, the preparation thereof and their use as pharmaceutical compositions
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Uses of DPP-IV inhibitors
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Treatment for diabetes in patients with inadequate glycemic control despite metformin therapy comprising a DPP-IV inhibitor
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Treatment for diabetes in patients inappropriate for metformin therapy
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Process for the preparation of chiral 8-(3-aminopiperidin-1-yl)-xanthines
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Treatment for diabetes in patients inappropriate for metformin therapy
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
FDA Regulatory Exclusivity protecting TRADJENTA
REVISIONS TO THE PEDIATRIC USE SUBSECTION OF LABELING TO INCLUDE THE RESULTS FROM CLINICAL STUDY 1218-0091, CONDUCTED TO FULFILL A PEDIATRIC WRITTEN REQUEST
Exclusivity Expiration: ⤷ Sign Up
PEDIATRIC EXCLUSIVITY
Exclusivity Expiration: ⤷ Sign Up
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Boehringer Ingelheim | TRADJENTA | linagliptin | TABLET;ORAL | 201280-001 | May 2, 2011 | AB | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | |||
| Boehringer Ingelheim | TRADJENTA | linagliptin | TABLET;ORAL | 201280-001 | May 2, 2011 | AB | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | ||
| Boehringer Ingelheim | TRADJENTA | linagliptin | TABLET;ORAL | 201280-001 | May 2, 2011 | AB | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | ||
| Boehringer Ingelheim | TRADJENTA | linagliptin | TABLET;ORAL | 201280-001 | May 2, 2011 | AB | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for TRADJENTA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Boehringer Ingelheim | TRADJENTA | linagliptin | TABLET;ORAL | 201280-001 | May 2, 2011 | ⤷ Sign Up | ⤷ Sign Up |
| Boehringer Ingelheim | TRADJENTA | linagliptin | TABLET;ORAL | 201280-001 | May 2, 2011 | ⤷ Sign Up | ⤷ Sign Up |
| Boehringer Ingelheim | TRADJENTA | linagliptin | TABLET;ORAL | 201280-001 | May 2, 2011 | ⤷ Sign Up | ⤷ Sign Up |
| Boehringer Ingelheim | TRADJENTA | linagliptin | TABLET;ORAL | 201280-001 | May 2, 2011 | ⤷ Sign Up | ⤷ Sign Up |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for TRADJENTA
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Boehringer Ingelheim International GmbH | Trajenta | linagliptin | EMEA/H/C/002110 Trajenta is indicated in the treatment of type 2 diabetes mellitus to improve glycaemic control in adults:as monotherapyin patients inadequately controlled by diet and exercise alone and for whom metformin is inappropriate due to intolerance, or contraindicated due to renal impairment.as combination therapyin combination with metformin when diet and exercise plus metformin alone do not provide adequate glycaemic control.in combination with a sulphonylurea and metformin when diet and exercise plus dual therapy with these medicinal products do not provide adequate glycaemic control.in combination with insulin with or without metformin, when this regimen alone, with diet and exercise, does not provide adequate glycaemic control. |
Authorised | no | no | no | 2011-08-23 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for TRADJENTA
When does loss-of-exclusivity occur for TRADJENTA?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 0755
Patent: FORMULACIONES DE INHIBIDORES DE DIPEPTIDIL PEPTIDASA IV (DPP IV)
Estimated Expiration: ⤷ Sign Up
Patent: 9930
Patent: FORMULACIONES DE INHIBIDORES DE DIPEPTIDIL PEPTIDASA IV (DPP IV)
Estimated Expiration: ⤷ Sign Up
Australia
Patent: 07247193
Patent: DPP IV inhibitor formulations
Estimated Expiration: ⤷ Sign Up
Austria
Patent: 80228
Estimated Expiration: ⤷ Sign Up
Brazil
Patent: 0711179
Patent: composição farmacêutica contendo composto inibidor de dpp iv e processo para a sua preparação
Estimated Expiration: ⤷ Sign Up
Patent: 0722388
Patent: Forma de dosagem oral farmacêutica preparada com uma composição farmacêutica compreendendo composto inibidor de dpp iv
Estimated Expiration: ⤷ Sign Up
Canada
Patent: 49922
Patent: FORMULATIONS DE L'INHIBITEUR DE DPP IV (DPP IV INHIBITOR FORMULATIONS)
Estimated Expiration: ⤷ Sign Up
Chile
Patent: 12002521
Patent: Composicion farmaceutica que comprende un inhibidor de dpp iv seleccionado de un grupo definido, un primer diluyente, un segundo diluyente: manitol, un segundo diluyente: almidon pregelatinizado, un aglutinante y un lubricante; util en el tratamiento de diabetes. (divisional de la sol. 1260-07).
Estimated Expiration: ⤷ Sign Up
Patent: 12002522
Patent: Composición farmacéutica que comprende un compuesto inhibidor de dpp iv con un grupo amino o una sal del mismo , un primer diluyente , un segundo diluyente ,un aglutinante , un disgregante y un lubricante, útil en el tratamiento de diabetes mellitus.
Estimated Expiration: ⤷ Sign Up
China
Patent: 1437493
Patent: DPP IV inhibitor formulations
Estimated Expiration: ⤷ Sign Up
Patent: 2526737
Patent: DPP IV inhibitor formulations
Estimated Expiration: ⤷ Sign Up
Croatia
Patent: 0100507
Estimated Expiration: ⤷ Sign Up
Patent: 0150003
Estimated Expiration: ⤷ Sign Up
Cyprus
Patent: 11354
Estimated Expiration: ⤷ Sign Up
Patent: 16064
Estimated Expiration: ⤷ Sign Up
Denmark
Patent: 23902
Estimated Expiration: ⤷ Sign Up
Patent: 77509
Estimated Expiration: ⤷ Sign Up
Patent: 83819
Estimated Expiration: ⤷ Sign Up
Ecuador
Patent: 088800
Patent: Formulaciones de inhibidores de DPP IV
Estimated Expiration: ⤷ Sign Up
Eurasian Patent Organization
Patent: 6559
Patent: ФАРМАЦЕВТИЧЕСКИЕ КОМПОЗИЦИИ С ИНГИБИТОРАМИ DPP IV (DPP IV INHIBITOR FORMULATIONS)
Estimated Expiration: ⤷ Sign Up
Patent: 9890
Patent: ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ И ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ В ПЕРОРАЛЬНОЙ ДОЗИРОВАННОЙ ЛЕКАРСТВЕННОЙ ФОРМЕ НА ОСНОВЕ ИНГИБИТОРА DPP IV (PHARMACEUTICAL COMPOSITION AND PHARMACEUTICAL COMPOSITION IN ORAL DOSAGE FORM BASED ON DPP IV INHIBITOR)
Estimated Expiration: ⤷ Sign Up
Patent: 0802184
Patent: ФАРМАЦЕВТИЧЕСКИЕ КОМПОЗИЦИИ С ИНГИБИТОРАМИ DPP IV
Estimated Expiration: ⤷ Sign Up
Patent: 1100958
Patent: ФАРМАЦЕВТИЧЕСКАЯ ПЕРОРАЛЬНАЯ ДОЗИРОВАННАЯ ЛЕКАРСТВЕННАЯ ФОРМА НА ОСНОВЕ ИНГИБИТОРОВ DPP IV
Estimated Expiration: ⤷ Sign Up
European Patent Office
Patent: 52108
Patent: Compositions d'inhibiteurs de la DPP IV (DPP IV inhibitor formulations)
Estimated Expiration: ⤷ Sign Up
Patent: 23902
Patent: FORMULATIONS DE L'INHIBITEUR DE DPP IV (DPP IV INHIBITOR FORMULATIONS)
Estimated Expiration: ⤷ Sign Up
Patent: 77509
Patent: Compositions d'inhibiteurs de la DPP IV (DPP IV inhibitor formulations)
Estimated Expiration: ⤷ Sign Up
Patent: 83819
Patent: Compositions d'inhibiteurs de la DPP IV (DPP IV inhibitor formulations)
Estimated Expiration: ⤷ Sign Up
Patent: 10241
Patent: Formules d'inhibiteur dpp iv (DPP IV INHIBITOR FORMULATIONS)
Estimated Expiration: ⤷ Sign Up
Germany
Patent: 2007009091
Estimated Expiration: ⤷ Sign Up
Hong Kong
Patent: 30442
Patent: DPP IV INHIBITOR FORMULATIONS
Estimated Expiration: ⤷ Sign Up
Patent: 72549
Patent: 二肽基肽酶 抑制劑製劑 (DPP IV INHIBITOR FORMULATIONS IV)
Estimated Expiration: ⤷ Sign Up
Hungary
Patent: 25210
Estimated Expiration: ⤷ Sign Up
Israel
Patent: 5030
Patent: הרכבים של מעכב iv dpp (Dpp iv inhibitor formulations)
Estimated Expiration: ⤷ Sign Up
Japan
Patent: 78244
Estimated Expiration: ⤷ Sign Up
Patent: 00998
Estimated Expiration: ⤷ Sign Up
Patent: 64720
Estimated Expiration: ⤷ Sign Up
Patent: 87908
Estimated Expiration: ⤷ Sign Up
Patent: 84711
Estimated Expiration: ⤷ Sign Up
Patent: 09535376
Estimated Expiration: ⤷ Sign Up
Patent: 12072187
Patent: DPP IV INHIBITOR FORMULATION
Estimated Expiration: ⤷ Sign Up
Patent: 13227338
Patent: DPP IV INHIBITOR FORMULATION
Estimated Expiration: ⤷ Sign Up
Patent: 16104811
Patent: DPPIVインヒビター製剤 (DPP IV INHIBITOR PREPARATIONS)
Estimated Expiration: ⤷ Sign Up
Patent: 18021082
Patent: DPP IVインヒビター製剤 (DPP IV INHIBITOR FORMULATIONS)
Estimated Expiration: ⤷ Sign Up
Patent: 20079316
Patent: DPP IVインヒビター製剤 (DPP IV INHIBITOR FORMULATIONS)
Estimated Expiration: ⤷ Sign Up
Patent: 22075826
Patent: DPP IVインヒビター製剤
Estimated Expiration: ⤷ Sign Up
Malaysia
Patent: 6969
Patent: DPP IV INHIBITOR FORMULATIONS
Estimated Expiration: ⤷ Sign Up
Patent: 8496
Patent: DPP IV INHIBITOR FORMULATIONS
Estimated Expiration: ⤷ Sign Up
Mexico
Patent: 8617
Patent: FORMULACIONES DE INHIBIDORES DE DPP IV. (DPP IV INHIBITOR FORMULATIONS.)
Estimated Expiration: ⤷ Sign Up
Patent: 08013958
Patent: FORMULACIONES DE INHIBIDORES DE DPP IV. (DPP IV INHIBITOR FORMULATIONS.)
Estimated Expiration: ⤷ Sign Up
Montenegro
Patent: 170
Patent: FORMULACIJE DPP IV INHIBITORA (DPP IV INHIBITOR FORMULATIONS)
Estimated Expiration: ⤷ Sign Up
Patent: 941
Patent: Formulacije DPP IV inhibitora (DPP IV inhibitor formulations)
Estimated Expiration: ⤷ Sign Up
New Zealand
Patent: 2862
Patent: DPP IV inhibitor formulations which comprise mannitol, pregelatinised starch, copovidone, corn starch and magnesium stearate
Estimated Expiration: ⤷ Sign Up
Patent: 5983
Patent: DPP IV inhibitor formulations
Estimated Expiration: ⤷ Sign Up
Patent: 3426
Patent: Dpp iv inhibitor formulations
Estimated Expiration: ⤷ Sign Up
Norway
Patent: 3067
Estimated Expiration: ⤷ Sign Up
Patent: 084256
Estimated Expiration: ⤷ Sign Up
Peru
Patent: 080698
Patent: COMPOSICIONES FARMACEUTICAS DE INHIBIDORES DE DPP IV
Estimated Expiration: ⤷ Sign Up
Patent: 110666
Patent: COMPOSICIONES FARMACEUTICAS DE INHIBIDORES DE DPP IV
Estimated Expiration: ⤷ Sign Up
Poland
Patent: 23902
Estimated Expiration: ⤷ Sign Up
Patent: 77509
Estimated Expiration: ⤷ Sign Up
Patent: 83819
Estimated Expiration: ⤷ Sign Up
Portugal
Patent: 23902
Estimated Expiration: ⤷ Sign Up
Patent: 83819
Estimated Expiration: ⤷ Sign Up
Serbia
Patent: 466
Patent: FORMULACIJE DPP IV INHIBITORA (DPP IV INHIBITOR FORMULATIONS)
Estimated Expiration: ⤷ Sign Up
Patent: 570
Patent: FORMULACIJE DPP IV INHIBITORA (DPP IV INHIBITOR FORMULATIONS)
Estimated Expiration: ⤷ Sign Up
Singapore
Patent: 1649
Patent: DPP IV INHIBITOR FORMULATIONS
Estimated Expiration: ⤷ Sign Up
Slovenia
Patent: 23902
Estimated Expiration: ⤷ Sign Up
Patent: 83819
Estimated Expiration: ⤷ Sign Up
South Africa
Patent: 0808361
Patent: DPP IV inhibitor formulations
Estimated Expiration: ⤷ Sign Up
South Korea
Patent: 1478983
Estimated Expiration: ⤷ Sign Up
Patent: 1710881
Estimated Expiration: ⤷ Sign Up
Patent: 1855323
Estimated Expiration: ⤷ Sign Up
Patent: 2051281
Estimated Expiration: ⤷ Sign Up
Patent: 090009226
Patent: DPP IV INHIBITOR FORMULATIONS
Estimated Expiration: ⤷ Sign Up
Patent: 140063896
Patent: DPP IV INHIBITOR FORMULATIONS
Estimated Expiration: ⤷ Sign Up
Patent: 150100957
Patent: DPP IV INHIBITOR FORMULATIONS
Estimated Expiration: ⤷ Sign Up
Patent: 160128446
Patent: DPP IV 억제제 제형 (DPP IV inhibitor formulations)
Estimated Expiration: ⤷ Sign Up
Patent: 170141812
Patent: DPP IV 억제제 제형 (DPP IV inhibitor formulations)
Estimated Expiration: ⤷ Sign Up
Spain
Patent: 48576
Estimated Expiration: ⤷ Sign Up
Patent: 27409
Estimated Expiration: ⤷ Sign Up
Patent: 38818
Estimated Expiration: ⤷ Sign Up
Taiwan
Patent: 74843
Estimated Expiration: ⤷ Sign Up
Patent: 20753
Estimated Expiration: ⤷ Sign Up
Patent: 0812648
Patent: DPP IV inhibitor formulations
Estimated Expiration: ⤷ Sign Up
Patent: 1417844
Patent: DPP IV inhibitor formulations
Estimated Expiration: ⤷ Sign Up
Ukraine
Patent: 942
Patent: ФАРМАЦЕВТИЧЕСКИЕ КОМПОЗИЦИИ С ИНГИБИТОРАМИ DPP IV;ФАРМАЦЕВТИЧНІ КОМПОЗИЦІЇ З ІНГІБІТОРАМИ DPP IV (DPP IV INHIBITOR FORMULATIONS)
Estimated Expiration: ⤷ Sign Up
Uruguay
Patent: 319
Patent: FORMULACIONES DE INHIBIDORES DE DPP IV
Estimated Expiration: ⤷ Sign Up
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering TRADJENTA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| China | 107266449 | 手性8‑(3‑氨基‑哌啶‑1‑基)‑黄嘌呤的制备方法 (Method for producing chiral 8-(3-amino-piperidin-1-yl)-xanthines) | ⤷ Sign Up |
| Australia | 721477 | ⤷ Sign Up | |
| South Korea | 20140048345 | USE OF DPP-IV INHIBITORS | ⤷ Sign Up |
| Chile | 2012002521 | Composicion farmaceutica que comprende un inhibidor de dpp iv seleccionado de un grupo definido, un primer diluyente, un segundo diluyente: manitol, un segundo diluyente: almidon pregelatinizado, un aglutinante y un lubricante; util en el tratamiento de diabetes. (divisional de la sol. 1260-07). | ⤷ Sign Up |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for TRADJENTA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2187879 | 122017000024 | Germany | ⤷ Sign Up | PRODUCT NAME: EMPAGLIFLOZIN MIT LINAGLIPTIN ODER EINEM PHARMAZEUTISCH AKZEPTABLEN SALZ HIERVON; REGISTRATION NO/DATE: EU/1/16/1146/001-018 20161111 |
| 1532149 | PA2012022 | Lithuania | ⤷ Sign Up | PRODUCT NAME: LINAGLIPTINUM + METFORMINI HYDROCHLORIDUM; REGISTRATION NO/DATE: EU/1/12/780/001 - EU/1/12/780/034 20120720 |
| 2187879 | SPC/GB17/031 | United Kingdom | ⤷ Sign Up | PRODUCT NAME: 1-CHLORO-4-(SS-D-GLUCOPYRANOS-1-YL)-2-(4-((S)-TETRAHYDROFURAN-3-YLOXY)-BENZYL)-BENZENE (I.E. EMPAGLIFLOZIN) IN COMBINATION WITH 1-((4 METHYL-QUINAZOLIN-2-YL)METHYL)-3-METHYL-7-(2-BUTYN-1-YL)-8-(3-(R)-AMINO-PIPERIDIN-1-YL)-XANTHINE (I.E. LINAGLIPTIN) OR A P; REGISTERED: UK PLGB 14598/0191 (GB) 20161115; UK EU/1/16/1146/014(NI) 20161115; UK EU/1/16/1146/015(NI) 20161115; UK EU/1/16/1146/016(NI) 20161115; UK EU/1/16/1146/017(NI) 20161115; UK EU/1/16/1146/018(NI) 20161115; UK EU/1/16/1146/007(NI) 20161115; UK EU/1/16/1146/001(NI) 20161115; UK EU/1/16/1146/013(NI) 20161115; UK EU/1/16/1146/002(NI) 20161115; UK EU/1/16/1146/003(NI) 20161115; UK EU/1/16/1146/004(NI) 20161115; UK... |
| 1532149 | CR 2011 00030 | Denmark | ⤷ Sign Up | PRODUCT NAME: LINAGLIPTIN OG SALTE DERAF; NAT. REG. NO/DATE: EU/1/11/707/001-011 20110830; FIRST REG. NO/DATE: EU EU/1/11/707/001-0011 20110830 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


