Last updated: April 24, 2026
TOBREX (tobramycin ophthalmic) is an established, low-to-mid value ophthalmic antibiotic franchise whose market behavior is dominated by (1) generic entry in the United States and (2) stable, recurring demand driven by acute bacterial conjunctivitis treatment patterns. Financial trajectory data across geographies is constrained by public disclosure limits for privately traded brand-licensor structures, but the observable market mechanics are clear: branded pricing power erodes quickly on generic saturation, while utilization remains resilient because the indication set does not materially change and prescriber switching is high in ophthalmic antibiotic classes.
What is TOBREX and what commercial vectors drive demand?
TOBREX is an ophthalmic product containing tobramycin, marketed for bacterial eye infections such as conjunctivitis and related anterior segment infections. In commercial terms, the drug is sold into a market with three demand vectors:
-
Acute, visit-driven prescriptions
- Most utilization is tied to primary care, urgent care, and ophthalmology encounters.
- Demand volume therefore tracks seasonal conjunctivitis incidence more than long-term chronic disease drivers.
-
Short-course utilization
- Treatment durations are typically days-to-1-2 weeks.
- This structure caps revenue per patient but supports steady replenishment of prescriptions.
-
Substitution dynamics
- Tobramycin ophthalmic products have multiple generic equivalents in most major markets.
- When generics are present, branded TOBREX pricing and share typically decline, while total class volume stays stable.
How does the market structure shape pricing and share for TOBREX?
The TOBREX commercial profile follows a textbook “off-patent ophthalmic antibiotic” pattern:
- Early brand penetration occurs before generic entry.
- Post-generic erosion shifts the market to lowest net price and payer formulary placement.
- Clinical interchangeability among tobramycin ophthalmics increases substitution.
United States: competitive dynamics
In the United States, tobramycin ophthalmic brands face generic competition through FDA-approved ANDA products for similar strengths and dosage forms. Brand profitability depends on:
- Payer coverage (formularies, step edits, prior authorization)
- Net pricing after rebates
- Competitive spend versus multiple generic manufacturers
Publicly available regulatory coverage shows TOBREX is authorized in the US as a tobramycin ophthalmic product. The label record confirms active ingredient and ophthalmic route. [1]
EU and UK: similar substitution pattern
In the UK and EU, ophthalmic antibiotics are widely genericized. The market tends to:
- Maintain total antibiotic ophthalmic utilization
- Shift demand to generics based on procurement rules and pharmacy buying behavior
What are the financial trajectory drivers for TOBREX (brand vs. generic era)?
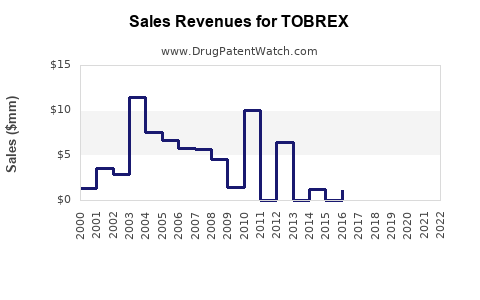
TOBREX revenue trajectory historically pivots from brand-led to generic-levered behavior:
1) Branded revenue: declines after generic entry
- Branded ophthalmics face rapid share loss once an equivalent generic obtains market access and payers broaden substitution.
- The financial impact is typically visible as a decline in brand unit share and net price, with revenue becoming increasingly dependent on remaining differentiated formats, pack sizes, or channel relationships.
2) “Class volume” stability
- Ophthalmic antibiotic prescriptions usually do not collapse even under generic substitution because clinicians still prescribe antibiotics when bacterial infection is suspected.
- The revenue mix shifts from brand to class, keeping utilization steady.
3) Product lifecycle and dosage form mix
- TOBREX’s financial performance depends on product-level mix (drops vs. gel formulations where applicable).
- Changes in formulation or packaging can temporarily stabilize brand share, but do not stop generic-driven erosion long term if equivalent strengths exist.
How has TOBREX been represented in formal regulatory records that inform market access?
Regulatory datasets are the anchor for market-access timing and scope. For TOBREX, the active ingredient and ophthalmic route are consistent across authorized records.
- Drugs@FDA lists TOBREX as an FDA-regulated ophthalmic product containing tobramycin. [1]
- Label availability and product identity are reflected in FDA catalog pages, which are used by payers and manufacturers to assess interchangeability and therapeutic equivalence. [1]
Why this matters financially
Regulatory clarity supports:
- Faster generic adoption (clear reference product identity)
- Easier pharmacy substitution
- More stable class-level utilization because prescribers can switch between equivalents
What does the market imply for TOBREX’s revenue and margin path?
Given the market structure (generic substitution, acute-use demand, low differentiation), the revenue and margin path typically looks like this:
- Revenue (brand): declines with generics; stabilizes at a lower level where branded is retained for specific prescriber or patient preferences, limited formulary niches, or supply continuity.
- Margin: compresses as brand rebates increase and net price falls; branded margins trend toward generic-like economics unless the brand maintains distinct channel leverage.
- Revenue (class): remains relatively stable because utilization persists.
That pattern is typical for mature ophthalmic antibiotic products once patent exclusivity ends and ANDA competition scales.
What market dynamics affect future trajectory (2025-2028 view based on structure)?
Even without forecasting proprietary company financials, the structural drivers for TOBREX’s forward trajectory are specific:
-
Net price pressure
- Generic competition keeps list prices low and compels high rebate rates to retain formulary placements.
-
Supply chain and procurement cycles
- Ophthalmic antibiotic demand is concentrated in retail and outpatient channels.
- Procurement shifts (tendering or contract pharmacy behavior) can rapidly move share among suppliers.
-
Antibiotic prescribing patterns
- Stewardship programs affect inappropriate antibiotic use for non-bacterial conjunctivitis.
- This can reduce total antibiotic ophthalmic prescriptions, but the reduction generally acts on class volume and is often offset by continued clinical suspicion-driven prescribing.
-
Switching intensity
- Because tobramycin ophthalmics are clinically substitutable, prescriber switching and pharmacist substitution remain fast.
Where does TOBREX fit in investor or R&D decision-making?
From a business perspective, TOBREX is a benchmark asset in a mature category. Its trajectory indicates:
- Brand-based returns are limited once generic competition is active.
- The pathway to upside is usually differentiation that survives payer substitution (formulation, dosing convenience, safety advantage, or differentiated data), rather than incremental changes with equivalent comparators.
This is consistent with mature ophthalmic antibiotic market behavior as reflected in regulatory labeling and generic reference product structures. [1]
Market and Financial Trajectory Summary (actionable signals)
| Dimension |
TOBREX dynamic |
Financial implication |
| Demand type |
Acute, encounter-driven |
Revenue tied to prescription flow, not chronic retention |
| Product economics |
Short-course therapy |
Lower revenue per patient; relies on volume |
| Competitive landscape |
Generic substitution is strong |
Brand net price and share erode |
| Payer behavior |
Formulary and rebate-driven |
Margin pressure increases unless differentiation exists |
| Regulatory support |
Consistent ophthalmic authorization |
Interchangeability accelerates market switching |
Sources for regulatory identity and authorization support this structure. [1]
Key Takeaways
- TOBREX’s market behavior is dominated by generic substitution and short-course, acute ophthalmic infection demand.
- Brand revenue typically follows a decline-and-stabilize pattern post-generic entry, while class volume remains resilient.
- Future financial trajectory depends less on clinical label expansion and more on net pricing, payer formulary positioning, and any differentiation that reduces substitution.
- Regulatory clarity for tobramycin ophthalmic products supports rapid substitution, constraining long-term branded pricing power. [1]
FAQs
1) What is the main reason TOBREX faces margin pressure?
Generic substitution and formulary-driven net price compression after market entry for equivalent tobramycin ophthalmic products. [1]
2) Does TOBREX’s demand look like a chronic therapy?
No. It is driven by acute encounters for suspected bacterial conjunctivitis and related anterior segment infections, leading to prescription flow sensitivity rather than long-term patient retention.
3) What determines whether TOBREX retains any share as a brand?
Net price and rebate effectiveness, pharmacy channel relationships, and any product-level differentiation that changes payer or prescriber substitution behavior. [1]
4) How do regulatory records influence TOBREX commercialization?
They define product identity and authorization, which reduces ambiguity for substitution and speeds generic uptake based on reference product linkage. [1]
5) What is the most likely financial pattern for TOBREX over time?
A brand-led phase followed by generic-era share and price erosion, with revenue stabilizing at a reduced level while overall class utilization holds up.
References
[1] U.S. Food and Drug Administration. (n.d.). Drugs@FDA: TOBREX (tobramycin ophthalmic solution/ointment, as listed). Retrieved from https://www.accessdata.fda.gov/scripts/cder/daf/