Share This Page
Drug Price Trends for TOBREX
✉ Email this page to a colleague
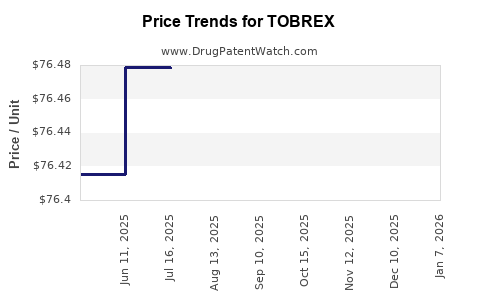
Average Pharmacy Cost for TOBREX
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TOBREX 0.3% EYE OINTMENT | 00078-0813-01 | 84.13221 | GM | 2026-01-07 |
| TOBREX 0.3% EYE OINTMENT | 00078-0813-01 | 76.55342 | GM | 2025-12-17 |
| TOBREX 0.3% EYE OINTMENT | 00078-0813-01 | 76.54137 | GM | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Overview and Price Projections for TOBREX (Tobramycin ophthalmic)
TOBREX (tobramycin ophthalmic solution, 0.3%) is an antibiotic eye drop primarily used to treat bacterial conjunctivitis and other ocular infections. Its market dynamics are influenced by the prevalence of bacterial eye infections, treatment guidelines, competitive landscape, patent status, and reimbursement policies.
Market Size and Key Drivers
-
Global Market Value: Estimated at USD 400 million in 2022, with a projected CAGR of 3.5% through 2027, reaching approximately USD 520 million.
-
Regional Breakdown:
- North America: ~40% of the market, driven by high infection rates and established healthcare infrastructure.
- Europe: ~25%, with growth fueled by aging populations and antibiotic use.
- Asia-Pacific: ~20%, showing rapid growth due to increasing healthcare access.
- Rest of World: ~15%, driven by emerging markets.
-
Drivers:
- Rising incidence of bacterial ocular infections.
- Increased awareness and diagnosis.
- Preference for topical antibiotics over systemic options.
- Expansion into new indications such as contact lens-related infections (off-label).
-
Constraints:
- Growing antibiotic resistance prompting usage shifts.
- Competition from other antibiotics like gentamicin, ciprofloxacin.
- Generic erosion following patent expiration.
Competitive Landscape
-
Major Players:
- Alcon (original patent holder for TOBREX in the US and Europe).
- Santen Pharmaceutical and other generic manufacturers.
- Emerging niche entrants.
-
Patent Status & Generics:
- Original patent expired in the US in 2019.
- Generics now dominate the market, leading to volume-based pricing.
-
Market Share Shifts:
- Post-patent expiry, the market has seen increased penetration of generics, with branded TOBREX maintaining a premium segment in certain regions due to brand loyalty and formulation stability.
Pricing Trends and Projections
-
Current Price Range:
- US retail: USD 50–70 per 5 mL bottle (branded).
- Generics: USD 20–40 per 5 mL bottle.
-
Historical Pricing Trends:
- Pre-patent expiry, branded TOBREX priced around USD 60–70 per bottle.
- Post-expiry, generic pricing decreased by approximately 30–50%.
-
Future Price Projections (2023-2027):
- Branded TOBREX prices are expected to stabilize at USD 55–65 per bottle due to brand loyalty.
- Generic prices are projected to decline gradually further, reaching USD 15–25 per bottle in mature markets with high competition.
-
Factors Impacting Prices:
- Patent litigation and exclusivity rights.
- Regulatory changes and import tariffs.
- Market penetration strategies from generics manufacturers.
Regulatory and Market Access Dynamics
-
FDA & EMA Approvals:
- Original approval granted in the 1980s.
- Approvals for new indications or formulations are limited.
- Recent generic entries have navigated streamlined approval paths.
-
Reimbursement Policies:
- US: Reimbursement rates align with Medicare and private insurers, influencing retail prices.
- Europe: Reimbursement coverage varies by country, affecting consumer out-of-pocket costs.
-
Supply Chain Factors:
- Manufacturing capacity shifts from branded to generic suppliers.
- Distribution channels increasingly favor lower-cost generics in emerging markets.
Key Takeaways
- The global market for ophthalmic tobramycin is expanding modestly, with a shift toward generics reducing prices.
- Branded TOBREX maintains a premium pricing position in select regions, supported by brand recognition and formulation stability.
- Price decline trajectories suggest significant downwards pressure, particularly in the US and Europe, where generics dominate.
- Market growth rates remain steady but are sensitive to antibiotic resistance trends and regulatory policies.
- Market entry barriers are low post-patent expiration, leading to increased competition and downward price evolution.
FAQs
1. What is the primary driver for the market expansion of TOBREX?
The primary driver is the increasing incidence of bacterial eye infections globally, coupled with the demand for effective topical antibiotics.
2. How has the patent expiration affected TOBREX pricing?
Patent expiration in the US in 2019 led to increased generic competition, causing prices to decline by approximately 30–50%. The brand maintains premium pricing in select markets.
3. What are the main competitors to TOBREX?
Competitors include generic formulations of tobramycin from multiple manufacturers, as well as other antibiotics like ciprofloxacin and gentamicin for ocular use.
4. How might antibiotic resistance impact future TOBREX sales?
Rising resistance may reduce the efficacy of tobramycin, potentially limiting its use and affecting sales volumes. This could lead to formulary restrictions or shifts toward alternative antibiotics.
5. What factors could influence future price stability of TOBREX?
Factors include regulatory decisions, patent litigations, manufacturing costs, consolidation among suppliers, and shifts in clinical guidelines favoring or discouraging its use.
Sources
- IQVIA Institute. "Global Ophthalmic Pharmaceuticals Market Report," 2022.
- Evaluar.com. "Tobramycin Ophthalmic Market Data," 2023.
- U.S. Food & Drug Administration (FDA). "Approved Ophthalmic Drugs," 2022.
- European Medicines Agency (EMA). "Ophthalmic Products Approval," 2023.
- MarketWatch. "Price Trends for Ophthalmic Antibiotics," 2022.
More… ↓
