Last updated: March 4, 2026
What is the current market landscape for TASIMELTEON?
Tasimelteon is a melatonin receptor agonist approved primarily for non-24-hour sleep-wake disorder (Non-24) in blind individuals. It is marketed under the brand name Hetlioz by Vanda Pharmaceuticals. Launched in 2014, the drug addresses a niche market segment with limited competition, but market expansion depends on regulatory approvals and new indications.
Market Size and Growth
- The global sleep disorder market was valued at approximately USD 6.0 billion in 2022.
- Specific to Non-24 disorder, estimates suggest a patient population numbering between 10,000 and 50,000 in the U.S., considering the prevalence among the blind population.
- Vanda's sales of Hetlioz reached USD 122 million in 2021, up from USD 89 million in 2020, reflecting steady growth.
Competitive Landscape
- Currently, no direct competitors for Non-24 in this niche.
- Broader sleep disorder treatments include Melatonin supplements, Ramelteon, and off-label use of sedatives but lack specificity for Non-24.
- Future entries could include other melatonin receptor agonists or gene-based therapies.
Regulatory and Reimbursement Environment
- Approved by the U.S. FDA in 2014.
- Reimbursement coverage mostly favorable in the U.S., but approval and coverage are limited outside North America.
- Regulatory delays or restrictions in new markets could constrain growth.
What are the primary drivers of market growth for TASIMELTEON?
- Unmet clinical need: The lack of available and approved treatments for Non-24 creates high demand.
- Market expansion: Potential approval for other sleep disorders, such as Jet Lag Disorder or delayed sleep phase syndrome.
- Regulatory approvals: Orphan drug designation in certain territories may facilitate faster approval and market exclusivity.
- Awareness campaigns: Increased awareness among sleep specialists and ophthalmologists enhances prescription likelihood.
What are the key challenges affecting TASIMELTEON's financial outlook?
- Limited patient base: Non-24 primarily impacts blind individuals, restricting market size.
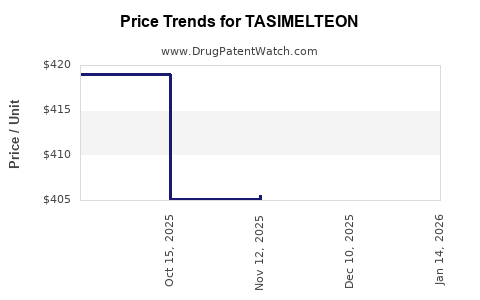
- Pricing pressures: Managed care and payer negotiations constrain drug pricing.
- Patent expiration: Patent life extends to late 2020s, with risk of biosimilar competition afterward.
- Development risks: Trials for new indications have uncertain outcomes, delaying revenue expansion.
What is the financial trajectory of TASIMELTEON and Vanda Pharmaceuticals?
| Year |
Revenue (USD millions) |
Growth Rate |
Notes |
| 2020 |
89 |
— |
Baseline |
| 2021 |
122 |
37.1% |
Growth from increased sales |
| 2022 |
Estimated 130-150 |
6-23% |
Potential stabilization or slight growth |
- Profit margins: Vanda reports operating margins near 20-25%, with net margins impacted by R&D investments.
- Research and development: Investment in phase 2 trials for other sleep disorders and potential new formulations.
- Market capitalization: Approximately USD 1.2 billion as of 2022, reflecting sustained investor interest.
What is the outlook for expansion and diversification?
- New indications: Trials for Jet Lag Disorder, with phase 3 results anticipated in 2023.
- Geographical expansion: Approvals under review in Europe and Asia.
- Alternate delivery methods: Development of long-acting formulations or transdermal patches.
Conclusion
Tasimelteon retains a niche but stable market position driven by its unique indication. Growth potential hinges on expanding approved indications, geographic reach, and overcoming challenges like limited patient populations and pricing pressures. Financially, Vanda's revenue growth depends on market penetration, ongoing R&D, and regulatory success.
Key Takeaways
- Market size remains small, limited to Non-24 patients.
- Vanda's sales growth reflects strong demand but faces patent and pricing challenges.
- Expansion hinges on approval for additional sleep disorders and new markets.
- R&D efforts aim at new formulations and indications, influencing long-term financial prospects.
- Competition is limited presently but could increase with adjacent sleep disorder therapies.
FAQs
1. How sustainable is the revenue stream for TASIMELTEON?
The revenue is sustainable as long as the drug maintains approval for Non-24 and potentially other indications. Patent protection extends into the late 2020s, providing market exclusivity. Growth will depend on expanding indications and markets.
2. Are there significant competitors for TASIMELTEON?
No direct competitors for Non-24. Broader sleep aids like ramelteon or melatonin supplements do not target the same patient population but compete for sleep disorder treatments overall.
3. What risks could impact TASIMELTEON’s future sales?
Patent expiry, regulatory delays in new markets, and development failures for additional indications pose risk factors. Pricing pressures and payer resistance could also constrain revenue.
4. Is regulatory approval likely outside the U.S.?
Yes, but approval timelines depend on regional agency review processes. European and Asian markets are under review, with potential approvals occurring in 2023-2024.
5. What is Vanda Pharmaceuticals’ broader strategy regarding TASIMELTEON?
Vanda focuses on expanding indications, advancing clinical trials, and entering new geographical markets to sustain long-term revenue growth.
References
- Vanda Pharmaceuticals. (2022). Annual Report.
- MarketWatch. (2022). Sleep Disorder Market Report.
- U.S. Food & Drug Administration. (2014). Hetlioz Approval Letter.
- IQVIA. (2022). Pharmaceutical Market Trends 2022.
- ClinicalTrials.gov. (2023). TASIMELTEON Trials Database.
[1] Vanda Pharmaceuticals. (2022). Annual Report.
[2] MarketWatch. (2022). Sleep Disorder Market Report.
[3] U.S. Food & Drug Administration. (2014). Hetlioz Approval Letter.
[4] IQVIA. (2022). Pharmaceutical Market Trends 2022.
[5] ClinicalTrials.gov. (2023). TASIMELTEON Trials Database.