Last updated: February 19, 2026
TACLONEX (calcipotriene and betamethasone dipropionate) is a topical combination product indicated for the treatment of plaque psoriasis. The drug's market performance is influenced by patent expirations, generic competition, and evolving treatment paradigms in dermatology.
What is the current market exclusivity status of TACLONEX?
TACLONEX is marketed by Leo Pharma. The primary U.S. patent for the drug's formulation and use, U.S. Patent No. 5,834,450, expired on March 14, 2017 [1]. Additional patents related to manufacturing processes and specific formulations may have later expiration dates. However, the loss of market exclusivity for the core patent has allowed for the introduction of generic versions.
Who are the key generic competitors for TACLONEX?
Following the expiration of the primary patent, several pharmaceutical companies have launched generic versions of calcipotriene and betamethasone dipropionate topical products. Notable generic manufacturers include:
- Teva Pharmaceuticals
- Mylan N.V. (now Viatris)
- Sun Pharmaceutical Industries
- Apotex Inc.
- Actavis plc (now part of Teva)
These companies offer bioequivalent products, typically in the same strengths and dosage forms (ointment and solution) as the branded TACLONEX. The availability of these generics has significantly impacted TACLONEX's market share and pricing.
How has patent expiration affected TACLONEX sales and market share?
The expiration of U.S. Patent No. 5,834,450 on March 14, 2017, directly led to the erosion of TACLONEX's market exclusivity [1]. This event triggered the entry of multiple generic competitors.
Sales Trajectory:
Prior to patent expiration, TACLONEX generated substantial revenue for Leo Pharma. Post-expiration, sales of the branded product have declined significantly due to price competition from generics. While precise, up-to-the-minute sales figures for branded TACLONEX are proprietary, industry reports indicate a sharp decrease in market share for branded topical psoriasis treatments once generics become available.
- Pre-2017: TACLONEX held a dominant position in the topical combination therapy market for plaque psoriasis, commanding premium pricing.
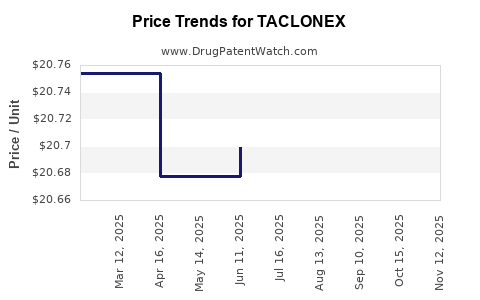
- Post-2017: The market share for branded TACLONEX has been largely ceded to generics. Generic versions are typically priced at a discount, often 30-60% lower than the branded product, depending on market dynamics and payer contracts [2].
Market Share:
The market share for branded TACLONEX has been substantially reduced. The combined market share of generic calcipotriene and betamethasone dipropionate products now represents the majority of prescriptions for this therapeutic combination. For example, data from various market research firms tracking prescription data (e.g., IQVIA) consistently shows a shift from branded to generic topical corticosteroids and vitamin D analogs.
What is the current regulatory landscape for topical psoriasis treatments?
The regulatory environment for topical psoriasis treatments involves approvals from agencies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). For TACLONEX, the key regulatory considerations include:
- FDA Approval: TACLONEX was approved by the FDA in 2002 for the treatment of plaque psoriasis in adults.
- Generic Approvals: Generic versions of calcipotriene and betamethasone dipropionate topical products undergo Abbreviated New Drug Application (ANDA) processes with the FDA. These applications demonstrate bioequivalence to the Reference Listed Drug (RLD), which is TACLONEX in this case.
- Labeling and Indications: Generic products must adhere to the labeling and indications approved for the RLD, unless significant differences are scientifically justified and approved by the FDA.
- Post-Marketing Surveillance: All approved drugs, including generics, are subject to post-marketing surveillance for safety and efficacy.
The regulatory framework ensures that generic alternatives are therapeutically equivalent, facilitating their widespread adoption and contributing to cost savings for healthcare systems and patients.
What are the primary therapeutic alternatives to TACLONEX?
The treatment landscape for plaque psoriasis is multifaceted, offering various therapeutic options beyond calcipotriene and betamethasone dipropionate combinations. Alternatives include:
- Other Topical Corticosteroids: Monotherapy with potent topical corticosteroids such as clobetasol propionate, halcinonide, and fluocinonide. These are often used for more severe or localized lesions.
- Topical Vitamin D Analogs: Calcipotriene (calcipotriol) or tacalcitol monotherapy.
- Topical Retinoids: Tazarotene is a retinoid that can be used alone or in combination with corticosteroids.
- Topical Calcineurin Inhibitors: Tacrolimus and pimecrolimus are options for sensitive areas like the face and intertriginous regions where steroids may cause adverse effects.
- Systemic Therapies: For moderate to severe psoriasis, systemic agents are employed, including:
- Traditional Oral Agents: Methotrexate, cyclosporine, acitretin.
- Biologics: Tumor necrosis factor (TNF)-alpha inhibitors (e.g., adalimumab, etanercept, infliximab), interleukin (IL)-17 inhibitors (e.g., secukinumab, ixekizumab), IL-12/23 inhibitors (e.g., ustekinumab), and IL-23 inhibitors (e.g., guselkumab, risankizumab).
- Phototherapy: Ultraviolet B (UVB) or psoralen plus ultraviolet A (PUVA) therapy.
The choice of therapy depends on psoriasis severity, affected body surface area, patient preference, comorbidities, and cost. The availability of effective systemic and biologic therapies has also influenced the market for topical agents.
What is the global market size and projected growth for topical psoriasis treatments?
The global market for topical psoriasis treatments is substantial, driven by the prevalence of the condition. While TACLONEX itself has seen its branded market shrink, the overall market for topical combination therapies and their generic equivalents remains significant.
Market Size:
Estimates for the global topical psoriasis treatment market vary but generally place it in the low billions of U.S. dollars annually. This market encompasses all topical formulations, including corticosteroids, vitamin D analogs, retinoids, and combination products.
Projected Growth:
The market for topical psoriasis treatments is projected to experience moderate growth. Key drivers include:
- Increasing Psoriasis Prevalence: A growing global population and potentially rising incidence rates contribute to a larger patient pool.
- Demand for Convenient Formulations: Topical treatments offer a convenient and accessible first-line option for many patients.
- Advancements in Topical Formulations: Development of new delivery systems and improved formulations that enhance efficacy and tolerability.
However, growth may be tempered by the increasing adoption of more advanced systemic and biologic therapies for moderate-to-severe psoriasis, which can achieve higher efficacy rates. The rise of generic alternatives will also exert downward pressure on overall market value, even as prescription volumes remain stable or increase.
Key Market Trends:
- Dominance of Generics: The market for established topical combinations like calcipotriene and betamethasone dipropionate is largely driven by generic competition, with price being a primary differentiator.
- Development of Novel Topicals: Pharmaceutical companies continue to invest in developing novel topical agents or improved formulations of existing molecules to address unmet needs, such as enhanced efficacy, better tolerability, and simplified dosing regimens.
- Shift to Systemics: For patients with more severe disease, there is a clear trend towards systemic and biologic therapies, which can offer more profound and sustained disease control.
What is the financial outlook for Leo Pharma concerning TACLONEX post-patent expiration?
Following the patent expiration of TACLONEX, Leo Pharma's financial strategy has shifted. The company has focused on:
- Expanding its Biosimilar and Biologic Portfolio: Leo Pharma is actively developing and marketing biologics and biosimilars for dermatological conditions, aiming to capture value in advanced therapies.
- Geographic Expansion: Seeking growth in emerging markets where branded products may retain market share longer.
- Pipeline Development: Investing in R&D for new dermatological treatments, including novel biologics and small molecules, to compensate for declining revenue from older, off-patent products.
The financial impact on Leo Pharma from TACLONEX has been a reduction in high-margin revenue. The company must balance this with the revenue generated from its generic portfolio and its strategic investments in new therapeutic areas. For a company like Leo Pharma, which relies heavily on dermatology, the transition from a branded product like TACLONEX to a more diversified portfolio including generics and biologics is a critical business imperative.
What are the intellectual property considerations for competitors entering the calcipotriene and betamethasone dipropionate market?
Competitors seeking to enter or expand their presence in the calcipotriene and betamethasone dipropionate market must navigate a complex intellectual property landscape.
- Composition of Matter Patents: The primary composition of matter patent (U.S. Patent No. 5,834,450) for the specific combination has expired [1]. This opens the door for generic formulation.
- Formulation Patents: Companies may hold patents on specific formulations of calcipotriene and betamethasone dipropionate, such as unique ointment bases, creams, or solution vehicles designed to improve stability, penetration, or patient adherence. Competitors must ensure their generic formulations do not infringe on these active formulation patents.
- Method of Use Patents: While the primary indication for plaque psoriasis is well-established, patents may exist for specific methods of using the combination for particular patient subsets or disease severities.
- Manufacturing Process Patents: Patents can cover novel or improved methods of manufacturing the drug substance or the finished dosage form. Generic manufacturers must develop non-infringing manufacturing processes.
- Exclusivity Periods: In addition to patent protection, regulatory exclusivity periods (e.g., New Chemical Entity exclusivity, Orphan Drug exclusivity, Pediatric exclusivity) can provide market protection even after patent expiration. For TACLONEX, these would have largely expired by the time of patent expiry.
Due diligence is essential for generic companies to identify and assess the validity and scope of any remaining active patents. Freedom-to-operate (FTO) analyses are critical to mitigate the risk of patent litigation.
What are the key market dynamics influencing the future of topical combination therapies for psoriasis?
Several market dynamics are shaping the future of topical combination therapies like TACLONEX:
- Cost Containment Pressures: Healthcare payers (governments, insurance companies) are increasingly focused on reducing prescription drug costs. This drives a preference for generics over branded products and may influence formulary placement.
- Advancements in Biologics and Systemics: The development of highly effective biologic and systemic treatments for moderate to severe psoriasis has raised the bar for treatment expectations. This can lead to earlier escalation of therapy for some patients, potentially reducing the duration of topical treatment.
- Patient Preference and Adherence: While effective, topical treatments can be challenging for patients to adhere to due to factors like greasy texture, application frequency, and potential side effects. Newer formulations or combination products that improve convenience and tolerability may gain traction.
- Emergence of Novel Topical Agents: Research continues into new topical molecules and delivery systems, such as topical JAK inhibitors or novel corticosteroid/vitamin D analog formulations, which could offer improved efficacy or safety profiles.
- Psoriasis Subtypes and Severity: Treatment decisions are increasingly tailored to specific psoriasis subtypes and severity levels. Topical combination therapies remain a cornerstone for mild to moderate plaque psoriasis.
- Digital Health and Telemedicine: The increasing use of telemedicine may impact how topical treatments are prescribed and monitored, potentially facilitating remote patient management and adherence support.
The market for established topical combination therapies will likely remain substantial, driven by cost-effectiveness and suitability for mild-to-moderate disease. However, growth will be constrained by competition from generics and the increasing availability of advanced systemic therapies.
Key Takeaways
- TACLONEX's primary U.S. patent expired in March 2017, enabling generic competition.
- Multiple generic manufacturers, including Teva and Mylan, now offer bioequivalent versions of calcipotriene and betamethasone dipropionate.
- The introduction of generics has led to a significant decline in branded TACLONEX sales and market share.
- The topical psoriasis market is competitive, with alternatives including other topical agents, phototherapy, and advanced systemic/biologic therapies.
- Leo Pharma's strategy has shifted to focus on new product development, biosimilars, and biologics to offset revenue decline from off-patent products.
- Future market dynamics will be influenced by cost containment, advancements in biologics, patient adherence, and the development of novel topical agents.
Frequently Asked Questions
- When did the primary patent for TACLONEX expire in the United States?
The primary U.S. patent for TACLONEX, U.S. Patent No. 5,834,450, expired on March 14, 2017.
- What impact has generic competition had on the pricing of TACLONEX?
Generic versions of TACLONEX are typically priced at a significant discount, often 30-60% lower than the branded product, due to market competition and cost pressures.
- Are there any remaining patents that prevent generic entry?
While the main composition of matter patent has expired, competitors must still navigate potential patents related to specific formulations, manufacturing processes, or methods of use to ensure freedom to operate.
- What is the primary indication for TACLONEX?
TACLONEX is indicated for the topical treatment of plaque psoriasis in adults.
- What therapeutic class does TACLONEX belong to?
TACLONEX is a combination topical therapy consisting of a corticosteroid (betamethasone dipropionate) and a vitamin D analog (calcipotriene).
Citations
[1] United States Patent No. 5,834,450. (1998). Combination of calcipotriol and a corticosteroid. Retrieved from USPTO Patent Full-Text and Image Database.
[2] Industry Market Analysis Reports (Various, proprietary). (2017-2023). Analysis of prescription drug pricing and generic penetration in the US dermatology market.