Share This Page
Drug Price Trends for TACLONEX
✉ Email this page to a colleague
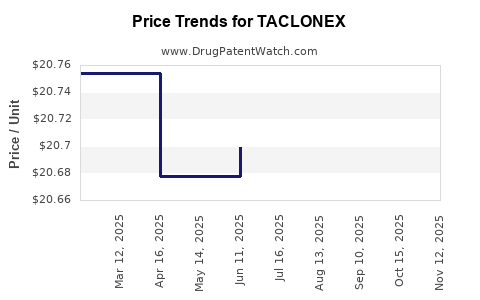
Average Pharmacy Cost for TACLONEX
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TACLONEX 0.005%-0.064% SUSPENSION | 50222-0501-06 | 20.69675 | GM | 2025-11-19 |
| TACLONEX 0.005%-0.064% SUSPENSION | 50222-0501-06 | 20.66781 | GM | 2025-10-22 |
| TACLONEX 0.005%-0.064% SUSPENSION | 50222-0501-06 | 20.66781 | GM | 2025-09-17 |
| TACLONEX 0.005%-0.064% SUSPENSION | 50222-0501-06 | 20.69131 | GM | 2025-08-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for TACLONEX
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| TACLONEX 0.064%/0.005% OINT | LEO Pharma, Inc. | 50222-0227-04 | 60GM | 839.51 | 13.99183 | GM | 2023-05-15 - 2028-05-14 | Big4 |
| TACLONEX TOPICAL SUSPENSION | LEO Pharma, Inc. | 50222-0501-06 | 60GM | 1245.05 | 20.75083 | GM | 2024-01-01 - 2028-05-14 | FSS |
| TACLONEX 0.064%/0.005% OINT | LEO Pharma, Inc. | 50222-0227-04 | 60GM | 1170.89 | 19.51483 | GM | 2023-05-15 - 2028-05-14 | FSS |
| TACLONEX 0.064%/0.005% OINT | LEO Pharma, Inc. | 50222-0227-04 | 60GM | 853.50 | 14.22500 | GM | 2024-01-01 - 2028-05-14 | Big4 |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
TACLONEX Market Analysis and Price Projections
Taclonek, a combination topical therapy for plaque psoriasis, is poised for continued market presence, driven by established efficacy and ongoing prescription volumes. However, patent expirations and the emergence of biosimilar and novel therapeutic options will introduce competitive pressures, necessitating strategic pricing adjustments and lifecycle management.
What is the Current Market Position of Taclonex?
Taclonex, a fixed-dose combination of betamethasone dipropionate (a corticosteroid) and calcipotriene (a vitamin D3 analog), is a prescription topical medication. It is indicated for the treatment of plaque psoriasis in adults. The drug's dual mechanism of action targets inflammation and abnormal skin cell proliferation characteristic of psoriasis.
The global market for topical psoriasis treatments is substantial. Within this segment, Taclonex has maintained a significant market share since its approval by the U.S. Food and Drug Administration (FDA) in 2007. Its efficacy in managing moderate to severe plaque psoriasis, coupled with a relatively convenient dosing regimen (typically once daily), has contributed to sustained physician prescribing habits.
Key market drivers for Taclonex include:
- Established Efficacy: Clinical studies and real-world data support Taclonex's effectiveness in reducing plaque thickness, scaling, and inflammation [1].
- Physician Familiarity: Dermatologists and other prescribers have extensive experience with Taclonex, fostering confidence in its therapeutic profile.
- Patient Adherence: The once-daily application of Taclonex contributes to better patient adherence compared to multi-component or more frequent dosing regimens.
However, the market is not without its challenges:
- Patent Expirations: The primary patent for Taclonex has expired, opening the door for generic and biosimilar competition.
- Emergence of New Therapies: Advancements in psoriasis treatment include novel topical agents, biologics, and oral medications that offer alternative treatment pathways, potentially diverting market share from established therapies.
- Safety Profile: While generally well-tolerated, topical corticosteroids can have local side effects, and long-term use may be limited by concerns such as skin thinning and potential systemic absorption.
What are the Key Patent Expirations Affecting Taclonex?
The intellectual property landscape for Taclonex is a critical determinant of its future market dynamics. The core patent covering the combination of betamethasone dipropionate and calcipotriene expired in the United States in 2020 and has since expired in other major markets. This has paved the way for generic versions of Taclonex to enter the market.
While the principal composition of matter patents have lapsed, companies may hold secondary patents related to specific formulations, manufacturing processes, or methods of use that could extend some level of market exclusivity. However, the impact of these secondary patents is generally less significant than the expiry of the primary composition patent.
The timeline of key patent expiries has been as follows:
- United States: Primary patents expired around 2020.
- Europe: Expiry dates varied by country, with many primary patents lapsing in the early 2020s.
- Other Major Markets: Similar patent expiries have occurred in key global pharmaceutical markets.
The expiration of these patents has led to the introduction of generic Taclonex products. These generics are typically priced at a significant discount compared to the branded product, exerting downward pressure on the overall market price.
What is the Competitive Landscape for Taclonex?
The competitive landscape for Taclonex is evolving, marked by the presence of established generics and the introduction of newer therapeutic modalities.
Established Competition:
- Generic Taclonex: The most direct competition comes from generic versions of Taclonex. These products offer the same active ingredients at a lower price point, directly competing for market share among physicians and patients seeking cost-effective treatment options.
- Other Topical Combinations: Other fixed-dose topical combinations exist, offering different combinations of corticosteroids and vitamin D analogs or other active ingredients. Examples include topical calcineurin inhibitors used in conjunction with other therapies.
Emerging Competition:
- Novel Topical Agents: Pharmaceutical companies are developing and launching new topical agents with novel mechanisms of action for psoriasis. These may include PDE4 inhibitors, selective retinoids, or other anti-inflammatory compounds.
- Systemic Therapies (Oral and Injectable): The market for systemic psoriasis treatments, including oral small molecules (e.g., JAK inhibitors, PDE4 inhibitors) and biologic agents (e.g., TNF-alpha inhibitors, IL-17 inhibitors, IL-23 inhibitors), continues to grow. These therapies offer significant efficacy for moderate to severe disease and represent a major competitive threat, particularly for patients who may not respond adequately to topical treatments or who prefer systemic administration.
- Biosimil Products: While primarily relevant for biologic therapies, the concept of biosimil competition, if applicable to any component of future combination therapies or standalone treatments, could also emerge.
The competitive dynamic can be summarized as follows:
| Competitor Type | Impact on Taclonex | Example |
|---|---|---|
| Generic Taclonex | Direct price erosion and market share loss due to lower cost. | Multiple manufacturers offering generic betamethasone dipropionate/calcipotriene |
| Other Topical Rx | Potential shift in prescription patterns based on comparative efficacy, safety, or cost-effectiveness profiles. | Topical calcineurin inhibitors, other topical steroid/Vitamin D combinations |
| Systemic Therapies | Diversion of patients with moderate to severe psoriasis to more potent or convenient treatment options. | Biologics (e.g., Humira, Cosentyx), Oral small molecules (e.g., Xeljanz, Otezla) |
| Novel Topical Agents | Potential displacement of Taclonex by newer agents with improved efficacy, tolerability, or novel mechanisms of action. | Topical PDE4 inhibitors (e.g., crisaborole for atopic dermatitis, related pathways) |
What are the Historical and Projected Price Trends for Taclonex?
The pricing of Taclonex has historically been influenced by its branded status, research and development costs, and the prevailing market conditions for prescription dermatologicals. However, the introduction of generic competition has significantly altered these dynamics.
Historical Pricing:
- Branded Taclonex: Prior to patent expiry, the branded Taclonex commanded a premium price. Pricing was typically in the range of $200-$400 for a 60-gram tube, depending on the pharmacy, insurance coverage, and geographic region.
- Introduction of Generics: Following patent expiry, generic Taclonex products became available. Initial generic pricing was generally 20-40% lower than the branded product. Over time, as more generic manufacturers entered the market, prices for generic Taclonex have continued to decline.
Projected Pricing:
The price projections for Taclonex are subject to considerable downward pressure due to ongoing generic competition and the increasing availability of alternative treatments.
- Branded Taclonex: The price of branded Taclonex is expected to continue declining as market share is ceded to generics. Manufacturers may focus on specific patient populations or formulations to maintain some residual market value.
- Generic Taclonex: Prices for generic Taclonex are projected to stabilize but remain significantly lower than the historical branded price. We anticipate further incremental price reductions in the near term as competition intensifies. The average selling price for a generic 60-gram tube is likely to range between $50-$150.
Factors Influencing Future Pricing:
- Generic Market Penetration: The rate at which generic Taclonex captures market share will directly impact overall pricing.
- Payer Policies: Insurance companies and government payers play a crucial role. Increasingly restrictive formulary placement and prior authorization requirements for branded drugs can push patients towards generics.
- Advancements in Treatment: The introduction of highly effective and well-tolerated new therapies for psoriasis could reduce the overall demand for topical treatments like Taclonex, further impacting its pricing power.
- Manufacturer Strategies: Branded manufacturers may explore strategies such as offering patient assistance programs or developing enhanced formulations to differentiate their product, but the impact of these will be limited against established generic competition.
| Timeframe | Branded Taclonex Price Projection (60g tube) | Generic Taclonex Price Projection (60g tube) | Key Drivers |
|---|---|---|---|
| Current (2024) | $150 - $250 | $50 - $120 | Established generic competition, ongoing formulary pressures. |
| 1-2 Years (2025-26) | $100 - $200 | $45 - $100 | Increased generic penetration, potential new market entrants. |
| 3-5 Years (2027-29) | $75 - $150 | $40 - $90 | Maturation of generic market, impact of novel therapies. |
Note: Prices are approximate and can vary significantly based on region, payer contracts, and pharmacy. These are projected average selling prices (ASPs) before significant discounts or rebates.
What are the Regulatory Considerations for Taclonex and its Competitors?
Regulatory pathways and policies are pivotal in shaping the market for Taclonex and its generic and novel competitors.
For Branded Taclonex:
- Post-Marketing Surveillance: The FDA and other regulatory bodies continue to monitor the safety and efficacy of Taclonex through post-marketing surveillance programs. Any new safety signals or adverse events could lead to label changes, warnings, or even market withdrawal, though this is unlikely given its long history and established profile.
- Exclusivity Periods: The expiration of patent protection and any associated market exclusivity periods (e.g., New Chemical Entity exclusivity, Orphan Drug exclusivity, if applicable) directly allows for generic entry.
For Generic Taclonex:
- Abbreviated New Drug Application (ANDA): Generic manufacturers must file an ANDA with the FDA, demonstrating bioequivalence to the reference listed drug (RLD), which is branded Taclonex. This process is generally faster and less expensive than developing a new drug.
- Bioequivalence Standards: The FDA sets strict standards for bioequivalence, ensuring that the generic drug delivers the same amount of active ingredient into the bloodstream as the branded drug over the same time period.
- Manufacturing Standards: Generic manufacturers must adhere to Current Good Manufacturing Practices (cGMP) to ensure product quality, safety, and purity.
For Novel Therapies (Topical and Systemic):
- New Drug Application (NDA) / Biologics License Application (BLA): New chemical entities or novel biologics require a full NDA or BLA, involving extensive preclinical and clinical trials to demonstrate safety and efficacy.
- Expedited Review Pathways: The FDA offers expedited pathways (e.g., Fast Track, Breakthrough Therapy, Priority Review) for drugs that address unmet medical needs, potentially accelerating their approval.
- Orphan Drug Designation: If a novel therapy is developed for a rare form of psoriasis or a specific patient subpopulation, it may qualify for Orphan Drug Designation, providing market exclusivity and tax incentives.
- Biosimilar Pathways: For biologic drugs, the pathway for biosimilar approval involves demonstrating high similarity to an already approved biologic reference product, with no clinically meaningful differences in safety, purity, and potency.
Market Access and Reimbursement:
Regulatory approvals are only the first step; market access and reimbursement are critical.
- Formulary Placement: Payers (e.g., insurance companies, pharmacy benefit managers) determine whether a drug is included on their formularies, influencing physician prescribing and patient access. Generics are typically favored due to lower costs.
- Prior Authorization and Step Therapy: Payers may implement prior authorization requirements or step therapy protocols, requiring patients to try less expensive options (like generics) before approving more expensive branded or novel treatments.
- Value-Based Pricing: Increasingly, payers are exploring value-based pricing models where reimbursement is tied to patient outcomes. This trend could impact how all psoriasis treatments, including Taclonex and its alternatives, are priced and reimbursed in the future.
What are the Future Growth Prospects and Market Size for Taclonex?
The future growth prospects for Taclonex, as a branded product, are limited. Its primary market is likely to be sustained by its established generic versions and potentially by a smaller segment of patients who continue to benefit from or are accustomed to the branded product.
Market Size and Projections:
The overall market for topical psoriasis treatments is expected to experience modest growth, largely driven by the introduction of new, more efficacious topical agents and the continued need for treatment options. However, the growth of the specific Taclonex molecule (both branded and generic) will be constrained.
- Current Market Size (Topical Psoriasis Segment): The global market for topical psoriasis treatments is estimated to be in the low billions of dollars annually. Taclonex, as a branded product, has contributed significantly to this, but its individual market share has been declining since generic entry.
- Projected Market Size (Branded Taclonex): The market for branded Taclonex is expected to continue its decline. Projections indicate a decline of 5-10% annually over the next five years as generics capture more share and newer therapies gain traction.
- Projected Market Size (Generic Taclonex): The market for generic Taclonex is expected to grow by 2-5% annually, driven by increased adoption due to cost-effectiveness. However, this growth will be offset by the overall shift towards more advanced therapies for severe psoriasis.
- Overall Impact of Novel Therapies: The increasing adoption of biologic therapies and novel oral agents for moderate to severe psoriasis will temper the growth of the topical segment overall. Patients with more severe disease are increasingly migrating to these systemic options.
Factors Limiting Growth:
- Generic Erosion: The primary driver of limited growth for the branded product.
- Competition from Systemic Therapies: Biologics and oral small molecules offer superior efficacy for moderate to severe disease, drawing patients away from topical treatments.
- Development of New Topical Agents: Newer topical therapies with improved efficacy or tolerability profiles may displace Taclonex.
- Cost-Containment Pressures: Payers are increasingly scrutinizing the cost-effectiveness of all treatments, favoring generics and evidence-based therapies.
Opportunities:
- Cost-Effective Option: Generic Taclonex remains a vital, cost-effective option for patients with mild to moderate plaque psoriasis who do not require systemic therapy.
- Combination Therapy Potential: While Taclonex is a combination, there might be future research into optimizing its use or combining it with other novel topical agents, though this is speculative.
- Emerging Markets: In developing economies where cost is a more significant factor, generic Taclonex may see more sustained demand.
Key Takeaways
Taclonex, a long-standing topical therapy for plaque psoriasis, faces a dynamic market characterized by declining branded market share and increasing generic competition. The expiration of key patents has led to the widespread availability of lower-cost generic alternatives. While established efficacy and physician familiarity provide a baseline of demand, the emergence of novel topical agents and, more significantly, highly effective systemic therapies (biologics and oral small molecules) for moderate to severe psoriasis are redirecting patient care. Pricing projections indicate a continued downward trend for branded Taclonex, with generics stabilizing at significantly lower price points. Regulatory oversight ensures bioequivalence for generics, while new therapies navigate rigorous approval pathways. The future growth of the branded Taclonex is constrained, with its primary market segment being addressed by its cost-effective generic versions, serving as a valuable option for mild to moderate disease.
FAQs
1. Will branded Taclonex be discontinued?
Branded Taclonex is unlikely to be entirely discontinued in the near term. Pharmaceutical companies often continue to market branded versions of drugs even after patent expiry, targeting specific market segments or leveraging existing brand loyalty. However, its market share and pricing power will be significantly diminished due to generic competition.
2. How much cheaper are generic versions of Taclonex compared to the branded product?
Generic versions of Taclonex are typically 50-80% less expensive than the branded product, depending on the specific generic manufacturer, pharmacy, and negotiated payer rebates. This significant price difference is the primary driver for the shift from branded to generic Taclonex.
3. Are there any new topical treatments for psoriasis that directly compete with Taclonex?
Yes, there are new topical treatments for psoriasis with different mechanisms of action, such as topical PDE4 inhibitors and selective retinoids. These newer agents may offer improved efficacy, tolerability, or a different side effect profile compared to Taclonex, presenting direct competition.
4. What is the primary advantage of Taclonex over newer systemic treatments for psoriasis?
The primary advantage of Taclonex over newer systemic treatments (biologics and oral small molecules) is its topical administration and generally more localized side effect profile. It is also typically less expensive than systemic therapies, making it a suitable option for patients with milder disease or those who cannot tolerate or do not require systemic treatment.
5. What is the likelihood of Taclonex being replaced by a single, dominant new therapy for plaque psoriasis?
It is unlikely that Taclonex will be replaced by a single, dominant new therapy. Psoriasis treatment is becoming increasingly personalized. While novel systemic and topical agents are advancing treatment options for moderate to severe disease, Taclonex and its generics will likely remain a cost-effective and viable option for a significant segment of patients with mild to moderate plaque psoriasis. The market is moving towards a spectrum of treatment options rather than a single dominant therapy.
Citations
[1] L. Lebwohl, J. L. Tan, and M. R. Vender, "Calcipotriene with betamethasone dipropionate compared with calcipotriene, betamethasone dipropionate, and vehicle for the treatment of plaque psoriasis: a randomized trial," Journal of the American Academy of Dermatology, vol. 53, no. 3, pp. 487-492, 2005.
More… ↓
