STEGLATRO Drug Patent Profile
✉ Email this page to a colleague
When do Steglatro patents expire, and when can generic versions of Steglatro launch?
Steglatro is a drug marketed by Msd Sub Merck and is included in one NDA. There is one patent protecting this drug and one Paragraph IV challenge.
This drug has sixty-one patent family members in forty-nine countries.
The generic ingredient in STEGLATRO is ertugliflozin. One supplier is listed for this compound. Additional details are available on the ertugliflozin profile page.
DrugPatentWatch® Generic Entry Outlook for Steglatro
Steglatro was eligible for patent challenges on December 19, 2021.
There have been three patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for STEGLATRO?
- What are the global sales for STEGLATRO?
- What is Average Wholesale Price for STEGLATRO?
Summary for STEGLATRO
| International Patents: | 61 |
| US Patents: | 1 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 56 |
| Clinical Trials: | 4 |
| Patent Applications: | 1,067 |
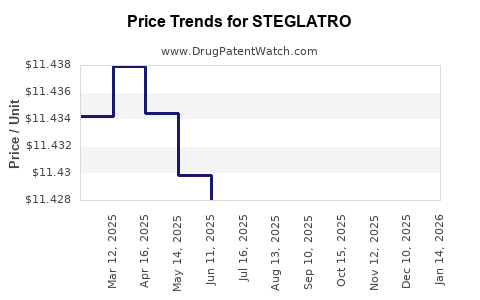
| Drug Prices: | Drug price information for STEGLATRO |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for STEGLATRO |
| What excipients (inactive ingredients) are in STEGLATRO? | STEGLATRO excipients list |
| DailyMed Link: | STEGLATRO at DailyMed |
Recent Clinical Trials for STEGLATRO
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Klinik Ottakring | Phase 3 |
| Wilhelminenspital Vienna | Phase 3 |
| Klinikum Wiener Neustadt | Phase 3 |
Paragraph IV (Patent) Challenges for STEGLATRO
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| STEGLATRO | Tablets | ertugliflozin | 5 mg and 15 mg | 209803 | 3 | 2021-12-20 |
US Patents and Regulatory Information for STEGLATRO
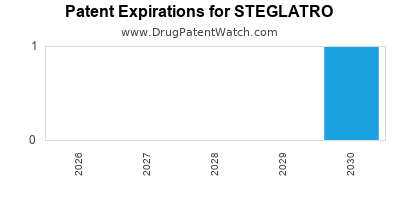
STEGLATRO is protected by one US patents.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Msd Sub Merck | STEGLATRO | ertugliflozin | TABLET;ORAL | 209803-001 | Dec 19, 2017 | RX | Yes | No | 8,080,580 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Msd Sub Merck | STEGLATRO | ertugliflozin | TABLET;ORAL | 209803-002 | Dec 19, 2017 | RX | Yes | Yes | 8,080,580 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for STEGLATRO
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Merck Sharp & Dohme B.V. | Steglatro | ertugliflozin | EMEA/H/C/004315Steglatro is indicated in adults aged 18 years and older with type 2 diabetes mellitus as an adjunct to diet and exercise to improve glycaemic control:as monotherapy in patients for whom the use of metformin is considered inappropriate due to intolerance or contraindications.in addition to other medicinal products for the treatment of diabetes. | Authorised | no | no | no | 2018-03-21 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for STEGLATRO
When does loss-of-exclusivity occur for STEGLATRO?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
African Regional IP Organization (ARIPO)
Patent: 28
Estimated Expiration: ⤷ Start Trial
Argentina
Patent: 3138
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 09286380
Estimated Expiration: ⤷ Start Trial
Austria
Patent: 40040
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 0918841
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 33795
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 11000394
Estimated Expiration: ⤷ Start Trial
China
Patent: 2149717
Estimated Expiration: ⤷ Start Trial
Patent: 3497199
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 41636
Estimated Expiration: ⤷ Start Trial
Costa Rica
Patent: 110077
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0120104
Estimated Expiration: ⤷ Start Trial
Cuba
Patent: 003
Estimated Expiration: ⤷ Start Trial
Patent: 110041
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 12497
Estimated Expiration: ⤷ Start Trial
Patent: 18024
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 34687
Estimated Expiration: ⤷ Start Trial
Dominican Republic
Patent: 011000058
Estimated Expiration: ⤷ Start Trial
Ecuador
Patent: 11010854
Estimated Expiration: ⤷ Start Trial
El Salvador
Patent: 11003842
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 8492
Estimated Expiration: ⤷ Start Trial
Patent: 1100266
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 34687
Estimated Expiration: ⤷ Start Trial
France
Patent: C1036
Estimated Expiration: ⤷ Start Trial
Georgia, Republic of
Patent: 0135803
Estimated Expiration: ⤷ Start Trial
Honduras
Patent: 09001652
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 56616
Estimated Expiration: ⤷ Start Trial
Patent: 93606
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 800031
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 1226
Estimated Expiration: ⤷ Start Trial
Patent: 6804
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 25322
Estimated Expiration: ⤷ Start Trial
Patent: 12500842
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 334687
Estimated Expiration: ⤷ Start Trial
Patent: 2018510
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 5418
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 11002166
Estimated Expiration: ⤷ Start Trial
Montenegro
Patent: 285
Estimated Expiration: ⤷ Start Trial
Morocco
Patent: 590
Estimated Expiration: ⤷ Start Trial
Netherlands
Patent: 0943
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 1027
Estimated Expiration: ⤷ Start Trial
Nicaragua
Patent: 1100043
Estimated Expiration: ⤷ Start Trial
Norway
Patent: 18019
Estimated Expiration: ⤷ Start Trial
Panama
Patent: 40801
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 110288
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 34687
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 34687
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 236
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 34687
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1101341
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1338540
Estimated Expiration: ⤷ Start Trial
Patent: 1446454
Estimated Expiration: ⤷ Start Trial
Patent: 110045093
Estimated Expiration: ⤷ Start Trial
Patent: 130116078
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 80408
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1014863
Estimated Expiration: ⤷ Start Trial
Patent: 87598
Estimated Expiration: ⤷ Start Trial
Tunisia
Patent: 11000066
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 3626
Estimated Expiration: ⤷ Start Trial
Uruguay
Patent: 073
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering STEGLATRO around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Israel | 236804 | ⤷ Start Trial | |
| Portugal | 2334687 | ⤷ Start Trial | |
| Chile | 2011000394 | ⤷ Start Trial | |
| Cyprus | 2018024 | ⤷ Start Trial | |
| Serbia | 52236 | ⤷ Start Trial | |
| New Zealand | 591027 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for STEGLATRO
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2334687 | 1890026-6 | Sweden | ⤷ Start Trial | PRODUCT NAME: ERTUGLIFLOZIN,OPTIONALLY IN THE FORM OF ERTUGLIFOZIN L- PYROGLUTAMIC ACID; FIRST MARKETING AUTHORIZATION NUMBER SE: EG EU/1/18/1267, 2018-03-23 1390008-9 1890026-6 2190052-7 (INKLUSIVE FOERLAENGNING 2191052-6) 2290013-8 2290007-0 SKYDDSTIDEN FOER SAMTLIGA DESSA TILLAEGGSSKYDD AER FOERLAENGD MED EN DAG, I ENLIGHET MED PATENT- OCH MARKNADSDOMSTOLENS BESLUT I PMAE 7804-24. DEN BESLUTADE SKYDDSTIDEN FRAMGAR AV SVENSK PATENTDATABAS. |
| 2334687 | 201840022 | Slovenia | ⤷ Start Trial | PRODUCT NAME: ERTUGLIFLOZIN, OPTIONALLY AS A CRYSTAL FORM, PARTICULARLY AS A CO-CRYSTAL WITH L-PYROGLUTAMIC ACID, AND SPECIFICALLY AS ERTUGLIFLOZIN L-PYROGLUTAMIC ACID; NATIONAL AUTHORISATION NUMBER: EU/1/18/1267/001-012; DATE OF NATIONAL AUTHORISATION: 20180321; AUTHORITY FOR NATIONAL AUTHORISATION: EU |
| 2334687 | C02334687/01 | Switzerland | ⤷ Start Trial | FORMER OWNER: PFIZER INC., US |
| 2334687 | SPC/GB18/026 | United Kingdom | ⤷ Start Trial | PRODUCT NAME: ERTUGLIFOZIN, OPTIONALLY AS A CRYSTAL FORM, PARTICULARLY AS A CO-CRYSTAL WITH L-PYROGLUTAMIC ACID, AND SPECIFICALLY AS ERTUGLIFOZIN L-PYROGLUTAMIC ACID; REGISTERED: UK EU/1/18/1267/001(NI) 20180321; UK EU/1/18/1267/002(NI) 20180321; UK EU/1/18/1267/003(NI) 20180321; UK EU/1/18/1267/004(NI) 20180321; UK EU/1/18/1267/005(NI) 20180321; UK EU/1/18/1267/006(NI) 20180321; UK PLGB 53095/0064 20180321; UK PLGB 53095/0065 20180321; UK EU/1/18/1267/007(NI) 20180321; UK EU/1/18/1267/008(NI) 20180321; UK EU/1/18/1267/009(NI) 20180321; UK EU/1/18/1267/010(NI) 20180321; UK EU/1/18/1267/011(NI) 20180321; UK EU/1/18/1267/012(NI) 20180321 |
| 2334687 | 392 18-2018 | Slovakia | ⤷ Start Trial | PRODUCT NAME: ERTUGLIFLOZIN VO VSETKYCH FORMACH CHRANENYCH ZAKLADNYM PATENTOM; REGISTRATION NO/DATE: EU/1/18/1267 20180323 |
| 2334687 | PA2018510 | Lithuania | ⤷ Start Trial | PRODUCT NAME: ERTUGLIFLOZINAS, PASIRINKTINAI KAIP KRISTALINE FORMA, YPAC KAIP KO-KRISTALAS SU L-PIROGLUTAMO RUGSTIMI, IR YPAC KAIP ERTUGLIFLOZINO L-PIROGLUTAMO RUGSTIS; REGISTRATION NO/DATE: EU/1/18/1267 20180321 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
STEGLATRO Market Dynamics and Financial Trajectory
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.