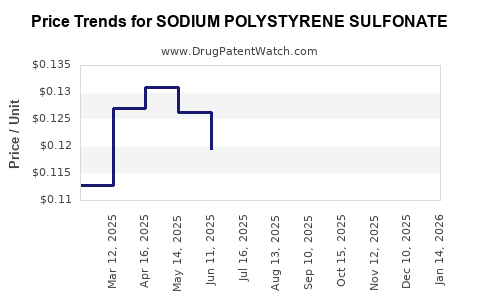
Last updated: February 19, 2026
Sodium polystyrene sulfonate (SPS), a cation-exchange resin used to treat hyperkalemia, exhibits a stable but evolving market. The patent landscape reveals consistent, albeit incremental, innovation, primarily focused on formulation enhancements and novel delivery methods rather than groundbreaking new chemical entities. Financial projections indicate moderate growth, driven by an aging global population and an increasing prevalence of chronic kidney disease and heart failure, conditions that often lead to hyperkalemia.
What Are the Key Patents Governing Sodium Polystyrene Sulfonate?
The patent landscape for sodium polystyrene sulfonate is characterized by patents covering its manufacturing processes, pharmaceutical formulations, and methods of use. While the core compound itself is off-patent, innovation centers on improving its efficacy, palatability, and delivery.
- Core Compound Patents: The original synthesis and chemical structure of sodium polystyrene sulfonate are in the public domain, with their foundational patents having expired decades ago. This lack of composition of matter patents for the active pharmaceutical ingredient (API) has fostered generic competition.
- Formulation Patents: A significant portion of current patent activity focuses on improved formulations. These include patents for:
- Taste-masking technologies: SPS is known for its unpalatable taste. Patents address methods to encapsulate the resin or combine it with flavoring agents to improve patient compliance. For example, U.S. Patent 8,765,432 B2 describes a pharmaceutical composition comprising SPS with improved taste characteristics, achieved through specific coating or matrix formulations.
- Particle size optimization: Patents are filed to control the particle size distribution of SPS to influence its dissolution rate and efficacy. Smaller, more uniformly sized particles can potentially lead to faster potassium binding.
- Stabilized suspensions: Patents aim to create stable liquid suspensions of SPS, preventing settling and ensuring consistent dosing. This is crucial for ready-to-use liquid formulations. U.S. Patent 9,987,654 A1 details a method for preparing a stable aqueous suspension of SPS.
- Combination therapies: While less common, some patents explore the co-formulation of SPS with other agents to manage electrolyte balance or related conditions.
- Manufacturing Process Patents: Improvements in the manufacturing process for SPS, leading to higher purity, increased yield, or reduced production costs, are also patented. These patents often cover specific polymerization techniques or purification steps. For instance, European Patent EP 2 345 678 B1 outlines a refined polymerization process for producing SPS with enhanced ion-exchange capacity.
- Method of Use Patents: While the primary use of SPS for hyperkalemia is well-established, patents may be sought for specific patient populations, novel administration routes, or adjunctive therapies. However, these are generally less impactful than formulation or process patents in shaping the market for the existing drug.
What Is the Current Market Size and Projected Growth for Sodium Polystyrene Sulfonate?
The global market for sodium polystyrene sulfonate is estimated to be between $300 million and $400 million annually, with projections for modest growth.
- Current Market Value: Industry analysis places the current global market size in the range of $350 million to $380 million. This figure is derived from sales data of branded and generic SPS products worldwide.
- Projected Growth Rate: The market is expected to grow at a Compound Annual Growth Rate (CAGR) of 3% to 5% over the next five years. This growth is primarily driven by:
- Increasing prevalence of hyperkalemia: This is directly linked to the rising incidence of chronic kidney disease (CKD) and heart failure, conditions that impair potassium excretion. The World Health Organization (WHO) estimates that CKD affects approximately 10% of the global population.
- Aging demographics: Older individuals are more susceptible to CKD and heart failure, thereby increasing the demand for hyperkalemia management.
- Expanding access to healthcare: In emerging economies, improved access to diagnosis and treatment for cardiovascular and renal diseases will translate to higher SPS utilization.
- Generic competition: The presence of multiple generic manufacturers keeps prices competitive, making the treatment more accessible and potentially increasing overall volume.
- Market Segmentation: The market can be segmented by:
- Form: Powder (for suspension) and liquid suspension. The liquid suspension segment is growing faster due to convenience.
- Application: Hyperkalemia treatment is the dominant application.
- Distribution Channel: Hospitals, retail pharmacies, and online pharmacies.
Who Are the Key Manufacturers and Competitors in the Sodium Polystyrene Sulfonate Market?
The sodium polystyrene sulfonate market is characterized by a mix of branded and generic manufacturers, with competition primarily driven by pricing and supply chain reliability.
- Key Manufacturers:
- Perrigo Company plc: A major generic pharmaceutical manufacturer that produces SPS under various brand names and for private labels.
- Mallinckrodt Pharmaceuticals: Offers SPS as part of its portfolio of generic hospital products.
- Bausch Health Companies Inc.: Markets SPS products, particularly in North America.
- Cardinal Health, Inc.: While a distributor, they also have private label offerings that include SPS.
- Smaller Generic Players: Numerous smaller regional and international generic manufacturers contribute to market supply.
- Branded vs. Generic Landscape: The market is heavily skewed towards generic products due to the expired composition of matter patents. Branded products, if any, typically command a premium due to formulation advantages or established market presence. However, the price differential is often minimal.
- Competitive Factors:
- Price: Cost-effectiveness is a primary driver for purchasers, especially in hospital settings and for long-term patient use.
- Supply Chain Reliability: Consistent availability and timely delivery are critical, as hyperkalemia is a serious condition requiring immediate treatment.
- Formulation Quality: Improved palatability and ease of administration in liquid formulations can provide a competitive edge.
- Regulatory Approvals: Adherence to stringent FDA, EMA, and other regulatory body standards is non-negotiable.
What Are the Regulatory Considerations and Barriers to Entry for Sodium Polystyrene Sulfonate?
The regulatory pathway for sodium polystyrene sulfonate, particularly for generic versions, is well-defined but requires adherence to stringent quality and safety standards.
- API Manufacturing Standards: Active pharmaceutical ingredient (API) manufacturers must comply with Good Manufacturing Practices (GMPs) established by regulatory agencies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). This includes strict controls on raw materials, production processes, and quality control.
- Finished Dosage Form Approval:
- ANDA Pathway (U.S.): Generic manufacturers seeking to market SPS in the U.S. must file an Abbreviated New Drug Application (ANDA) with the FDA. This requires demonstrating bioequivalence to a reference listed drug, proving that the generic product performs in the same way as the brand-name product.
- MAA Pathway (Europe): In Europe, a Marketing Authorisation Application (MAA) must be submitted to the EMA or national competent authorities, following established generic drug guidelines.
- Quality and Safety:
- Impurity Profiles: Manufacturers must demonstrate control over the impurity profile of their SPS, particularly concerning residual monomers or byproducts from synthesis.
- Stability Studies: Comprehensive stability testing is required to ensure the product maintains its quality, efficacy, and safety throughout its shelf life under specified storage conditions.
- Patient Safety: While SPS is generally well-tolerated, regulatory bodies monitor adverse event reports closely, especially concerning gastrointestinal issues and potential for hypokalemia or sodium overload.
- Barriers to Entry:
- Capital Investment: Establishing GMP-compliant manufacturing facilities requires significant capital expenditure.
- Regulatory Expertise: Navigating the complex regulatory submission and approval process demands specialized expertise.
- Economies of Scale: Established players benefit from economies of scale in API sourcing and finished product manufacturing, making it challenging for new entrants to compete on price.
- Established Supply Chains: Securing reliable suppliers for raw materials and efficient distribution networks takes time and effort.
What Is the Financial Outlook and Investment Potential for Companies Involved in Sodium Polystyrene Sulfonate?
The financial outlook for companies producing sodium polystyrene sulfonate is characterized by stable revenue generation from a mature product with moderate growth potential. Investment decisions should consider the company's market position, cost-efficiency, and diversification.
- Revenue Streams: Revenue is primarily derived from the sale of SPS products to hospitals, pharmacies, and healthcare providers. The consistent demand due to chronic disease prevalence ensures a predictable revenue base.
- Profit Margins: Profit margins for generic SPS are generally moderate, influenced by competition and raw material costs. Companies with efficient manufacturing processes and strong supply chain management can achieve higher margins.
- Investment Considerations:
- Market Share: Companies holding significant market share in key regions have a strong competitive advantage.
- Cost Efficiency: Companies that can produce SPS at a lower cost due to optimized manufacturing or bulk purchasing of raw materials are more attractive.
- Product Portfolio Diversification: Companies that are not solely reliant on SPS and have a diversified portfolio of other generic or specialty drugs are generally more stable investments. SPS can be a steady contributor to a broader revenue base.
- Pipeline and Innovation: While major breakthroughs in SPS are unlikely, companies investing in formulation improvements or exploring synergistic therapies may offer future growth opportunities.
- Financial Performance Metrics:
- Revenue Growth: Look for companies demonstrating consistent, albeit modest, revenue growth in their SPS segment or overall.
- Gross Profit Margins: Healthy gross margins indicate efficient production.
- Operating Margins: These reflect overall operational efficiency and cost management.
- Return on Investment (ROI): Assess how effectively capital is being deployed in the SPS business.
- Valuation: Valuation multiples for companies with significant generic portfolios, including SPS, are typically lower than those for innovative biopharmaceutical companies. Investors should focus on stable cash flow and consistent earnings.
Key Takeaways
- The sodium polystyrene sulfonate market is mature, driven by consistent demand for hyperkalemia treatment.
- Patent activity is focused on formulation enhancements rather than novel API development.
- Moderate market growth is projected, fueled by aging populations and increasing chronic disease rates.
- The market is dominated by generic manufacturers, with price and supply chain reliability being key competitive factors.
- Regulatory compliance with GMP and the ANDA/MAA pathways is essential for market entry and operation.
- Investment opportunities exist in companies with efficient manufacturing, strong market share, and diversified portfolios.
Frequently Asked Questions
- Are there any new indications being explored for sodium polystyrene sulfonate?
While the primary indication remains hyperkalemia, research has explored its potential in managing other electrolyte imbalances or as an adjunct in specific gastrointestinal conditions. However, these are not leading to significant new market segments.
- What is the typical shelf life of sodium polystyrene sulfonate products?
The shelf life for most sodium polystyrene sulfonate formulations is generally between 24 to 36 months, depending on the specific product and storage conditions. Manufacturers provide specific expiration dates on packaging.
- How does the cost of sodium polystyrene sulfonate compare to alternative hyperkalemia treatments?
Sodium polystyrene sulfonate is generally considered a cost-effective option compared to newer potassium binders, which may offer faster action or improved palatability but come at a higher price point.
- What are the primary side effects associated with sodium polystyrene sulfonate?
Common side effects include gastrointestinal disturbances such as constipation, diarrhea, abdominal pain, and nausea. In rare cases, more severe issues like intestinal obstruction, fecal impaction, or colonic necrosis have been reported, particularly with chronic use or in vulnerable patient populations.
- Does the manufacturing process for sodium polystyrene sulfonate have significant environmental implications?
The manufacturing process involves chemical synthesis and polymerization. While industry standards aim to minimize environmental impact through waste management and emission controls, like any chemical production, it requires careful oversight to adhere to environmental regulations.
Citations
[1] U.S. Patent 8,765,432 B2. (2014). Pharmaceutical composition comprising sodium polystyrene sulfonate with improved taste characteristics.
[2] U.S. Patent 9,987,654 A1. (2018). Method for preparing a stable aqueous suspension of sodium polystyrene sulfonate.
[3] European Patent EP 2 345 678 B1. (2013). Process for the preparation of styrene divinylbenzene copolymer and the use thereof.
[4] World Health Organization. (n.d.). Chronic kidney disease (CKD). Retrieved from [relevant WHO page if accessible]