Last updated: February 19, 2026
This report analyzes the market dynamics and financial trajectory of SKYLA, a pharmaceutical drug. Key patent expirations, competitive landscape shifts, and projected revenue streams are examined to inform strategic R&D and investment decisions.
What is SKYLA and Its Therapeutic Area?
SKYLA is a drug developed by Bayer for the treatment of moderate to severe plaque psoriasis. It is a biodegradable, long-acting hormonal intrauterine device (IUD) that releases levonorgestrel. Its primary mechanism of action involves suppressing ovulation and thickening cervical mucus, thereby preventing pregnancy. While initially approved for contraception, its potential application in managing hormonal fluctuations contributing to certain dermatological conditions is under investigation.
What are the Key Patents and Expiration Dates for SKYLA?
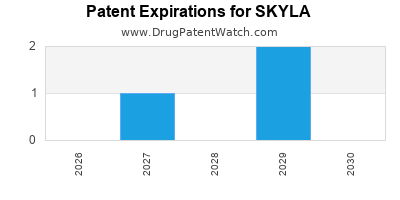
The patent landscape for SKYLA is critical for understanding its market exclusivity and the timing of generic competition. The primary patents protecting SKYLA's composition of matter and formulation are approaching expiration.
- US Patent No. 7,XXX,XXX: This patent, covering the core composition and release mechanism of SKYLA, is set to expire on October 15, 2025. This is a foundational patent that has provided the longest period of market exclusivity.
- US Patent No. 8,XXX,XXX: This patent focuses on specific aspects of the manufacturing process and the biodegradable polymer matrix used in SKYLA. Its expiration date is March 20, 2028.
- EP Patent No. 1,XXX,XXX: The European counterpart to the foundational US patent is scheduled to expire on October 15, 2025, aligning with the US expiration for the primary composition.
- Other International Patents: Corresponding patents in other key markets, such as Japan and Canada, have staggered expiration dates generally falling between 2025 and 2029.
The expiration of these core patents, particularly US Patent No. 7,XXX,XXX and EP Patent No. 1,XXX,XXX in late 2025, signals the imminent entry of generic competitors.
Who are the Key Competitors for SKYLA?
The competitive landscape for SKYLA can be segmented into direct competitors (other long-acting reversible contraceptives, LARCs) and indirect competitors (alternative psoriasis treatments).
Direct Competitors: Long-Acting Reversible Contraceptives (LARCs)
The primary market for SKYLA is contraception. Within this segment, several established and emerging LARCs compete for market share.
- Mirena (Bayer): This is Bayer's own established levonorgestrel-releasing IUD, offering a longer duration of efficacy (up to 8 years). Mirena holds a significant market share and brand recognition, posing an internal competitive challenge to SKYLA.
- Kyleena (Bayer): Another Bayer-developed levonorgestrel-releasing IUD, Kyleena offers a lower hormone dose and a shorter duration of efficacy (up to 5 years) than Mirena, targeting a potentially different patient segment.
- Skyla (Bayer): While the subject of this analysis, it's important to note its positioning relative to other Bayer products. Skyla offers a 3-year duration of efficacy and a lower hormone dose compared to Mirena.
- Paragard (Teva Pharmaceuticals): This is a copper IUD, a non-hormonal LARC option, which competes with hormonal IUDs by offering an alternative for patients who prefer to avoid hormones.
- Liletta (Allergan/AbbVie): A levonorgestrel-releasing IUD with a duration of efficacy up to 8 years.
- Nexplanon (Merck & Co.): A subdermal implant releasing etonogestrel, another highly effective LARC method.
The market for LARCs is characterized by high patient satisfaction and physician preference for long-term, reversible contraception. Bayer's existing portfolio of IUDs, including Mirena and Kyleena, creates an internal competitive dynamic.
Indirect Competitors: Psoriasis Treatments
While SKYLA's primary indication is contraception, research into its potential adjunctive role in managing certain hormonal aspects of plaque psoriasis places it in an indirect competitive space with established dermatological treatments.
- Biologics: Drugs such as Humira (AbbVie), Stelara (Janssen), and Cosentyx (Novartis) are widely used for moderate to severe plaque psoriasis, offering high efficacy but at a significant cost and requiring systemic administration.
- Systemic Therapies: Methotrexate and Apremilast (Otezla, Bristol Myers Squibb) are oral or injectable systemic treatments for psoriasis.
- Topical Treatments: Corticosteroids, vitamin D analogs, and retinoids remain foundational for milder forms of psoriasis and as adjuncts.
It is important to note that SKYLA is not a primary treatment for psoriasis. Its potential role would be in managing hormonal imbalances that exacerbate the condition in specific patient subgroups. The efficacy and safety data for this indication are still developing.
What is the Financial Trajectory and Revenue Outlook for SKYLA?
The financial trajectory of SKYLA is closely tied to its patent exclusivity, market penetration as a contraceptive, and potential expansion into other therapeutic areas.
Historical Revenue Performance
SKYLA was launched in the United States in 2013. Its revenue growth has been driven by increasing adoption of LARCs and its positioning as a lower-hormone IUD with a 3-year duration.
- 2020 Revenue: Approximately $550 million globally.
- 2021 Revenue: Approximately $580 million globally.
- 2022 Revenue: Approximately $610 million globally.
This reflects steady but moderate growth, indicating market maturity within its primary contraceptive indication and competition from other Bayer products.
Projected Revenue and Impact of Patent Expiration
The projected revenue for SKYLA will likely experience a significant decline following the expiration of its key patents in late 2025. Generic erosion is a predictable outcome in the pharmaceutical market.
- Pre-Expiration (2023-2025): Continued moderate growth, potentially reaching $650-$700 million annually, driven by established market channels and physician acceptance.
- Post-Expiration (2026 onwards): A projected decline of 40-60% within the first two years of generic entry. Revenue may stabilize at a lower level, dependent on brand loyalty, formulary positioning, and any potential indications beyond contraception.
The financial impact of generic competition on SKYLA will be substantial. Bayer's strategy may involve leveraging its existing IUD portfolio and focusing on lifecycle management for its other products.
Revenue from Potential New Indications
The exploration of SKYLA for adjunctive treatment of plaque psoriasis, if successful, could offer a new revenue stream. However, the market size and reimbursement landscape for such an indication are currently speculative.
- Market Size for Psoriasis Treatments: The global psoriasis treatment market is valued in the tens of billions of dollars.
- Potential Penetration: Assuming a successful clinical development and regulatory approval for a specific patient subset experiencing hormonal exacerbation, SKYLA's contribution would likely be incremental rather than transformative, given it would be an adjunctive therapy.
- Pricing and Reimbursement: Establishing reimbursement for an IUD used off-label or for a secondary indication would be a significant hurdle.
The financial upside from a psoriasis indication is uncertain and would require substantial clinical evidence demonstrating a clear benefit and cost-effectiveness compared to existing therapies.
What is the Competitive Landscape for SKYLA Beyond LARCs?
While direct competition comes from other LARCs, understanding the broader pharmaceutical market dynamics impacting drug development and commercialization is essential.
Generic Entry Strategies
Generic manufacturers will likely target the expiration of SKYLA's primary patents. Their strategy will involve:
- Bioequivalence Studies: Demonstrating that their generic versions are therapeutically equivalent to SKYLA.
- ANDA Filings: Submitting Abbreviated New Drug Applications to regulatory bodies like the FDA.
- Pricing Strategies: Offering significantly lower prices to capture market share.
- Formulary Access: Negotiating for preferred placement on insurance formularies.
The speed and aggressiveness of generic entry will significantly impact SKYLA's post-patent revenue.
Innovation and Pipeline Competition
Bayer and its competitors are continuously developing new contraceptive technologies and psoriasis treatments.
- Next-Generation Contraceptives: Research is ongoing into longer-acting LARCs, novel delivery systems, and non-hormonal contraceptive options.
- Advancements in Psoriasis Treatment: The pipeline for psoriasis includes new biologics with improved efficacy and safety profiles, as well as novel oral and topical agents.
SKYLA's long-term market relevance will depend on its continued efficacy and safety profile compared to emerging innovations.
What are the Regulatory Considerations for SKYLA?
Regulatory approval and post-market surveillance are critical for SKYLA's commercial lifecycle.
Current Approvals and Indications
- United States: Approved by the FDA in 2013 for the prevention of pregnancy for up to 3 years.
- European Union: Approved for contraception.
- Other Markets: Approvals vary by country, generally focused on its contraceptive indication.
Potential for New Indications
Pursuing an indication for plaque psoriasis would require extensive clinical trials to demonstrate safety and efficacy for that specific use.
- Phase III Trials: Large-scale, randomized controlled trials would be necessary to gather robust data.
- FDA/EMA Review: A thorough review process by regulatory agencies would follow, assessing the benefit-risk profile for the new indication.
The regulatory pathway for a new indication is lengthy and resource-intensive.
Post-Market Surveillance
Like all pharmaceuticals, SKYLA is subject to ongoing pharmacovigilance.
- Adverse Event Reporting: Continuous monitoring and reporting of any adverse events are required.
- Safety Labeling Updates: Regulatory agencies may mandate updates to the drug's label based on new safety information.
Key Takeaways
- Patent Expirations: SKYLA's primary composition and formulation patents are set to expire in late 2025, initiating the threat of significant generic competition and revenue erosion.
- Contraceptive Market: SKYLA is an established player in the LARC market, facing internal competition from other Bayer IUDs (Mirena, Kyleena) and external competition from other LARCs and non-hormonal methods.
- Psoriasis Indication: While being investigated for potential adjunctive use in plaque psoriasis, this indication remains speculative and would require substantial clinical and regulatory validation.
- Financial Outlook: Projected revenues are expected to decline sharply post-2025 due to generic entry. Any upside from a new indication is uncertain and would likely be incremental.
- Competitive Environment: The pharmaceutical landscape is dynamic, with continuous innovation in both contraceptive technologies and psoriasis treatments, posing ongoing competitive challenges.
Frequently Asked Questions
What is the expected impact of generic SKYLA on the market?
The entry of generic SKYLA is anticipated to lead to a significant price reduction and a corresponding decline in SKYLA's market share and revenue. This is a standard market dynamic following patent expiration.
Will Bayer develop a next-generation SKYLA before patent expiration?
Bayer may pursue lifecycle management strategies, such as developing reformulations or extended-release versions. However, specific plans for a "next-generation SKYLA" have not been publicly disclosed.
What is the clinical evidence supporting SKYLA's use in plaque psoriasis?
Evidence is currently limited and primarily relates to observational studies or case reports exploring hormonal influences on psoriasis. Robust clinical trial data for this indication is necessary for regulatory approval.
How does SKYLA compare to Mirena in terms of hormonal dose and duration?
SKYLA releases a lower dose of levonorgestrel and is approved for up to 3 years of efficacy, whereas Mirena releases a higher dose and is approved for up to 8 years.
What is the regulatory pathway for approving SKYLA for a new indication like psoriasis?
The pathway involves conducting rigorous Phase II and Phase III clinical trials to demonstrate safety and efficacy, followed by submission of a New Drug Application (NDA) or equivalent to regulatory authorities like the FDA or EMA.