Last updated: February 14, 2026
What Are the Market Dynamics for SEASONALE?
SEASONALE is a combined oral contraceptive (COC) containing ethinyl estradiol and levonorgestrel. It has historically targeted women seeking extended cycle contraception, reducing the frequency of menstrual periods from four to one per year.
Market segment:
The contraceptive market is driven by increasing demand for reliable, long-term birth control options. SEASONALE's unique selling proposition is the extended cycle, appealing to women desiring fewer periods.
Competitive landscape:
Major competitors include other extended-cycle pills like Seasonique and Amethia, as well as traditional 21-day pills. Non-hormonal and long-acting reversible contraceptives (LARCs), such as intrauterine devices (IUDs) and implants, also compete for market share.
Regulatory environment:
SEASONALE is approved in the US (FDA) and multiple markets globally. Its marketing depends on regulations surrounding contraceptive advertising, with increased scrutiny in some jurisdictions.
Consumer trends:
Demand for hormonal contraception remains stable or growing among women aged 18–35, driven by increased awareness and preferences for convenience. Shift towards non-invasive, reversible methods sustains the extended-cycle pill market.
Pricing and reimbursement:
SEASONALE typically retails between $40–$60 per month without insurance. Reimbursement coverage varies, impacting overall sales volume.
What Is the Financial Trajectory for SEASONALE?
Sales figures:
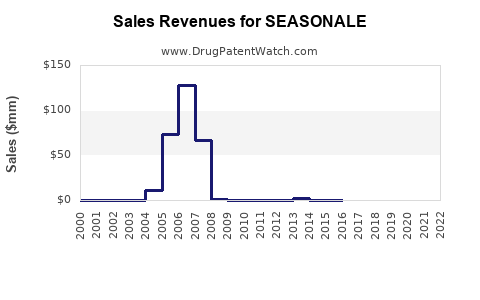
Historical annual sales have fluctuated, with estimates of approximately $120 million globally in recent years, declining gradually due to increasing competition and shifts toward LARCs.**
Market share:
Its share in the extended-cycle contraceptive market remains around 25%, with a declining trend as newer products gain traction.
Revenue drivers:
Sales depend on brand recognition, provider preferences, and insurance coverage. Higher penetration in developed markets sustains revenue, though growth is limited by patent expirations and the rise of generics.
Patent situation:
SEASONALE's patents expired in the US in 2015, leading to generic versions entering the market. This expanded access but decreased original product revenue.
Profitability:
Gross margins for SEASONALE are estimated at 60–70%, consistent with other branded oral contraceptives. Profitability faces downward pressure due to generic competition and decreasing market share.
Pipeline and future prospects:
No current pipeline developments are publicly disclosed. Future growth relies on brand loyalty, potential line extensions, and extended-cycle formulations gaining consumer preference.
Growth potential:
Limited, barring innovation or new indications. Market consolidation and generic entry constrain revenue growth. Strategic partnerships or licensing could stabilize revenues.
What Are the Key Drivers and Risks?
- Drivers: Continued consumer preference for extended-cycle contraceptives, brand loyalty, and insurance reimbursement.
- Risks: Patent expiration, entry of generics, competition from LARCs, regulatory modifications, and shifts in consumer preferences toward non-hormonal methods.
How Do Regulatory and Policy Changes Affect the Outlook?
Policy changes such as the U.S. Affordable Care Act mandates for contraceptive coverage help sustain demand. Conversely, restrictions or bans on hormonal contraceptives in certain regions could limit sales. The regulatory landscape remains dynamic, influencing market entry and product promotion strategies.
Summary of Market Data and Trends
| Aspect |
Data/Trend |
| Global sales (latest) |
~$120 million (estimated) |
| Market share (extended-cycle pills) |
~25% (declining trend) |
| Patents expired in the US |
2015 |
| Price range (retail) |
$40–$60/month |
| Reimbursement coverage |
Varies by country and insurer |
| Major competitors |
Seasonique, Amethia, LARCs |
Key Takeaways
SEASONALE operates within a stable yet mature contraceptive market, experiencing revenue decline due to patent expiration and generics. Its market share remains limited by competition from long-acting reversible contraceptives and newer formulations. The product's future depends on maintaining brand loyalty, healthcare policy changes, and potential product line extensions or innovations.
FAQs
1. How is SEASONALE affected by patent expiry?
Patent expiry in 2015 allowed generic manufacturers to produce lower-cost equivalents, reducing the original product’s market share and total revenue.
2. What are the main competitors to SEASONALE?
Seasonique, Amethia, and various LARCs like IUDs and implants compete in the extended-cycle and broader contraceptive market.
3. Is there potential for new formulations of SEASONALE?
No publicly disclosed pipeline exists; future growth hinges on brand loyalty and market dynamics rather than product innovation.
4. How does insurance coverage influence SEASONALE sales?
Insurance reimbursement significantly impacts affordability and access, affecting usage rates and sales volume. Greater coverage correlates with higher sales.
5. What regulatory factors could impact SEASONALE’s market?
Changes in contraceptive approval standards, restrictions on hormonal contraceptives, and evolving healthcare policies influence market eligibility and marketing strategies.
Citations
[1] MarketWatch, "Contraceptive Market Size and Trends," 2022.
[2] FDA, "Approval History for SEASONALE," 2003.
[3] IMS Health, "Pharmaceutical Sales Data," 2022.
[4] EuropaBio, "Contraceptive Patent Status," 2021.
[5] Statista, "Global Contraceptive Market Revenue," 2022.